Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00535
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
Antimicrobial peptides (AMP) 4 - Tobramycin conjugate
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
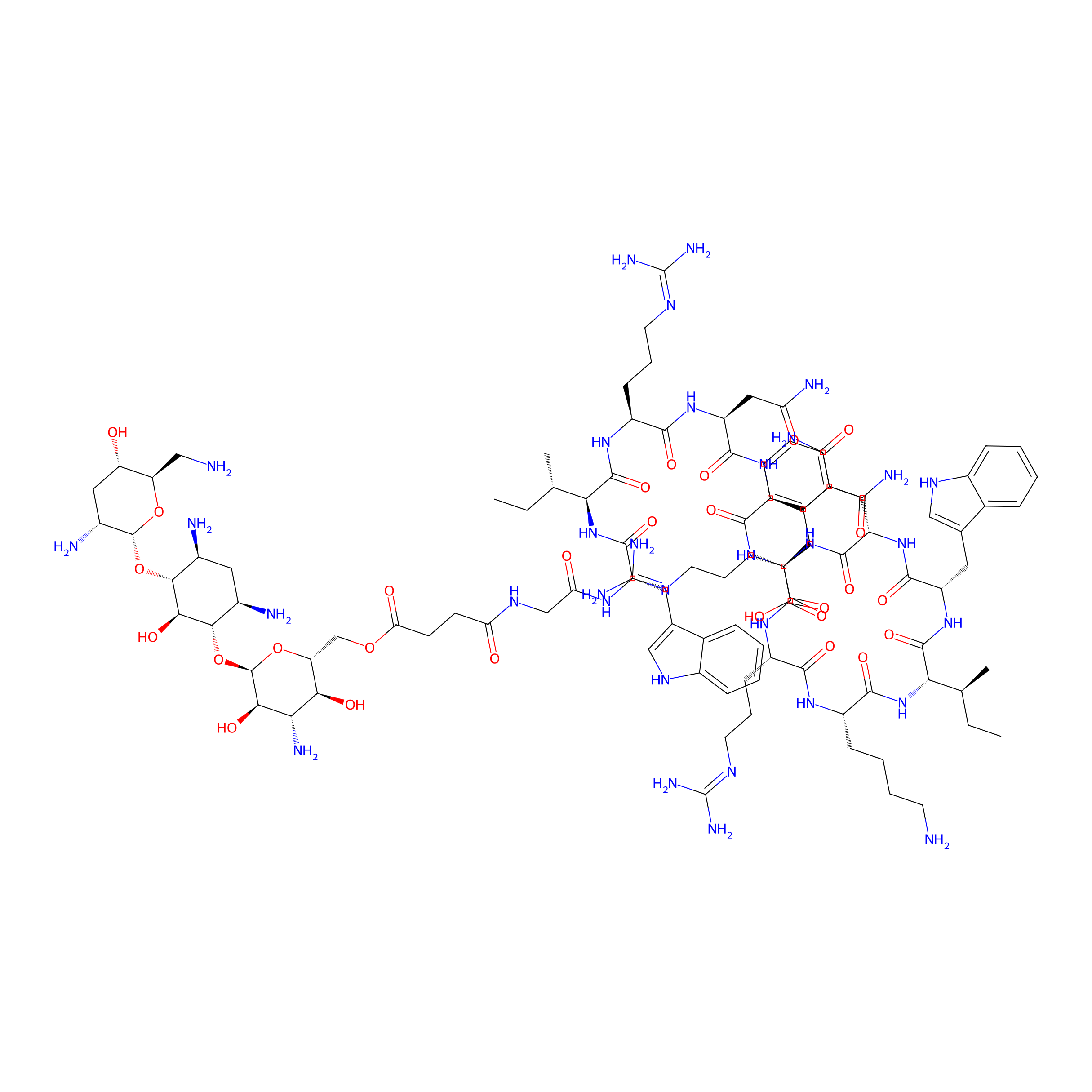
|
|||||
| Peptide Name |
Antimicrobial peptides (AMP) 4
|
Peptide Info | ||||
| Drug Name |
Tobramycin
|
Drug Info | ||||
| Therapeutic Target |
Bacterial 30S ribosomal RNA (Bact 30S rRNA)
|
Target Info | ||||
| Linker Name |
Succinic Acid
|
Linker Info | ||||
| Peptide Modified Type |
Cyclization modification
|
|||||
| Formula |
C105H165N33O28
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 2337.678 | ||||
| Lipid-water partition coefficient (xlogp) | -11.4672 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 35 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 36 | |||||
| Rotatable Bond Count (rotbonds) | 70 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 1000 μM | |||
| Administration Time | 1 h | ||||
| MOA of PDC |
We have synthesized a series of MAAPCs incorporating tobramycin with variations in the composition, sequence, and conjugation site of the peptide transporter. The MAAPCs exhibit good selectivity for bacterial cell membranes over mammalian cell membranes and do not induce any significant hemolysis of human red blood cells. MAAPCs exhibit better antibacterial activity against actively growing Gram-negative E. coli (MIC < 15 μM) than actively growing Gram-positive S. aureus (MIC < 25 μM). MAAPC01 exhibits the highest permeabilization of the outer membrane, with all other MAAPCs showing less permeation activity; tobramycin exhibits no outer membrane activity. MAAPC01 and MAAPC05 exhibit the highest inner membrane permeability, comparable to the control melittin. MAAPC02 and 03 exhibit less membrane activity, and MAAPC04 and tobramycin show negligible inner membrane activity. Higher levels of membrane activity correlate well with antimicrobial activity against persisters, where MAAPC01 and MAAPC05 show much better activity than tobramycin alone or MAAPC04.
Click to Show/Hide
|
||||
| Description |
One of the limitations to the clinical use of AMPs as antimicrobial agents is their hemolytic activity which is associated with increased toxicity. Thus, as a first assessment of the MAAPC toxicity, we investigated whether the conjugates may cause lysis of human red blood cells (hRBCs). Notably, all MAAPCs exhibited negligible hemolytic activity against hRBCs (HC50 > 500 μM), which is the first indicator of the MAAPC safety toward eukaryotic cells. Bacterial and animal cell membranes significantly differ in composition. Indeed, microbial cell surfaces contain more anionic lipids (overall negatively charged) whereas mammalian cell membranes have more lipids with neutral zwitterionic head groups (overall neutrally charged). The MAAPCs were designed to selectively discriminate between bacterial and mammalian cells. The selectivity indexes (SIs), defined as HC50/MIC, for E. coli and S. aureus demonstrated great selectivity of all MAAPCs to bacteria over hRBCs, which implies that the MAAPCs are effective against bacteria without causing harm to human cells. MAAPC04 displayed the highest bacterial selectivity (SI > 640 and SI > 160 against E. coli and S. aureus, respectively) likely due to its reduced hydrophobic content.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human red blood cells | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Minimum bactericidal concentration (MBC) |
3.1 ± 0.0 μM
|
|||
| Administration Time | 18 h | ||||
| MOA of PDC |
We have synthesized a series of MAAPCs incorporating tobramycin with variations in the composition, sequence, and conjugation site of the peptide transporter. The MAAPCs exhibit good selectivity for bacterial cell membranes over mammalian cell membranes and do not induce any significant hemolysis of human red blood cells. MAAPCs exhibit better antibacterial activity against actively growing Gram-negative E. coli (MIC < 15 μM) than actively growing Gram-positive S. aureus (MIC < 25 μM). MAAPC01 exhibits the highest permeabilization of the outer membrane, with all other MAAPCs showing less permeation activity; tobramycin exhibits no outer membrane activity. MAAPC01 and MAAPC05 exhibit the highest inner membrane permeability, comparable to the control melittin. MAAPC02 and 03 exhibit less membrane activity, and MAAPC04 and tobramycin show negligible inner membrane activity. Higher levels of membrane activity correlate well with antimicrobial activity against persisters, where MAAPC01 and MAAPC05 show much better activity than tobramycin alone or MAAPC04.
Click to Show/Hide
|
||||
| Description |
We first investigated the antimicrobial potency of these new MAAPCs by determining their minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) against Escherichia coli (E. coli MG1655) and Staphylococcus aureus (SA113) pathogens with a turbidity-based microdilution broth assay. Overall, all the MAAPCs display good antimicrobial activity and tend to be more selective to E. coli (MIC range 3.1-12.5 μM) over S. aureus (MIC range 12.5-25 μM), which follows the general observation that tobramycin is particularly active against Gram-negative organisms. Their MBC values are either equal to or slightly higher than their respective MIC values suggesting that the compounds not only are inhibiting bacterial growth but are also bactericidal. In contrast, the unconjugated peptides (P1-P4) exhibited no or little antimicrobial activity against E. coli and S. aureus. However, the MAAPCs did not show better activity than tobramycin, which is expected because our conjugates are not designed to target lab strains but instead to have high efficacy against clinically relevant pathogens such as persisters, resistant bacteria, and anaerobes that are impermeable to existing antibiotics. It should be noted that a mixture of the peptide itself with tobramycin does not enhance the antimicrobial activity compared to tobramycin alone, indicating there is no synergistic effect between the two entities when simply mixed. Whereas MAAPC05 showed similar activity against S. aureus in comparison to MAAPC01 (indicating that the terminus of conjugation did not greatly impact activity), it displayed a 2-fold increase of the MIC against E. coli. In contrast, MAAPC02, MAAPC03, and MAAPC04 displayed improved activity compared to MAAPC01. These results indicate the importance of the peptide transporter sequence as well as the conjugation site for tobramycin. Interestingly, MAAPC02 and MAAPC03 have nearly identical amino-acid composition as MAAPC01 (the only difference is a single glycine), but the amino acids are in a different sequence. In the MAAPC02 analog, the hydrophobic amino acids are clustered along one side of the -helical wheel of the peptide, which confers higher amphiphilic character and greater helical propensity. In the MAAPC03 analog, the peptide has the reversed sequence of MAAPC01. There are examples in the literature that the reversed analogs of antimicrobial peptides possess equal or enhanced antimicrobial activities. The amphiphilicity and peptide sequence orientation might play a role on the membrane-peptide interaction. However, further experiments are required to elucidate the reasons for this improved antibacterial activity.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 511145 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Minimum bactericidal concentration (MBC) |
20.8 ± 7.2 μM
|
|||
| Administration Time | 18 h | ||||
| MOA of PDC |
We have synthesized a series of MAAPCs incorporating tobramycin with variations in the composition, sequence, and conjugation site of the peptide transporter. The MAAPCs exhibit good selectivity for bacterial cell membranes over mammalian cell membranes and do not induce any significant hemolysis of human red blood cells. MAAPCs exhibit better antibacterial activity against actively growing Gram-negative E. coli (MIC < 15 μM) than actively growing Gram-positive S. aureus (MIC < 25 μM). MAAPC01 exhibits the highest permeabilization of the outer membrane, with all other MAAPCs showing less permeation activity; tobramycin exhibits no outer membrane activity. MAAPC01 and MAAPC05 exhibit the highest inner membrane permeability, comparable to the control melittin. MAAPC02 and 03 exhibit less membrane activity, and MAAPC04 and tobramycin show negligible inner membrane activity. Higher levels of membrane activity correlate well with antimicrobial activity against persisters, where MAAPC01 and MAAPC05 show much better activity than tobramycin alone or MAAPC04.
Click to Show/Hide
|
||||
| Description |
We first investigated the antimicrobial potency of these new MAAPCs by determining their minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) against Escherichia coli (E. coli MG1655) and Staphylococcus aureus (SA113) pathogens with a turbidity-based microdilution broth assay. Overall, all the MAAPCs display good antimicrobial activity and tend to be more selective to E. coli (MIC range 3.1-12.5 μM) over S. aureus (MIC range 12.5-25 μM), which follows the general observation that tobramycin is particularly active against Gram-negative organisms. Their MBC values are either equal to or slightly higher than their respective MIC values suggesting that the compounds not only are inhibiting bacterial growth but are also bactericidal. In contrast, the unconjugated peptides (P1-P4) exhibited no or little antimicrobial activity against E. coli and S. aureus. However, the MAAPCs did not show better activity than tobramycin, which is expected because our conjugates are not designed to target lab strains but instead to have high efficacy against clinically relevant pathogens such as persisters, resistant bacteria, and anaerobes that are impermeable to existing antibiotics. It should be noted that a mixture of the peptide itself with tobramycin does not enhance the antimicrobial activity compared to tobramycin alone, indicating there is no synergistic effect between the two entities when simply mixed. Whereas MAAPC05 showed similar activity against S. aureus in comparison to MAAPC01 (indicating that the terminus of conjugation did not greatly impact activity), it displayed a 2-fold increase of the MIC against E. coli. In contrast, MAAPC02, MAAPC03, and MAAPC04 displayed improved activity compared to MAAPC01. These results indicate the importance of the peptide transporter sequence as well as the conjugation site for tobramycin. Interestingly, MAAPC02 and MAAPC03 have nearly identical amino-acid composition as MAAPC01 (the only difference is a single glycine), but the amino acids are in a different sequence. In the MAAPC02 analog, the hydrophobic amino acids are clustered along one side of the -helical wheel of the peptide, which confers higher amphiphilic character and greater helical propensity. In the MAAPC03 analog, the peptide has the reversed sequence of MAAPC01. There are examples in the literature that the reversed analogs of antimicrobial peptides possess equal or enhanced antimicrobial activities. The amphiphilicity and peptide sequence orientation might play a role on the membrane-peptide interaction. However, further experiments are required to elucidate the reasons for this improved antibacterial activity.
Click to Show/Hide
|
||||
| In Vitro Model | Staphylococcus aureus infection | Staphylococcus aureus | 1280 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Minimum inhibitory concentration (MIC) |
3.1 ± 0.0 μM
|
|||
| Administration Time | 18 h | ||||
| MOA of PDC |
We have synthesized a series of MAAPCs incorporating tobramycin with variations in the composition, sequence, and conjugation site of the peptide transporter. The MAAPCs exhibit good selectivity for bacterial cell membranes over mammalian cell membranes and do not induce any significant hemolysis of human red blood cells. MAAPCs exhibit better antibacterial activity against actively growing Gram-negative E. coli (MIC < 15 μM) than actively growing Gram-positive S. aureus (MIC < 25 μM). MAAPC01 exhibits the highest permeabilization of the outer membrane, with all other MAAPCs showing less permeation activity; tobramycin exhibits no outer membrane activity. MAAPC01 and MAAPC05 exhibit the highest inner membrane permeability, comparable to the control melittin. MAAPC02 and 03 exhibit less membrane activity, and MAAPC04 and tobramycin show negligible inner membrane activity. Higher levels of membrane activity correlate well with antimicrobial activity against persisters, where MAAPC01 and MAAPC05 show much better activity than tobramycin alone or MAAPC04.
Click to Show/Hide
|
||||
| Description |
We first investigated the antimicrobial potency of these new MAAPCs by determining their minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) against Escherichia coli (E. coli MG1655) and Staphylococcus aureus (SA113) pathogens with a turbidity-based microdilution broth assay. Overall, all the MAAPCs display good antimicrobial activity and tend to be more selective to E. coli (MIC range 3.1-12.5 μM) over S. aureus (MIC range 12.5-25 μM), which follows the general observation that tobramycin is particularly active against Gram-negative organisms. Their MBC values are either equal to or slightly higher than their respective MIC values suggesting that the compounds not only are inhibiting bacterial growth but are also bactericidal. In contrast, the unconjugated peptides (P1-P4) exhibited no or little antimicrobial activity against E. coli and S. aureus. However, the MAAPCs did not show better activity than tobramycin, which is expected because our conjugates are not designed to target lab strains but instead to have high efficacy against clinically relevant pathogens such as persisters, resistant bacteria, and anaerobes that are impermeable to existing antibiotics. It should be noted that a mixture of the peptide itself with tobramycin does not enhance the antimicrobial activity compared to tobramycin alone, indicating there is no synergistic effect between the two entities when simply mixed. Whereas MAAPC05 showed similar activity against S. aureus in comparison to MAAPC01 (indicating that the terminus of conjugation did not greatly impact activity), it displayed a 2-fold increase of the MIC against E. coli. In contrast, MAAPC02, MAAPC03, and MAAPC04 displayed improved activity compared to MAAPC01. These results indicate the importance of the peptide transporter sequence as well as the conjugation site for tobramycin. Interestingly, MAAPC02 and MAAPC03 have nearly identical amino-acid composition as MAAPC01 (the only difference is a single glycine), but the amino acids are in a different sequence. In the MAAPC02 analog, the hydrophobic amino acids are clustered along one side of the -helical wheel of the peptide, which confers higher amphiphilic character and greater helical propensity. In the MAAPC03 analog, the peptide has the reversed sequence of MAAPC01. There are examples in the literature that the reversed analogs of antimicrobial peptides possess equal or enhanced antimicrobial activities. The amphiphilicity and peptide sequence orientation might play a role on the membrane-peptide interaction. However, further experiments are required to elucidate the reasons for this improved antibacterial activity.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 511145 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Minimum inhibitory concentration (MIC) |
12.5 ± 0.0 μM
|
|||
| Administration Time | 18 h | ||||
| MOA of PDC |
We have synthesized a series of MAAPCs incorporating tobramycin with variations in the composition, sequence, and conjugation site of the peptide transporter. The MAAPCs exhibit good selectivity for bacterial cell membranes over mammalian cell membranes and do not induce any significant hemolysis of human red blood cells. MAAPCs exhibit better antibacterial activity against actively growing Gram-negative E. coli (MIC < 15 μM) than actively growing Gram-positive S. aureus (MIC < 25 μM). MAAPC01 exhibits the highest permeabilization of the outer membrane, with all other MAAPCs showing less permeation activity; tobramycin exhibits no outer membrane activity. MAAPC01 and MAAPC05 exhibit the highest inner membrane permeability, comparable to the control melittin. MAAPC02 and 03 exhibit less membrane activity, and MAAPC04 and tobramycin show negligible inner membrane activity. Higher levels of membrane activity correlate well with antimicrobial activity against persisters, where MAAPC01 and MAAPC05 show much better activity than tobramycin alone or MAAPC04.
Click to Show/Hide
|
||||
| Description |
We first investigated the antimicrobial potency of these new MAAPCs by determining their minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) against Escherichia coli (E. coli MG1655) and Staphylococcus aureus (SA113) pathogens with a turbidity-based microdilution broth assay. Overall, all the MAAPCs display good antimicrobial activity and tend to be more selective to E. coli (MIC range 3.1-12.5 μM) over S. aureus (MIC range 12.5-25 μM), which follows the general observation that tobramycin is particularly active against Gram-negative organisms. Their MBC values are either equal to or slightly higher than their respective MIC values suggesting that the compounds not only are inhibiting bacterial growth but are also bactericidal. In contrast, the unconjugated peptides (P1-P4) exhibited no or little antimicrobial activity against E. coli and S. aureus. However, the MAAPCs did not show better activity than tobramycin, which is expected because our conjugates are not designed to target lab strains but instead to have high efficacy against clinically relevant pathogens such as persisters, resistant bacteria, and anaerobes that are impermeable to existing antibiotics. It should be noted that a mixture of the peptide itself with tobramycin does not enhance the antimicrobial activity compared to tobramycin alone, indicating there is no synergistic effect between the two entities when simply mixed. Whereas MAAPC05 showed similar activity against S. aureus in comparison to MAAPC01 (indicating that the terminus of conjugation did not greatly impact activity), it displayed a 2-fold increase of the MIC against E. coli. In contrast, MAAPC02, MAAPC03, and MAAPC04 displayed improved activity compared to MAAPC01. These results indicate the importance of the peptide transporter sequence as well as the conjugation site for tobramycin. Interestingly, MAAPC02 and MAAPC03 have nearly identical amino-acid composition as MAAPC01 (the only difference is a single glycine), but the amino acids are in a different sequence. In the MAAPC02 analog, the hydrophobic amino acids are clustered along one side of the -helical wheel of the peptide, which confers higher amphiphilic character and greater helical propensity. In the MAAPC03 analog, the peptide has the reversed sequence of MAAPC01. There are examples in the literature that the reversed analogs of antimicrobial peptides possess equal or enhanced antimicrobial activities. The amphiphilicity and peptide sequence orientation might play a role on the membrane-peptide interaction. However, further experiments are required to elucidate the reasons for this improved antibacterial activity.
Click to Show/Hide
|
||||
| In Vitro Model | Staphylococcus aureus infection | Staphylococcus aureus | 1280 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Selectivity index | > 80 | |||
| MOA of PDC |
We have synthesized a series of MAAPCs incorporating tobramycin with variations in the composition, sequence, and conjugation site of the peptide transporter. The MAAPCs exhibit good selectivity for bacterial cell membranes over mammalian cell membranes and do not induce any significant hemolysis of human red blood cells. MAAPCs exhibit better antibacterial activity against actively growing Gram-negative E. coli (MIC < 15 μM) than actively growing Gram-positive S. aureus (MIC < 25 μM). MAAPC01 exhibits the highest permeabilization of the outer membrane, with all other MAAPCs showing less permeation activity; tobramycin exhibits no outer membrane activity. MAAPC01 and MAAPC05 exhibit the highest inner membrane permeability, comparable to the control melittin. MAAPC02 and 03 exhibit less membrane activity, and MAAPC04 and tobramycin show negligible inner membrane activity. Higher levels of membrane activity correlate well with antimicrobial activity against persisters, where MAAPC01 and MAAPC05 show much better activity than tobramycin alone or MAAPC04.
Click to Show/Hide
|
||||
| Description |
One of the limitations to the clinical use of AMPs as antimicrobial agents is their hemolytic activity which is associated with increased toxicity. Thus, as a first assessment of the MAAPC toxicity, we investigated whether the conjugates may cause lysis of human red blood cells (hRBCs). Notably, all MAAPCs exhibited negligible hemolytic activity against hRBCs (HC50 > 500 μM), which is the first indicator of the MAAPC safety toward eukaryotic cells. Bacterial and animal cell membranes significantly differ in composition. Indeed, microbial cell surfaces contain more anionic lipids (overall negatively charged) whereas mammalian cell membranes have more lipids with neutral zwitterionic head groups (overall neutrally charged). The MAAPCs were designed to selectively discriminate between bacterial and mammalian cells. The selectivity indexes (SIs), defined as HC50/MIC, for E. coli and S. aureus demonstrated great selectivity of all MAAPCs to bacteria over hRBCs, which implies that the MAAPCs are effective against bacteria without causing harm to human cells. MAAPC04 displayed the highest bacterial selectivity (SI > 640 and SI > 160 against E. coli and S. aureus, respectively) likely due to its reduced hydrophobic content.
Click to Show/Hide
|
||||
| In Vitro Model | Staphylococcus aureus infection | Staphylococcus aureus | 1280 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Selectivity index | > 320 | |||
| MOA of PDC |
We have synthesized a series of MAAPCs incorporating tobramycin with variations in the composition, sequence, and conjugation site of the peptide transporter. The MAAPCs exhibit good selectivity for bacterial cell membranes over mammalian cell membranes and do not induce any significant hemolysis of human red blood cells. MAAPCs exhibit better antibacterial activity against actively growing Gram-negative E. coli (MIC < 15 μM) than actively growing Gram-positive S. aureus (MIC < 25 μM). MAAPC01 exhibits the highest permeabilization of the outer membrane, with all other MAAPCs showing less permeation activity; tobramycin exhibits no outer membrane activity. MAAPC01 and MAAPC05 exhibit the highest inner membrane permeability, comparable to the control melittin. MAAPC02 and 03 exhibit less membrane activity, and MAAPC04 and tobramycin show negligible inner membrane activity. Higher levels of membrane activity correlate well with antimicrobial activity against persisters, where MAAPC01 and MAAPC05 show much better activity than tobramycin alone or MAAPC04.
Click to Show/Hide
|
||||
| Description |
One of the limitations to the clinical use of AMPs as antimicrobial agents is their hemolytic activity which is associated with increased toxicity. Thus, as a first assessment of the MAAPC toxicity, we investigated whether the conjugates may cause lysis of human red blood cells (hRBCs). Notably, all MAAPCs exhibited negligible hemolytic activity against hRBCs (HC50 > 500 μM), which is the first indicator of the MAAPC safety toward eukaryotic cells. Bacterial and animal cell membranes significantly differ in composition. Indeed, microbial cell surfaces contain more anionic lipids (overall negatively charged) whereas mammalian cell membranes have more lipids with neutral zwitterionic head groups (overall neutrally charged). The MAAPCs were designed to selectively discriminate between bacterial and mammalian cells. The selectivity indexes (SIs), defined as HC50/MIC, for E. coli and S. aureus demonstrated great selectivity of all MAAPCs to bacteria over hRBCs, which implies that the MAAPCs are effective against bacteria without causing harm to human cells. MAAPC04 displayed the highest bacterial selectivity (SI > 640 and SI > 160 against E. coli and S. aureus, respectively) likely due to its reduced hydrophobic content.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 511145 | ||
References
