Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00093
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
Chlorotoxin
|
|||||
| Structure |
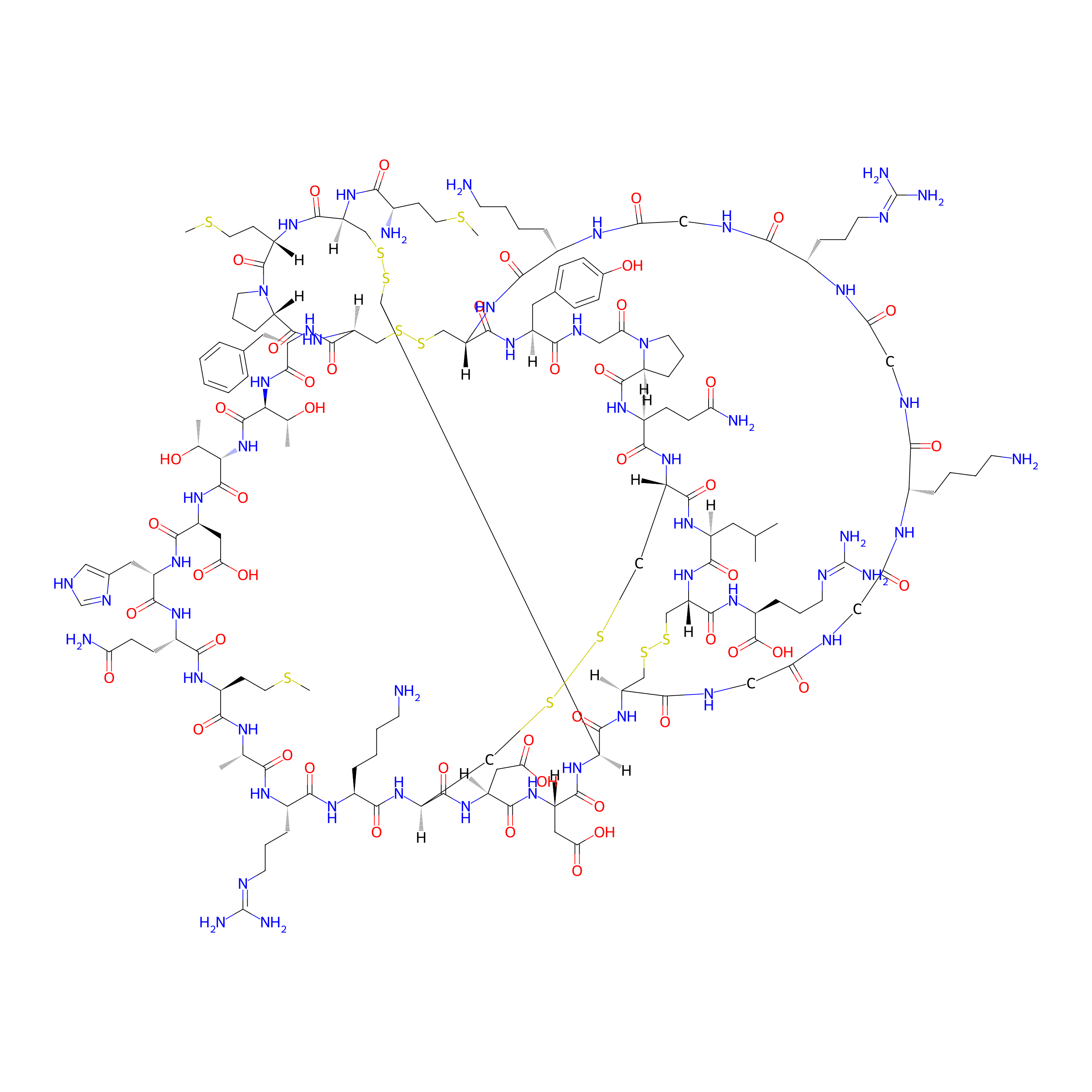
|
|||||
| Sequence |
MCMPCFTTDHQMARKCDDCCGGKGRGKCYGPQCLCR
|
|||||
| Peptide Type |
Linear
|
|||||
| Receptor Name |
Neuropilin-1 (NRP1)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C158H248N52O48S11
|
|||||
| Isosmiles |
[H]NCCCC[C@@H]1NC(=O)CNC(=O)CNC(=O)[C@@H]2CSSC[C@@H](C(=O)N[C@@H](CCC/N=C(\N)N[H])C(=O)O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H]3CSSC[C@@H]4NC(=O)[C@H](CCCCN[H])NC(=O)[C@H](CCC/N=C(\N)N[H])NC(=O)[C@H](C)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCC(=O)N[H])NC(=O)[C@H](Cc5cn([H])cn5)NC(=O)[C@H](CC(=O)O)NC(=O)[C@]([H])([C@@H](C)O[H])NC(=O)[C@]([H])([C@@H](C)O[H])NC(=O)[C@H](Cc5ccccc5)NC(=O)[C@H](CSSC[C@H](NC(=O)[C@H](CCCCN[H])NC(=O)CNC(=O)[C@H](CCC/N=C(\N)N[H])NC(=O)CNC1=O)C(=O)N[C@@H](Cc1ccc(O[H])cc1)C(=O)NCC(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(=O)N[H])C(=O)N3)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCSC)NC(=O)[C@@H](NC(=O)[C@H](CCSC)N[H])CSSC[C@H](NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CC(=O)O)NC4=O)C(=O)N2
|
|||||
| InChI |
InChI=1S/C158H248N52O48S11/c1-77(2)55-96-137(239)204-105(144(246)191-95(155(257)258)30-20-49-174-158(169)170)70-264-262-68-103-130(232)176-63-115(216)175-64-116(217)182-86(25-12-15-44-159)127(229)177-65-117(218)183-87(28-18-47-172-156(165)166)128(230)178-66-118(219)184-88(26-13-16-45-160)132(234)201-107-72-267-269-75-110-148(250)194-98(56-81-23-10-9-11-24-81)142(244)207-124(80(5)212)153(255)208-123(79(4)211)152(254)198-102(61-122(225)226)140(242)195-99(58-83-62-171-76-180-83)138(240)187-91(37-39-113(163)214)135(237)188-93(42-53-260-7)131(233)181-78(3)125(227)185-90(29-19-48-173-157(167)168)133(235)186-89(27-14-17-46-161)134(236)202-108(73-266-265-71-106(145(247)192-96)203-136(238)92(38-40-114(164)215)189-150(252)111-31-21-50-209(111)119(220)67-179-129(231)97(193-146(107)248)57-82-33-35-84(213)36-34-82)147(249)197-100(59-120(221)222)139(241)196-101(60-121(223)224)141(243)205-109(149(251)200-103)74-268-263-69-104(199-126(228)85(162)41-52-259-6)143(245)190-94(43-54-261-8)154(256)210-51-22-32-112(210)151(253)206-110/h9-11,23-24,33-36,62,76-80,85-112,123-124,211-213H,12-22,25-32,37-61,63-75,159-162H2,1-8H3,(H2,163,214)(H2,164,215)(H,171,180)(H,175,216)(H,176,232)(H,177,229)(H,178,230)(H,179,231)(H,181,233)(H,182,217)(H,183,218)(H,184,219)(H,185,227)(H,186,235)(H,187,240)(H,188,237)(H,189,252)(H,190,245)(H,191,246)(H,192,247)(H,193,248)(H,194,250)(H,195,242)(H,196,241)(H,197,249)(H,198,254)(H,199,228)(H,200,251)(H,201,234)(H,202,236)(H,203,238)(H,204,239)(H,205,243)(H,206,253)(H,207,244)(H,208,255)(H,221,222)(H,223,224)(H,225,226)(H,257,258)(H4,165,166,172)(H4,167,168,173)(H4,169,170,174)/t78-,79+,80+,85-,86-,87-,88-,89-,90-,91-,92-,93-,94-,95-,96-,97-,98-,99-,100-,101-,102-,103-,104-,105-,106-,107-,108-,109-,110-,111-,112-,123-,124-/m0/s1
|
|||||
| InChIKey |
DSHHZWDCJBIDQA-GQIQPHNSSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
3996.775
|
Polar area
|
1622.95
|
||
|
Complexity
|
3993.549139
|
xlogp Value
|
-19.1377
|
|||
|
Heavy Count
|
269
|
Rot Bonds
|
67
|
|||
|
Hbond acc
|
63
|
Hbond Donor
|
53
|
|||
The Activity Data of This Peptide
| Peptide Activity Information 1 | [1] | |||||
| Binding constant | 240 nM | |||||
|---|---|---|---|---|---|---|
| Binding Affinity Assay |
Octet binding studies required biotinylation of Cltx and derived peptides for loading onto streptavidin biosensors. Trypsin, (Promega Cat# V5280) (&tide; 40 μg/mL) was added to Cltx biotinylated at K27 or N-terminus (10 mg/mL) and incubated for 24 h at 37 °C, pH 8.5. The resulting peptides were separated by preparative HPLC and peptide fragments were characterized by MALDI. Small linear Cltx-derived peptides were also synthesized by routine methods and were biotinylated at the N-terminus. In some cases, peptides were reduced with TCEP at pH 8 and the cysteine residues were capped using iodoacetamide.
Click to Show/Hide
|
|||||
| Experimental Condition | Required biotinylation of Cltx | |||||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
ER-472 [Investigative]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
5.45%
|
|||
| Administration Time | 23days | ||||
| Administration Dosage | 1.25 mg/kg | ||||
| MOA of PDC |
Collectively, our data demonstrate that Cltx within ER-472 acts a cryptic peptide which is metabolized to peptides with C-terminal R-COOH in the tumor microenvironment. These peptides bind to tumor cell NRP1 to increase drug uptake, which consequently boosts the antitumor effect.
|
||||
| Description |
At the MTD dose, ER-472 treatment resulted in significant antitumor activity in all 3 models, but at 1/2 MTD no antitumor activity was observed in MIA PaCa-2 or BxPC-3 xenografts. In contrast, a much wider therapeutic window was observed for ER-472 in PC-3 xenografts: tumor regression and several tumor cures were observed at 1/2 MTD and significant antitumor activity was also recorded at the 1/4 MTD dose.
Click to Show/Hide
|
||||
| In Vivo Model | BxPC-3 xenograft model. | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
36.70%
|
|||
| Administration Time | 30 days | ||||
| Administration Dosage | 1.25 mg/kg | ||||
| MOA of PDC |
Collectively, our data demonstrate that Cltx within ER-472 acts a cryptic peptide which is metabolized to peptides with C-terminal R-COOH in the tumor microenvironment. These peptides bind to tumor cell NRP1 to increase drug uptake, which consequently boosts the antitumor effect.
|
||||
| Description |
At the MTD dose, ER-472 treatment resulted in significant antitumor activity in all 3 models, but at 1/2 MTD no antitumor activity was observed in MIA PaCa-2 or BxPC-3 xenografts. In contrast, a much wider therapeutic window was observed for ER-472 in PC-3 xenografts: tumor regression and several tumor cures were observed at 1/2 MTD and significant antitumor activity was also recorded at the 1/4 MTD dose.
Click to Show/Hide
|
||||
| In Vivo Model | MIA PaCa-2 xenograft model. | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
69.23%
|
|||
| Administration Time | 38 days | ||||
| Administration Dosage | 0.625 mg/kg | ||||
| MOA of PDC |
Collectively, our data demonstrate that Cltx within ER-472 acts a cryptic peptide which is metabolized to peptides with C-terminal R-COOH in the tumor microenvironment. These peptides bind to tumor cell NRP1 to increase drug uptake, which consequently boosts the antitumor effect.
|
||||
| Description |
At the MTD dose, ER-472 treatment resulted in significant antitumor activity in all 3 models, but at 1/2 MTD no antitumor activity was observed in MIA PaCa-2 or BxPC-3 xenografts. In contrast, a much wider therapeutic window was observed for ER-472 in PC-3 xenografts: tumor regression and several tumor cures were observed at 1/2 MTD and significant antitumor activity was also recorded at the 1/4 MTD dose.
Click to Show/Hide
|
||||
| In Vivo Model | PC-3 xenograft model. | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
74.54%
|
|||
| Administration Time | 23days | ||||
| Administration Dosage | 2.5 mg/kg | ||||
| MOA of PDC |
Collectively, our data demonstrate that Cltx within ER-472 acts a cryptic peptide which is metabolized to peptides with C-terminal R-COOH in the tumor microenvironment. These peptides bind to tumor cell NRP1 to increase drug uptake, which consequently boosts the antitumor effect.
|
||||
| Description |
At the MTD dose, ER-472 treatment resulted in significant antitumor activity in all 3 models, but at 1/2 MTD no antitumor activity was observed in MIA PaCa-2 or BxPC-3 xenografts. In contrast, a much wider therapeutic window was observed for ER-472 in PC-3 xenografts: tumor regression and several tumor cures were observed at 1/2 MTD and significant antitumor activity was also recorded at the 1/4 MTD dose.
Click to Show/Hide
|
||||
| In Vivo Model | BxPC-3 xenograft model. | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
100%
|
|||
| Administration Time | 38 days | ||||
| Administration Dosage | 1.25 mg/kg | ||||
| MOA of PDC |
Collectively, our data demonstrate that Cltx within ER-472 acts a cryptic peptide which is metabolized to peptides with C-terminal R-COOH in the tumor microenvironment. These peptides bind to tumor cell NRP1 to increase drug uptake, which consequently boosts the antitumor effect.
|
||||
| Description |
At the MTD dose, ER-472 treatment resulted in significant antitumor activity in all 3 models, but at 1/2 MTD no antitumor activity was observed in MIA PaCa-2 or BxPC-3 xenografts. In contrast, a much wider therapeutic window was observed for ER-472 in PC-3 xenografts: tumor regression and several tumor cures were observed at 1/2 MTD and significant antitumor activity was also recorded at the 1/4 MTD dose.
Click to Show/Hide
|
||||
| In Vivo Model | PC-3 xenograft model. | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
100%
|
|||
| Administration Time | 30 days | ||||
| Administration Dosage | 2.5 mg/kg | ||||
| MOA of PDC |
Collectively, our data demonstrate that Cltx within ER-472 acts a cryptic peptide which is metabolized to peptides with C-terminal R-COOH in the tumor microenvironment. These peptides bind to tumor cell NRP1 to increase drug uptake, which consequently boosts the antitumor effect.
|
||||
| Description |
At the MTD dose, ER-472 treatment resulted in significant antitumor activity in all 3 models, but at 1/2 MTD no antitumor activity was observed in MIA PaCa-2 or BxPC-3 xenografts. In contrast, a much wider therapeutic window was observed for ER-472 in PC-3 xenografts: tumor regression and several tumor cures were observed at 1/2 MTD and significant antitumor activity was also recorded at the 1/4 MTD dose.
Click to Show/Hide
|
||||
| In Vivo Model | MIA PaCa-2 xenograft model. | ||||
References
