Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00144
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
E1-3 doxorubicin
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
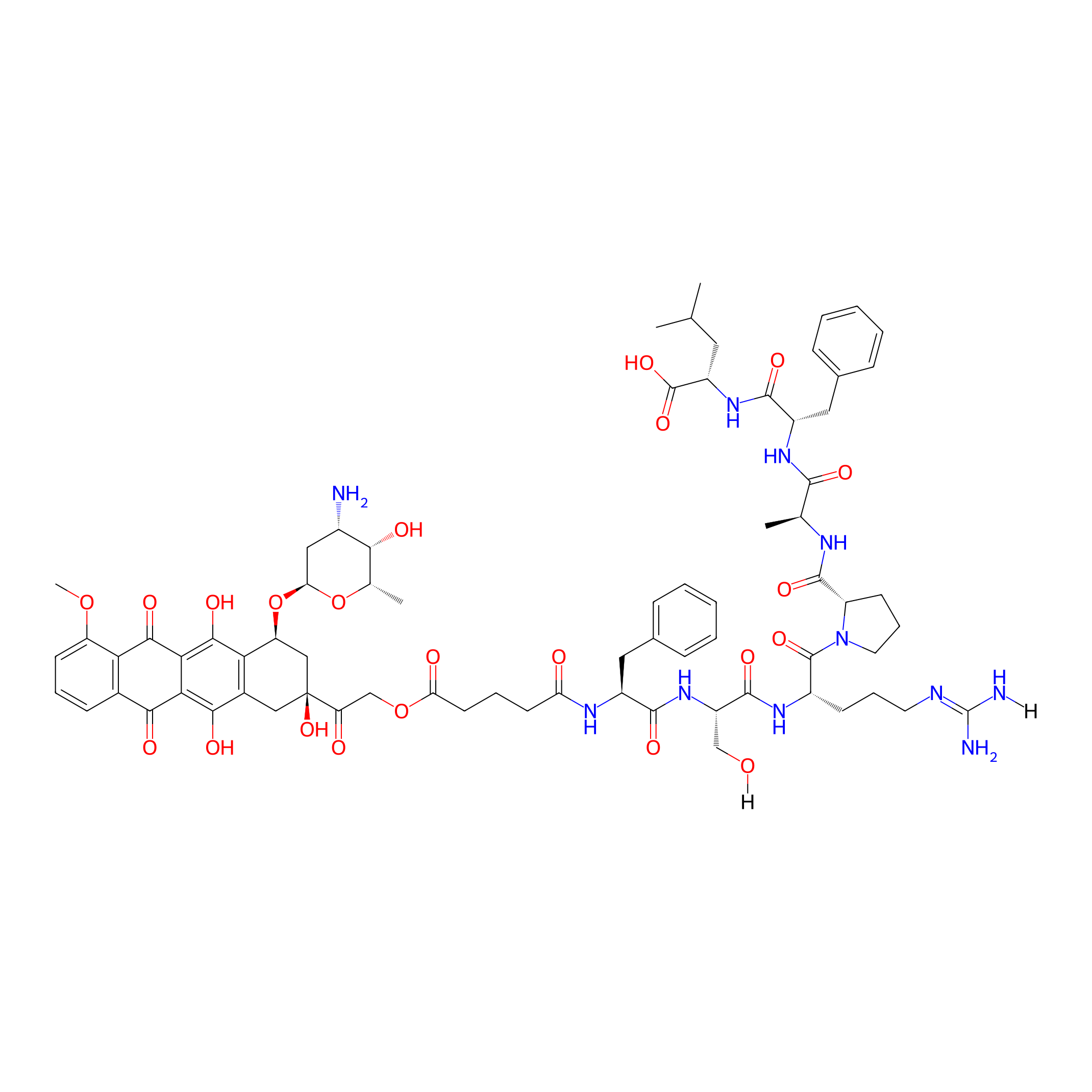
|
|||||
| Peptide Name |
E1-3
|
Peptide Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
Glutaric acid
|
Linker Info | ||||
| Formula |
C73H93N11O22
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 1476.602 | ||||
| Lipid-water partition coefficient (xlogp) | -0.6958 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 15 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 23 | |||||
| Rotatable Bond Count (rotbonds) | 35 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Blood-brain barrier permeability |
8
|
|||
| Administration Time | 30 min | ||||
| Description |
Notably, its permeability efficiency was significantly higher compared to free doxorubicin (5) (36.93 ± 0.7 uM and 28.93 ± 0.2 uM, respectively, p < 0.001) at 120 min post-treatment (Figure 8D).
|
||||
| In Vivo Model | Blood brain barrier model. | ||||
| In Vitro Model | Normal | HBEC-5i cell | CVCL_4D10 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Blood-brain barrier permeability |
20
|
|||
| Administration Time | 60 min | ||||
| Description |
Notably, its permeability efficiency was significantly higher compared to free doxorubicin (5) (36.93 ± 0.7 uM and 28.93 ± 0.2 uM, respectively, p < 0.001) at 120 min post-treatment (Figure 8D).
|
||||
| In Vivo Model | Blood brain barrier model. | ||||
| In Vitro Model | Normal | HBEC-5i cell | CVCL_4D10 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Blood-brain barrier permeability |
37
|
|||
| Administration Time | 120 min | ||||
| Description |
Notably, its permeability efficiency was significantly higher compared to free doxorubicin (5) (36.93 ± 0.7 uM and 28.93 ± 0.2 uM, respectively, p < 0.001) at 120 min post-treatment (Figure 8D).
|
||||
| In Vivo Model | Blood brain barrier model. | ||||
| In Vitro Model | Normal | HBEC-5i cell | CVCL_4D10 | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
25 ± 1.22 nM
|
|||
| Administration Time | 72 h | ||||
| Description |
This was confirmed with E1-7 doxorubicin conjugate (4) displaying a 5-fold reduction in cytotoxicity compared to E1-3 doxorubicin conjugate (3) (IC50 values of 130 ± 1.27 nM and 25 ± 1.22 nM, respectively) and 14-fold reduction in cytotoxicity compared to free doxorubicin (5) (IC50 values of 130 ± 1.27 nM and 8.8 ± 1.31 nM, respectively) (Figure 7).
Click to Show/Hide
|
||||
| In Vitro Model | Medulloblastoma | Medulloblastoma cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
842.0 ± 1.10 nM
|
|||
| Administration Time | 72 h | ||||
| Description |
E1-3 doxorubicin conjugate had a pronounced reduction in cytotoxicity (>72-fold reduction, IC50 value of 10754 ± 1.38 nM) compared to free doxorubicin (IC50 value of 148 ± 1.15 nM) in human fibroblasts. E1-7 doxorubicin was also able to reduce the cytotoxicity of doxorubicin on fibroblasts but not to the same degree as the E1-3 doxorubicin conjugate. E1-3 doxorubicin conjugate (3) also had reduced cytotoxicity compared to free doxorubicin (>7.4-fold reduction, IC50 values of 842 ± 1.10 nM and 113 ± 1.14 nM, respectively) in primary cultures of human astrocytes, a major cell type located in the brain and spinal cord.
Click to Show/Hide
|
||||
| In Vitro Model | Glioma | Brain astrocytes | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Medulloblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
10754 ± 1.38 nM
|
|||
| Administration Time | 72 h | ||||
| Description |
E1-3 doxorubicin conjugate had a pronounced reduction in cytotoxicity (>72-fold reduction, IC50 value of 10754 ± 1.38 nM) compared to free doxorubicin (IC50 value of 148 ± 1.15 nM) in human fibroblasts. E1-7 doxorubicin was also able to reduce the cytotoxicity of doxorubicin on fibroblasts but not to the same degree as the E1-3 doxorubicin conjugate. E1-3 doxorubicin conjugate (3) also had reduced cytotoxicity compared to free doxorubicin (>7.4-fold reduction, IC50 values of 842 ± 1.10 nM and 113 ± 1.14 nM, respectively) in primary cultures of human astrocytes, a major cell type located in the brain and spinal cord.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MRC-5 cell | CVCL_0440 | ||
References
