Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_02085
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
NT4-MON ether
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
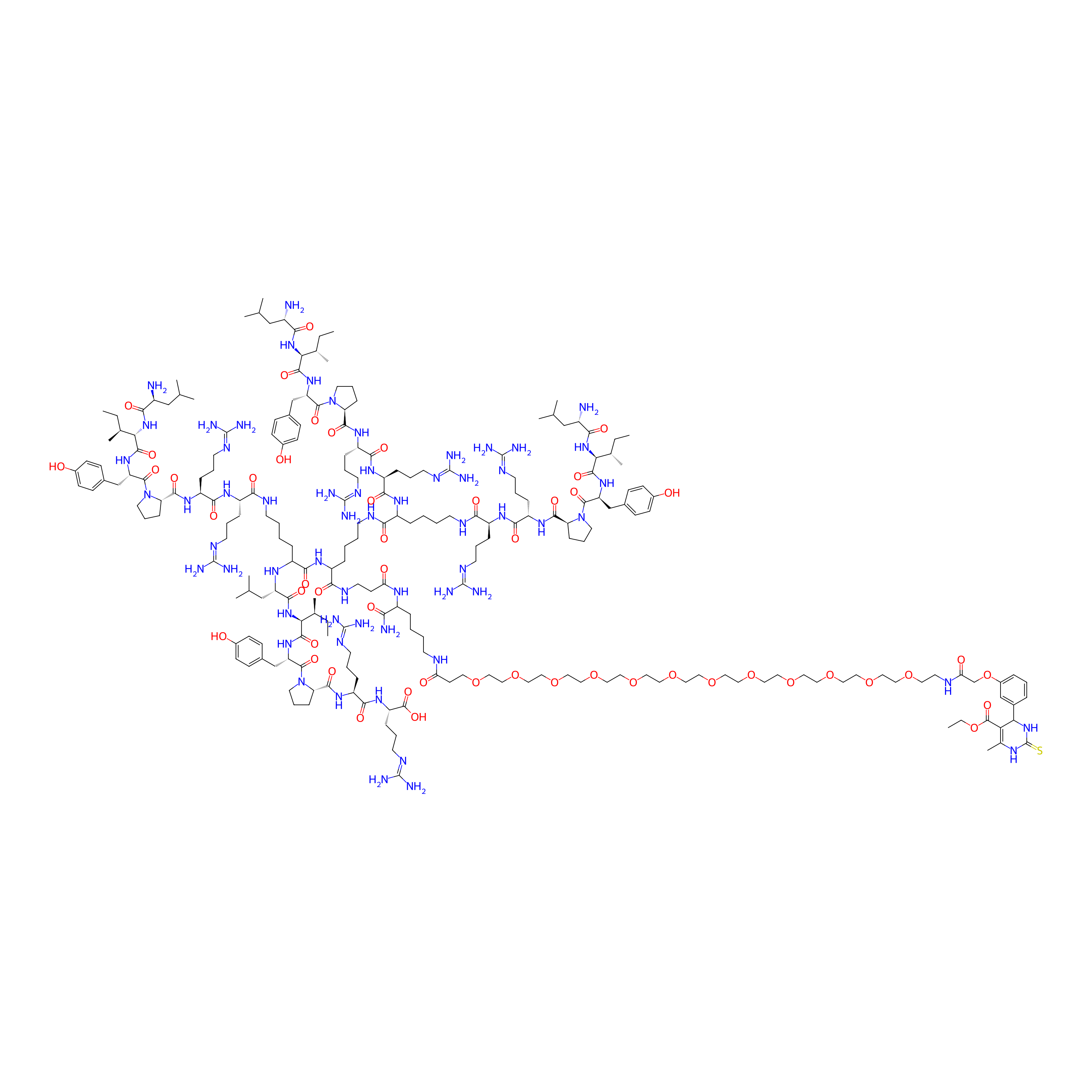
|
|||||
| Peptide Name |
Branched neurotensin (NT4)
|
Peptide Info | ||||
| Drug Name |
Monastrol
|
Drug Info | ||||
| Therapeutic Target |
Kinesin-like protein KIF11 (KIF11)
|
Target Info | ||||
| Linker Name |
Ether bond
|
Linker Info | ||||
| Formula |
C222H372N60O51S
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 4729.854 | ||||
| Lipid-water partition coefficient (xlogp) | -6.5763 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 53 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 63 | |||||
| Rotatable Bond Count (rotbonds) | 180 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability |
0.00%
|
|||
| Administration Time | 6 days | ||||
| Administration Dosage | 0.03 mM | ||||
| MOA of PDC |
We have been studying protease-resistant branched peptides as tumor targeting agents by using tetra-branched peptides (NT4) containing the human regulatory peptide neurotensin (NT) sequence. Neurotensin receptors are overexpressed in several human malignancies, such as colon, pancreatic, prostate and small-cell lung cancer. We have been using NT4 conjugated to different functional units for tumor imaging and therapy, and found that NT4 conjugated to methotrexate produced 60% reduction of tumor growth in xenografted mice. Results obtained with NT4 indicated that branched peptides are promising novel multifunctional targeting molecules, which might allow cancer detection and therapy by means of the same molecule, with no modification in target binding, but rather a simple exchange of functional units. Since cancer cells are very different from one another in terms of drug sensibility, not only in different tumors but in different patients and stages of the disease, this approach prefigures the synthesis of a number of constructs conjugated with differently acting chemotherapeutics. The type of linkage between effector unit (drug/imaging agent etc.) and peptide is obviously crucial for this type of approach. The choice must be driven by two issues: 1) The nature of the drug functional groups available for coupling with the peptide; and 2) the mechanism-of-action of the drug. When a prodrug acts without being released from the carrier unit, a strong linker is preferred. However, if a drug has to be released in order to interact with the intracellular target, the linker must be cleavable. In latter, the linker has to be chosen properly-not too labile or leakage will occur during drug distribution, but not too robust or the pharmacological action will be impaired.
Click to Show/Hide
|
||||
| Description |
This clearly indicated that the chemotherapeutic moiety was released into the cell medium and the drug was internalized into the cells, probably by membrane diffusion. This became evident when observing the behavior of the unrelated conjugated peptides.
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability |
5.00%
|
|||
| Administration Time | 6 days | ||||
| Administration Dosage | 0.03 mM | ||||
| MOA of PDC |
We have been studying protease-resistant branched peptides as tumor targeting agents by using tetra-branched peptides (NT4) containing the human regulatory peptide neurotensin (NT) sequence. Neurotensin receptors are overexpressed in several human malignancies, such as colon, pancreatic, prostate and small-cell lung cancer. We have been using NT4 conjugated to different functional units for tumor imaging and therapy, and found that NT4 conjugated to methotrexate produced 60% reduction of tumor growth in xenografted mice. Results obtained with NT4 indicated that branched peptides are promising novel multifunctional targeting molecules, which might allow cancer detection and therapy by means of the same molecule, with no modification in target binding, but rather a simple exchange of functional units. Since cancer cells are very different from one another in terms of drug sensibility, not only in different tumors but in different patients and stages of the disease, this approach prefigures the synthesis of a number of constructs conjugated with differently acting chemotherapeutics. The type of linkage between effector unit (drug/imaging agent etc.) and peptide is obviously crucial for this type of approach. The choice must be driven by two issues: 1) The nature of the drug functional groups available for coupling with the peptide; and 2) the mechanism-of-action of the drug. When a prodrug acts without being released from the carrier unit, a strong linker is preferred. However, if a drug has to be released in order to interact with the intracellular target, the linker must be cleavable. In latter, the linker has to be chosen properly-not too labile or leakage will occur during drug distribution, but not too robust or the pharmacological action will be impaired.
Click to Show/Hide
|
||||
| Description |
This clearly indicated that the chemotherapeutic moiety was released into the cell medium and the drug was internalized into the cells, probably by membrane diffusion. This became evident when observing the behavior of the unrelated conjugated peptides.
|
||||
| In Vitro Model | Colon adenocarcinoma | HT-29 cell | CVCL_0320 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability |
50.00%
|
|||
| Administration Time | 6 days | ||||
| Administration Dosage | 0.03 mM | ||||
| MOA of PDC |
We have been studying protease-resistant branched peptides as tumor targeting agents by using tetra-branched peptides (NT4) containing the human regulatory peptide neurotensin (NT) sequence. Neurotensin receptors are overexpressed in several human malignancies, such as colon, pancreatic, prostate and small-cell lung cancer. We have been using NT4 conjugated to different functional units for tumor imaging and therapy, and found that NT4 conjugated to methotrexate produced 60% reduction of tumor growth in xenografted mice. Results obtained with NT4 indicated that branched peptides are promising novel multifunctional targeting molecules, which might allow cancer detection and therapy by means of the same molecule, with no modification in target binding, but rather a simple exchange of functional units. Since cancer cells are very different from one another in terms of drug sensibility, not only in different tumors but in different patients and stages of the disease, this approach prefigures the synthesis of a number of constructs conjugated with differently acting chemotherapeutics. The type of linkage between effector unit (drug/imaging agent etc.) and peptide is obviously crucial for this type of approach. The choice must be driven by two issues: 1) The nature of the drug functional groups available for coupling with the peptide; and 2) the mechanism-of-action of the drug. When a prodrug acts without being released from the carrier unit, a strong linker is preferred. However, if a drug has to be released in order to interact with the intracellular target, the linker must be cleavable. In latter, the linker has to be chosen properly-not too labile or leakage will occur during drug distribution, but not too robust or the pharmacological action will be impaired.
Click to Show/Hide
|
||||
| Description |
This clearly indicated that the chemotherapeutic moiety was released into the cell medium and the drug was internalized into the cells, probably by membrane diffusion. This became evident when observing the behavior of the unrelated conjugated peptides.
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | ||
References
