Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00038
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
E1-3
|
|||||
| Structure |
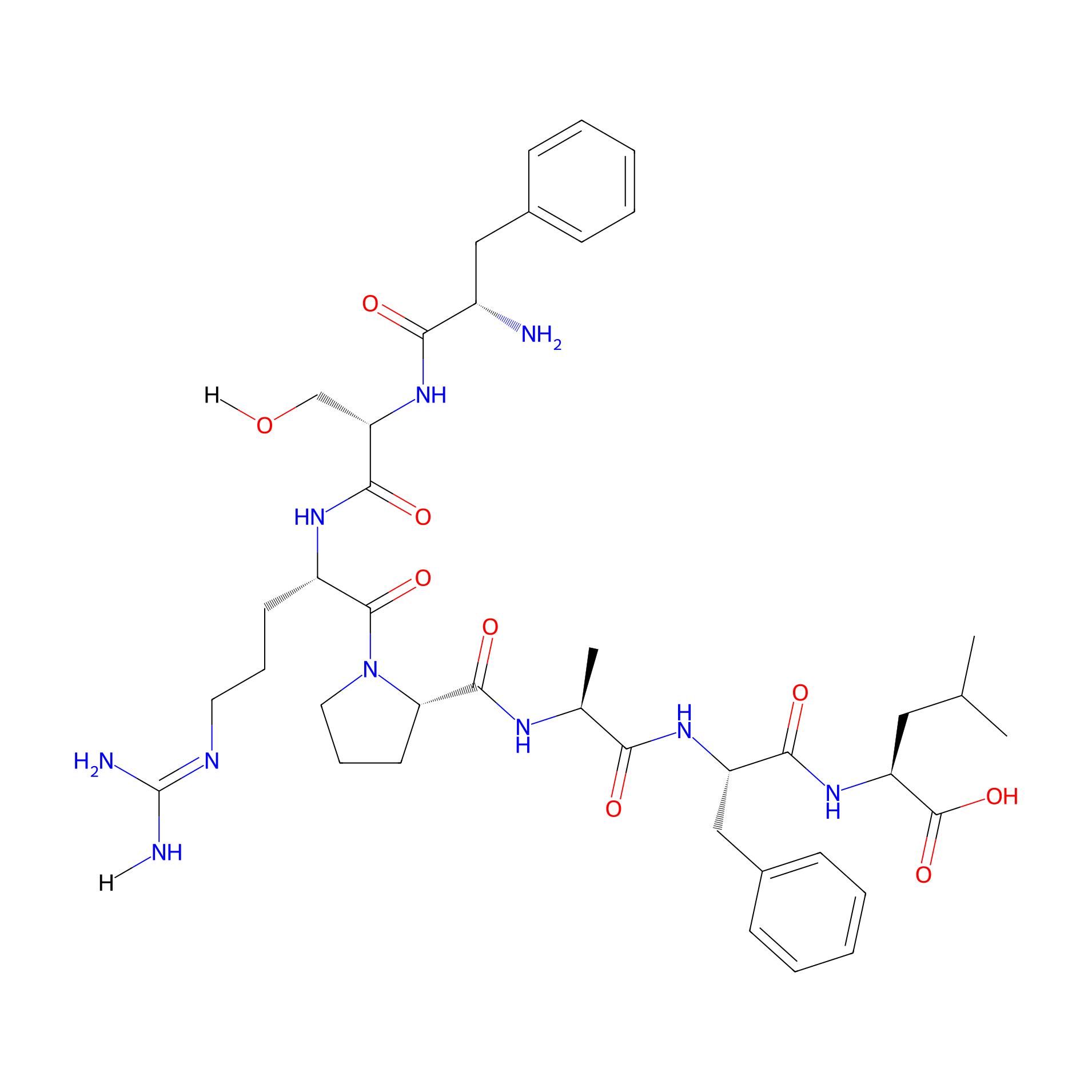
|
|||||
| Sequence |
FSRPAFL
|
|||||
| Peptide Type |
Linear
|
|||||
| PDC Transmembrane Types | Cell-penetrating peptides (CPPs) | |||||
| Formula |
C41H60N10O9
|
|||||
| Isosmiles |
[H]N/C(N)=N/CCC[C@H](NC(=O)[C@H](CO[H])NC(=O)[C@@H](N)Cc1ccccc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CC(C)C)C(=O)O
|
|||||
| InChI |
InChI=1S/C41H60N10O9/c1-24(2)20-31(40(59)60)49-36(55)30(22-27-14-8-5-9-15-27)48-34(53)25(3)46-38(57)33-17-11-19-51(33)39(58)29(16-10-18-45-41(43)44)47-37(56)32(23-52)50-35(54)28(42)21-26-12-6-4-7-13-26/h4-9,12-15,24-25,28-33,52H,10-11,16-23,42H2,1-3H3,(H,46,57)(H,47,56)(H,48,53)(H,49,55)(H,50,54)(H,59,60)(H4,43,44,45)/t25-,28-,29-,30-,31-,32-,33-/m0/s1
|
|||||
| InChIKey |
HLXZSWBPMOOQLL-GCNYMACQSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
836.992
|
Polar area
|
313.76
|
||
|
Complexity
|
836.4544735
|
xlogp Value
|
-1.5894
|
|||
|
Heavy Count
|
60
|
Rot Bonds
|
24
|
|||
|
Hbond acc
|
10
|
Hbond Donor
|
10
|
|||
The Activity Data of This Peptide
| Peptide Activity Information 1 | [1] | |||||
| Abs 410nm | 0.65 | |||||
|---|---|---|---|---|---|---|
| Binding Affinity Assay |
Medulloblastoma cells (DAOY) were plated into 96-multiwell ELISA plates (1 103 cells/well) the day prior to the assay. Cells were incubated in fresh culture medium without serum for 1 h at 37 °C and then fixed with freshly prepared 4% paraformaldehyde for 20 min at room temperature. Cells were washed three times in PBS and blocked for 1 h at room temperature in PBS containing 2% nonfat skim milk powder. The selected phage clones and a negative control M13KE phage were added (5 1010 pfu/well) individually to the cells and incubated for 90 min at room temperature. The phages were removed and cells washed in PBS containing 0.05% Tween. Anti-M13 antibody conjugated to horseradish peroxidase (HRP) was added to each well and incubated at room temperature for 1 h. Cells were washed with PBS containing 0.05% Tween and incubated with 2,2-azino-bis (3-ethylbenzothiazoline-6 sulfonic acid, ABTS) for 30 min at room temperature. The color reaction was stopped using HCl and the optimal density (OD) for each sample was measured at 410 nm using a microplate reader (PerkinElmer Victor3 plate reader).
Click to Show/Hide
|
|||||
| Experimental Condition | Medulloblastoma cells | |||||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
E1-3 doxorubicin [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Blood-brain barrier permeability |
8
|
|||
| Administration Time | 30 min | ||||
| Description |
Notably, its permeability efficiency was significantly higher compared to free doxorubicin (5) (36.93 ± 0.7 uM and 28.93 ± 0.2 uM, respectively, p < 0.001) at 120 min post-treatment (Figure 8D).
|
||||
| In Vivo Model | Blood brain barrier model. | ||||
| In Vitro Model | Normal | HBEC-5i cell | CVCL_4D10 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Blood-brain barrier permeability |
20
|
|||
| Administration Time | 60 min | ||||
| Description |
Notably, its permeability efficiency was significantly higher compared to free doxorubicin (5) (36.93 ± 0.7 uM and 28.93 ± 0.2 uM, respectively, p < 0.001) at 120 min post-treatment (Figure 8D).
|
||||
| In Vivo Model | Blood brain barrier model. | ||||
| In Vitro Model | Normal | HBEC-5i cell | CVCL_4D10 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Blood-brain barrier permeability |
37
|
|||
| Administration Time | 120 min | ||||
| Description |
Notably, its permeability efficiency was significantly higher compared to free doxorubicin (5) (36.93 ± 0.7 uM and 28.93 ± 0.2 uM, respectively, p < 0.001) at 120 min post-treatment (Figure 8D).
|
||||
| In Vivo Model | Blood brain barrier model. | ||||
| In Vitro Model | Normal | HBEC-5i cell | CVCL_4D10 | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
25 ± 1.22 nM
|
|||
| Administration Time | 72 h | ||||
| Description |
This was confirmed with E1-7 doxorubicin conjugate (4) displaying a 5-fold reduction in cytotoxicity compared to E1-3 doxorubicin conjugate (3) (IC50 values of 130 ± 1.27 nM and 25 ± 1.22 nM, respectively) and 14-fold reduction in cytotoxicity compared to free doxorubicin (5) (IC50 values of 130 ± 1.27 nM and 8.8 ± 1.31 nM, respectively) (Figure 7).
Click to Show/Hide
|
||||
| In Vitro Model | Medulloblastoma | Medulloblastoma cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
842.0 ± 1.10 nM
|
|||
| Administration Time | 72 h | ||||
| Description |
E1-3 doxorubicin conjugate had a pronounced reduction in cytotoxicity (>72-fold reduction, IC50 value of 10754 ± 1.38 nM) compared to free doxorubicin (IC50 value of 148 ± 1.15 nM) in human fibroblasts. E1-7 doxorubicin was also able to reduce the cytotoxicity of doxorubicin on fibroblasts but not to the same degree as the E1-3 doxorubicin conjugate. E1-3 doxorubicin conjugate (3) also had reduced cytotoxicity compared to free doxorubicin (>7.4-fold reduction, IC50 values of 842 ± 1.10 nM and 113 ± 1.14 nM, respectively) in primary cultures of human astrocytes, a major cell type located in the brain and spinal cord.
Click to Show/Hide
|
||||
| In Vitro Model | Glioma | Brain astrocytes | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
10754 ± 1.38 nM
|
|||
| Administration Time | 72 h | ||||
| Description |
E1-3 doxorubicin conjugate had a pronounced reduction in cytotoxicity (>72-fold reduction, IC50 value of 10754 ± 1.38 nM) compared to free doxorubicin (IC50 value of 148 ± 1.15 nM) in human fibroblasts. E1-7 doxorubicin was also able to reduce the cytotoxicity of doxorubicin on fibroblasts but not to the same degree as the E1-3 doxorubicin conjugate. E1-3 doxorubicin conjugate (3) also had reduced cytotoxicity compared to free doxorubicin (>7.4-fold reduction, IC50 values of 842 ± 1.10 nM and 113 ± 1.14 nM, respectively) in primary cultures of human astrocytes, a major cell type located in the brain and spinal cord.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MRC-5 cell | CVCL_0440 | ||
References
