Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_02034
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
R-C12-1
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
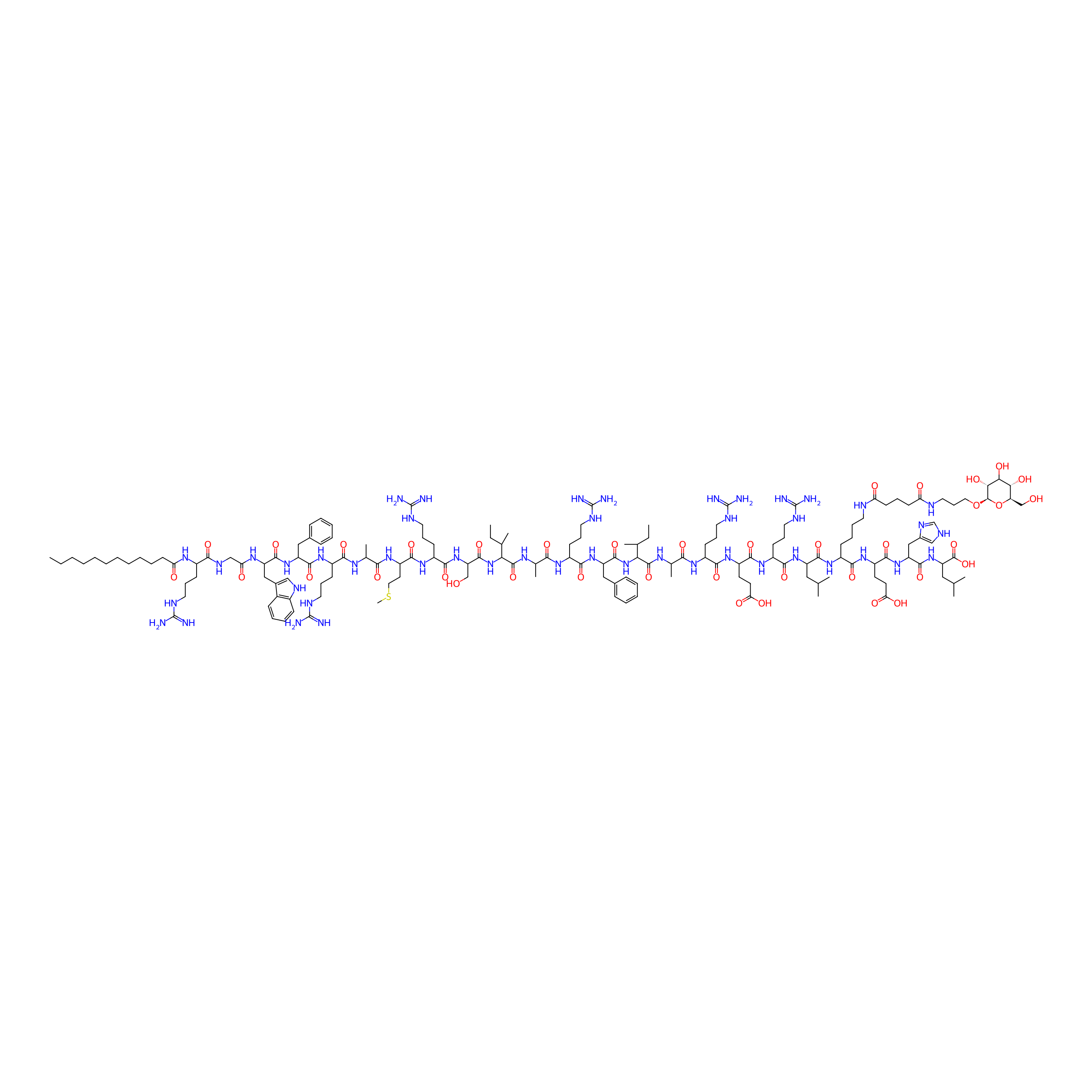
|
|||||
| Peptide Name |
R-C12
|
Peptide Info | ||||
| Drug Name |
D-Glucose
|
Drug Info | ||||
| Linker Name |
Glutaric acid
|
Linker Info | ||||
| Peptide Modified Type |
The modification of binding with chemical molecules
|
|||||
| Modified Segment |
Dodecanoic acid
|
|||||
| Ternimal Modification |
N-terminus
|
|||||
| Formula |
C156H256N46O38S
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 3416.115 | ||||
| Lipid-water partition coefficient (xlogp) | -6.97278 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 53 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 43 | |||||
| Rotatable Bond Count (rotbonds) | 121 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
3.5 μM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In this study, we would investigate the co-modification of fatty acids and monosaccharides in anticancer peptides, as we expect to optimize the selectivity and cytotoxicity of the peptide at the same time. Based on our previous study, the lipopeptide R-C12 was selected as a template to perform glucose derivative modification at different positions as we supposed that the glucose derivative modification position in R-C12 might be a crucial factor in its selectivity and activity optimization. The glucose derivative was covalently coupled to different sites in R-C12 through its -amino group of a Lys residue, which was obtained by replacing the Arg residue. R-C12 contains seven Arg residues, accordingly, seven glycolipid peptides were designed and synthesized, which were named R-C12-1, R-C12-2, R-C12-3, R-C12-4, R-C12-5, R-C12-6, and R-C12-7. Various physiochemical properties including the hydrodynamic size, ζ-potential, secondary structure, and hydrophobicity were determined to explain the different cytotoxicity and selectivity of these glycolipid peptides. On the other hand, the strength of the binding between glycolipid peptides and the cancer cells mediated by GLUT1 has also been explored. The most optimized glycolipid peptide R-C12-4 demonstrated significant cytotoxicity and antimetastasis in vitro and in vivo, which indicated that it may be a good lead for anticancer drug development.
Click to Show/Hide
|
||||
| Description |
Murine melanoma cells (B16-F10) were chosen as the object for evaluating the cytotoxicity of the seven glycolipid peptides and R-C12 by the CCK-8 assay. Based on our previous study, the HEK-293T cell line was also chosen as the noncancer cell line to evaluate the selectivity of glycolipid peptides. IC50 represents the concentration of glycolipid peptides and R-C12 that induce 50% cell death. As shown in Figure 1A, modification of the glucose derivative at different sites of R-C12 results in different cytotoxicity profiles. According to the IC50 values, for B16-F10 cells, the cytotoxic activity of peptides was arranged as follows R-C12-4 > R-C12-1 ≈ R-C12-2 ≈ R-C12-3 ≈ R-C12 > R-C12-5 > R-C12-6 > R-C12-7, in which R-C12-4 demonstrated the highest cytotoxicity on B16-F10 cells (IC50 = 1.9 μM)
Click to Show/Hide
|
||||
| In Vitro Model | Mouse melanoma | B16-F10 cell | CVCL_0159 | ||
References
