Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_02075
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
DPV1047-E-Dox
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
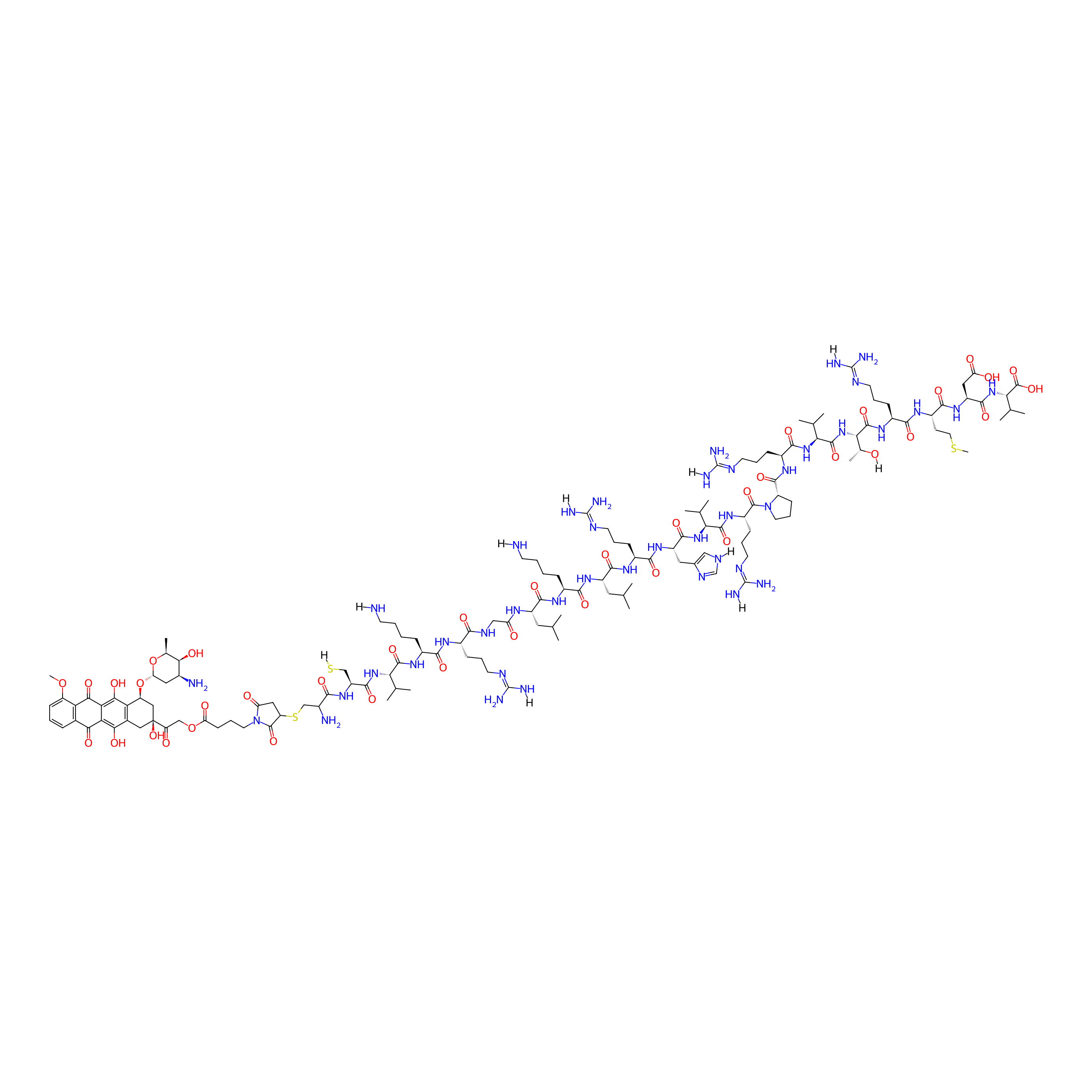
|
|||||
| Peptide Name |
DPV1047
|
Peptide Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
4-(3-((2-Amino-2-carboxyethyl)thio)-2,5-dioxopyrrolidin-1-yl)butanoic acid
|
Linker Info | ||||
| Formula |
C141H228N42O39S3
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 3231.831 | ||||
| Lipid-water partition coefficient (xlogp) | -10.815 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 42 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 50 | |||||
| Rotatable Bond Count (rotbonds) | 102 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
4 μM
|
|||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
In the case of Vp followed by DPV1047-E-Dox (8a) treatment, only a moderate increase in sensitivity was observed, with only a 3.6-5-fold increase in activity. The difference between doxorubicin (10) and DPV1047-E-Dox (8a) cytotoxicity following Vp treatment suggests that DPV1047-E-Dox (8a) is a poor substrate for Pgp-mediated cell extrusion, which could result in an increase in the intracellular concentration of the therapeutic molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Colon carcinoma | HCT 116 cell | CVCL_0291 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
5 μM
|
|||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
In the case of Vp followed by DPV1047-E-Dox (8a) treatment, only a moderate increase in sensitivity was observed, with only a 3.6-5-fold increase in activity. The difference between doxorubicin (10) and DPV1047-E-Dox (8a) cytotoxicity following Vp treatment suggests that DPV1047-E-Dox (8a) is a poor substrate for Pgp-mediated cell extrusion, which could result in an increase in the intracellular concentration of the therapeutic molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Colon carcinoma | HCT 116 cell | CVCL_0291 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
6 μM
|
|||
| Administration Time | Three injections per week for 3 weeks | ||||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
The Vectocell peptides alone showed no cytotoxic activity (data not shown). The Vectocell-doxorubicin conjugates 8a-c showed no loss of cytotoxicity compared to free doxorubicin. Compounds 9b and 9c showed a loss of activity in the HCT116 model but not in the MCF-7/6 cell model, compared to free doxorubicin (10). In contrast, the compounds 12a and 12b showed a significant loss of cytotoxicity in both tumor cell lines (loss of 1-2 logs in IC50). The in vitro data, therefore, suggest that the ester (8a-c) and thioether (9b and 9c) bonds are optimal for Vectocell-doxorubicin conjugate activity and that conjugation to the 3 position via an amide bond (12a and 12b) partially inactivates doxorubicin.
Click to Show/Hide
|
||||
| In Vitro Model | Colon carcinoma | HCT 116 cell | CVCL_0291 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
11 μM
|
|||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
In the case of Vp followed by DPV1047-E-Dox (8a) treatment, only a moderate increase in sensitivity was observed, with only a 3.6-5-fold increase in activity. The difference between doxorubicin (10) and DPV1047-E-Dox (8a) cytotoxicity following Vp treatment suggests that DPV1047-E-Dox (8a) is a poor substrate for Pgp-mediated cell extrusion, which could result in an increase in the intracellular concentration of the therapeutic molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Colon carcinoma | HCT 116 cell | CVCL_0291 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
22 μM
|
|||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
In the case of Vp followed by DPV1047-E-Dox (8a) treatment, only a moderate increase in sensitivity was observed, with only a 3.6-5-fold increase in activity. The difference between doxorubicin (10) and DPV1047-E-Dox (8a) cytotoxicity following Vp treatment suggests that DPV1047-E-Dox (8a) is a poor substrate for Pgp-mediated cell extrusion, which could result in an increase in the intracellular concentration of the therapeutic molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Uterine sarcoma | MES-SA/Dx5 cell | CVCL_2598 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
29 μM
|
|||
| Administration Time | Three injections per week for 3 weeks | ||||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
The Vectocell peptides alone showed no cytotoxic activity (data not shown). The Vectocell-doxorubicin conjugates 8a-c showed no loss of cytotoxicity compared to free doxorubicin. Compounds 9b and 9c showed a loss of activity in the HCT116 model but not in the MCF-7/6 cell model, compared to free doxorubicin (10). In contrast, the compounds 12a and 12b showed a significant loss of cytotoxicity in both tumor cell lines (loss of 1-2 logs in IC50). The in vitro data, therefore, suggest that the ester (8a-c) and thioether (9b and 9c) bonds are optimal for Vectocell-doxorubicin conjugate activity and that conjugation to the 3 position via an amide bond (12a and 12b) partially inactivates doxorubicin.
Click to Show/Hide
|
||||
| In Vitro Model | Breast cancer | MCF-7/6 cell | CVCL_W972 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
50 μM
|
|||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
In these resistant cell lines, DPV1047-E-Dox (8a) always showed better antiproliferative activity than doxorubicin. In MCF7-Adr and MES-SA-dx5 cells, which express high levels of Pgp, the enhanced efficacy of DPV1047-E-Dox (8a) was highly significant compared to that of doxorubicin alone (p < 0.001).
|
||||
| In Vitro Model | Colon adenocarcinoma | HCT 15 cell | CVCL_0292 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
50 μM
|
|||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
In the case of Vp followed by DPV1047-E-Dox (8a) treatment, only a moderate increase in sensitivity was observed, with only a 3.6-5-fold increase in activity. The difference between doxorubicin (10) and DPV1047-E-Dox (8a) cytotoxicity following Vp treatment suggests that DPV1047-E-Dox (8a) is a poor substrate for Pgp-mediated cell extrusion, which could result in an increase in the intracellular concentration of the therapeutic molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Colon carcinoma | HCT 116 cell | CVCL_0291 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
52 μM
|
|||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
In the case of Vp followed by DPV1047-E-Dox (8a) treatment, only a moderate increase in sensitivity was observed, with only a 3.6-5-fold increase in activity. The difference between doxorubicin (10) and DPV1047-E-Dox (8a) cytotoxicity following Vp treatment suggests that DPV1047-E-Dox (8a) is a poor substrate for Pgp-mediated cell extrusion, which could result in an increase in the intracellular concentration of the therapeutic molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7/ADR cell | CVCL_0031 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
79 μM
|
|||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
In the case of Vp followed by DPV1047-E-Dox (8a) treatment, only a moderate increase in sensitivity was observed, with only a 3.6-5-fold increase in activity. The difference between doxorubicin (10) and DPV1047-E-Dox (8a) cytotoxicity following Vp treatment suggests that DPV1047-E-Dox (8a) is a poor substrate for Pgp-mediated cell extrusion, which could result in an increase in the intracellular concentration of the therapeutic molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Uterine sarcoma | MES-SA/Dx5 cell | CVCL_2598 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
81 μM
|
|||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
In these resistant cell lines, DPV1047-E-Dox (8a) always showed better antiproliferative activity than doxorubicin. In MCF7-Adr and MES-SA-dx5 cells, which express high levels of Pgp, the enhanced efficacy of DPV1047-E-Dox (8a) was highly significant compared to that of doxorubicin alone (p < 0.001).
|
||||
| In Vitro Model | Uterine sarcoma | MES-SA/Dx5 cell | CVCL_2598 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
133 μM
|
|||
| Administration Time | Three injections per week for 3 weeks | ||||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
These experiments showed that it is possible to overcome the doxorubicin-resistant phenotype by conjugation of doxorubicin to Vectocell peptides. Vectocell peptides DPV1047 (8a and 12a) and DPV10 (8b and 9b) are able to inhibit the doxorubicin- resistant phenotype of MCF7-Adr cells. However, the compounds 8a and 9b exhibited the greatest cytotoxic activity in the doxorubicin-resistant cell model, although 9b showed a loss of cytotoxic activity in the HCT116 in vitro model (Table 1). The in vitro data therefore suggested that the optimal conjugate for both doxorubicin-sensitive and -resistant models was 8a.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7/ADR cell | CVCL_0031 | ||
| Experiment 13 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
260 μM
|
|||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
In these resistant cell lines, DPV1047-E-Dox (8a) always showed better antiproliferative activity than doxorubicin. In MCF7-Adr and MES-SA-dx5 cells, which express high levels of Pgp, the enhanced efficacy of DPV1047-E-Dox (8a) was highly significant compared to that of doxorubicin alone (p < 0.001).
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7/ADR cell | CVCL_0031 | ||
| Experiment 14 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
260 μM
|
|||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
In the case of Vp followed by DPV1047-E-Dox (8a) treatment, only a moderate increase in sensitivity was observed, with only a 3.6-5-fold increase in activity. The difference between doxorubicin (10) and DPV1047-E-Dox (8a) cytotoxicity following Vp treatment suggests that DPV1047-E-Dox (8a) is a poor substrate for Pgp-mediated cell extrusion, which could result in an increase in the intracellular concentration of the therapeutic molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7/ADR cell | CVCL_0031 | ||
| Experiment 15 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Relative tumor volume |
29.50%
|
|||
| Administration Time | Three injections per week for 3 weeks | ||||
| MOA of PDC |
The conjugation of Vectocell peptides to cytotoxic molecules can modify the in vivo distribution of the therapeutic molecules, improving pharmacokinetic properties and/or reducing systemic toxicity by driving tissue and intracellular delivery.2,5 In addition, conjugation of Vectocell peptides to small molecules/drugs may also provide a means of inhibiting the extracellular export of the therapeutic agents by proteins involved in multidrug resistance (MDR). Multidrug resistance can seriously limit cancer chemotherapy treatment, for example, through the overexpression of membrane transporters that mediate unidirectional energy-dependent drug efflux, thus reducing intracellular drug levels. These membrane transporters are normally expressed in high levels within cells involved in detoxification, such as the liver, kidney, and colon. Tumors arising from these cells are often resistant to chemotherapy treatment from their onset, while other tumors can acquire resistance through the induction of MDR transport proteins during treatment. Many inhibitors of MDR transporters have been identified. However, these inhibitors also interfere with the clearance of the cytotoxic drug, resulting in elevated plasma concentrations of the cytotoxic agent and associated toxicity. An alternative approach is to circumvent rather than to directly inhibit MDR mechanisms, by developing anticancer therapies that are not substrates for extracellular export. Doxorubicin, an anthracycline antibiotic, remains among the most widely used cytotoxic agents for the treatment of a broad spectrum of cancers, including breast, stomach, non-Hodgkin's lymphoma, and bladder cancer. As with many cytotoxic drugs, doxorubicin has severe short- and long-term side effects, in this case mostly associated with bone marrow and myocardial cell toxicity. Cardiotoxicity limits the cumulative dosage of doxorubicin to 500-600 mg/m2, which may be a dose at which tumor is still responding to treatment but for which no further doxorubicin treatment can be given. Another drawback of doxorubicin is the emergence of drug resistance that results in the reduction of the intracellular concentration of doxorubicin. The present study aimed to generate novel peptidic-doxorubicin conjugates by use of three Vectocell peptides that differ in terms of their charge, size, and intracellular delivery characteristics and to assess their ability to enhance the therapeutic potential of doxorubicin and to prevent the appearance of MDR. Different conjugation sites and linkers of different stabilities were used to generate Vectocell-doxorubicin conjugates in order to evaluate their effect on efficacy. Chemical routes were developed to allow the conjugation of doxorubicin to Vectocell peptides through ester, thioether, and amide chemical linkers. The ester and thioether involved carbon 14 of doxorubicin, and the amide carbon 3. In vitro and in vivo characterization has defined the optimal conjugate-linker combination that significantly increases efficacy above unconjugated doxorubicin in both doxorubicin-sensitive and -resistant models. The data presented therefore provide in vivo proof of concept for the use of Vectocell peptides to improve the therapeutic index of doxorubicin, and potentially many other cytotoxic or small molecule anticancer drugs.
Click to Show/Hide
|
||||
| Description |
The conjugates 8a-c all showed activity; the most active was 8a. Compound 12a showed no activity and confirmed the in vitro data. In a second experiment, conjugates 9c, 9b, and 8a were compared. Compound 9c was completely inactive and 9b was only partially active, which correlated with the reduction of in vitro cytotoxicity of both 9c and 9b compared to 8a observed in the HCT116 model. The in vivo evaluation of the Vectocell-doxorubicin conjugates confirmed that 8a (15 μmol/kg) is the optimal conjugate with a T/C value of 29.5% (Table 1), whereas T/C obtained with the other conjugates ranged from 34.1% (8c) to 86.2% (9c). Moreover, in this experiment, the conjugate 8a (15 μmol/kg) showed better efficacy than doxorubicin (10, 6.5 μmol/kg: doxorubucin's maximal tolerated dose, MTD), with a T/C of 49.6%. It should be noted that it is possible to administer 8a at twice the MTD of doxorubicin (10), demonstrating that 8a is less toxic and more active than 10. For this reason 8a was therefore selected for further preclinical evaluation in both doxorubicin-sensitive and -resistant models.
Click to Show/Hide
|
||||
| In Vitro Model | Colon carcinoma | HCT 116 cell | CVCL_0291 | ||
References
