Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00096
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
NALRDQTGLKNPVQLARAVC
|
|||||
| Structure |
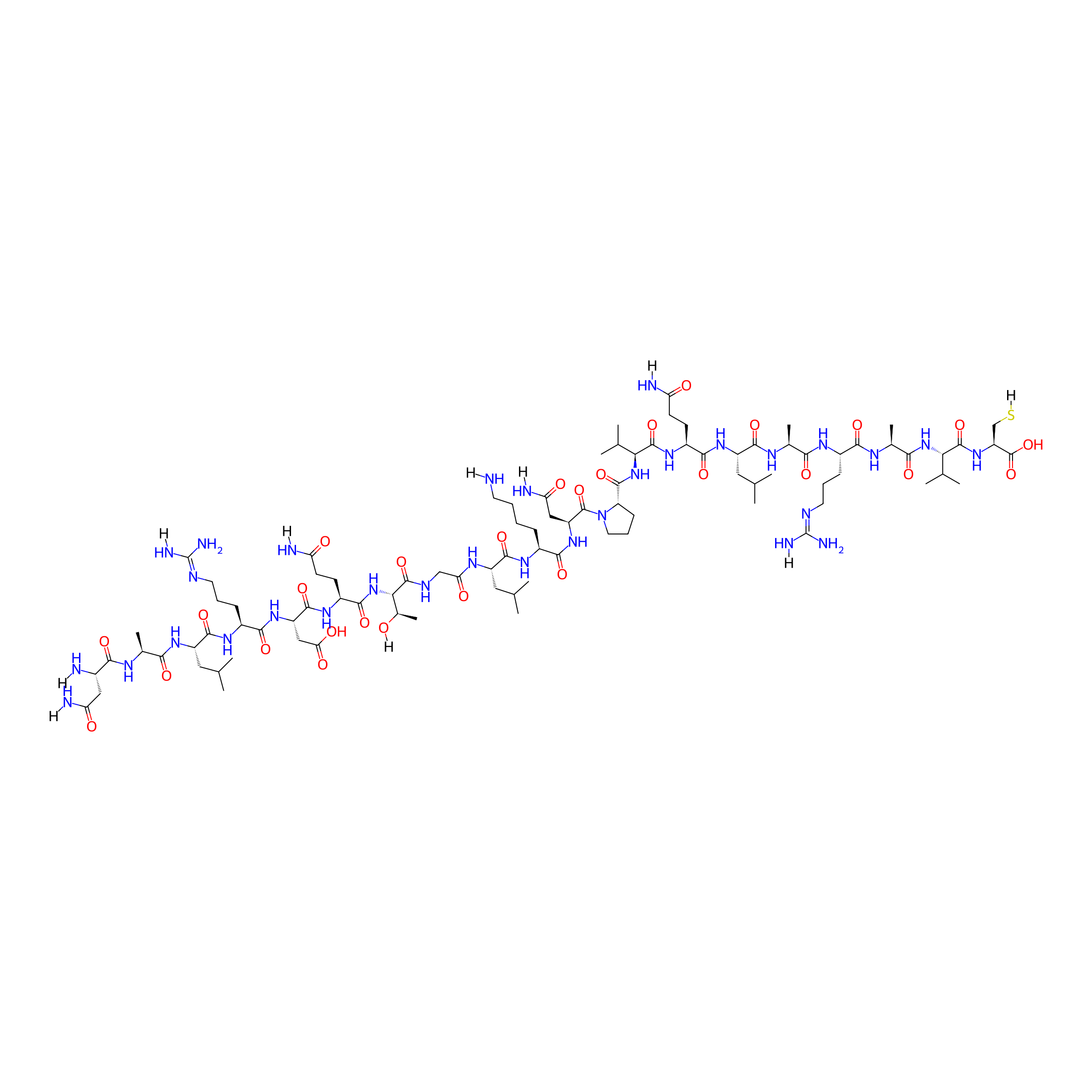
|
|||||
| Sequence |
NALRDQTGLKNPVQLARAVC
|
|||||
| Peptide Type |
Linear
|
|||||
| Receptor Name |
Integrin alpha-V; Integrin beta-6 (ITGAV; ITGB6)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C91H159N31O28S
|
|||||
| Isosmiles |
[H]NCCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@@]([H])(NC(=O)[C@H](CCC(=O)N[H])NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CCC/N=C(\N)N[H])NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CC(=O)N[H])N[H])[C@@H](C)O[H])C(=O)N[C@@H](CC(=O)N[H])C(=O)N1CCC[C@H]1C(=O)N[C@H](C(=O)N[C@@H](CCC(=O)N[H])C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC/N=C(\N)N[H])C(=O)N[C@@H](C)C(=O)N[C@H](C(=O)N[C@@H](CS[H])C(=O)O)C(C)C)C(C)C
|
|||||
| InChI |
InChI=1S/C91H159N31O28S/c1-40(2)32-55(108-66(128)38-104-85(145)70(48(14)123)121-79(139)54(25-27-63(95)125)112-83(143)58(37-67(129)130)116-76(136)52(22-18-30-103-91(100)101)111-82(142)57(34-42(5)6)114-72(132)45(11)105-74(134)49(93)35-64(96)126)81(141)110-50(20-15-16-28-92)77(137)117-59(36-65(97)127)88(148)122-31-19-23-61(122)84(144)120-69(44(9)10)86(146)113-53(24-26-62(94)124)78(138)115-56(33-41(3)4)80(140)107-46(12)71(131)109-51(21-17-29-102-90(98)99)75(135)106-47(13)73(133)119-68(43(7)8)87(147)118-60(39-151)89(149)150/h40-61,68-70,123,151H,15-39,92-93H2,1-14H3,(H2,94,124)(H2,95,125)(H2,96,126)(H2,97,127)(H,104,145)(H,105,134)(H,106,135)(H,107,140)(H,108,128)(H,109,131)(H,110,141)(H,111,142)(H,112,143)(H,113,146)(H,114,132)(H,115,138)(H,116,136)(H,117,137)(H,118,147)(H,119,133)(H,120,144)(H,121,139)(H,129,130)(H,149,150)(H4,98,99,102)(H4,100,101,103)/t45-,46-,47-,48+,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,68-,69-,70-/m0/s1
|
|||||
| InChIKey |
HHGCEFBWETUMSR-JGHQVEGBSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
2167.529
|
Polar area
|
992.14
|
||
|
Complexity
|
2166.169154
|
xlogp Value
|
-12.4396
|
|||
|
Heavy Count
|
151
|
Rot Bonds
|
76
|
|||
|
Hbond acc
|
31
|
Hbond Donor
|
32
|
|||
The Activity Data of This Peptide
| Peptide Activity Information 1 | [1] | |||||
| MTT activity | 89% | |||||
|---|---|---|---|---|---|---|
| Binding Affinity Assay |
Relative amounts of mitochondrial activity were quantified as an indirect measure of cell proliferation using the MTT assay (Promega). All cell lines were seeded at 3x103 cells/mL into 96-well plates. After 24h, cells were treated with serially-diluted concentrations of NT-peptide control, A20FMDV2, SG3199, SG3511 or SG3299 (0-500 nM, n=6 replicates/treatment) in serum-free media. Three biological repeats were performed. After 72 h, cells were subject to the MTT assay. Briefly, cell culture media was removed from each well and 100 μl of MTT reagent was added per well. After 1h at 37°C, MTT solution was replaced with 200μl DMSO and fluorescence measured at 550nm wavelength. Data were plotted relative to PBS (n=6, error bars represent standard deviation).
Click to Show/Hide
|
|||||
| Experimental Condition | A375Ppuro cell | |||||
| Peptide Activity Information 2 | [1] | |||||
| MTT activity | 90% | |||||
| Binding Affinity Assay |
Relative amounts of mitochondrial activity were quantified as an indirect measure of cell proliferation using the MTT assay (Promega). All cell lines were seeded at 3x103 cells/mL into 96-well plates. After 24h, cells were treated with serially-diluted concentrations of NT-peptide control, A20FMDV2, SG3199, SG3511 or SG3299 (0-500 nM, n=6 replicates/treatment) in serum-free media. Three biological repeats were performed. After 72 h, cells were subject to the MTT assay. Briefly, cell culture media was removed from each well and 100 μl of MTT reagent was added per well. After 1h at 37°C, MTT solution was replaced with 200μl DMSO and fluorescence measured at 550nm wavelength. Data were plotted relative to PBS (n=6, error bars represent standard deviation).
Click to Show/Hide
|
|||||
| Experimental Condition | Capan-1 cell | |||||
| Peptide Activity Information 3 | [1] | |||||
| MTT activity | 92% | |||||
| Binding Affinity Assay |
Relative amounts of mitochondrial activity were quantified as an indirect measure of cell proliferation using the MTT assay (Promega). All cell lines were seeded at 3x103 cells/mL into 96-well plates. After 24h, cells were treated with serially-diluted concentrations of NT-peptide control, A20FMDV2, SG3199, SG3511 or SG3299 (0-500 nM, n=6 replicates/treatment) in serum-free media. Three biological repeats were performed. After 72 h, cells were subject to the MTT assay. Briefly, cell culture media was removed from each well and 100 μl of MTT reagent was added per well. After 1h at 37°C, MTT solution was replaced with 200μl DMSO and fluorescence measured at 550nm wavelength. Data were plotted relative to PBS (n=6, error bars represent standard deviation).
Click to Show/Hide
|
|||||
| Experimental Condition | A375Pβ6 cell | |||||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
SG3511 [Investigative]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
34% (Day 28)
|
|||
| Administration Time | Bi-weekly for 4 weeks | ||||
| Administration Dosage | 20 µg/kg | ||||
| Evaluation Method | Tumor volume detection assay | ||||
| MOA of PDC |
Non-targeting SG3511 also produced a significant increase in apoptosis and DNA damage (P<0.05) again, probably as a result of the inherent lipophilicity of tesirine, but there was no significant change in tumor cell number, as measured by tumour cell area, CK-expression and confirmed with E-cadherin expression.
|
||||
| Description |
SG3299 significantly reduced Panc0403/PS1 xenograft tumor growth by 75.8±6% (P<0.001) compared with PBS treatment and by 60.4±9.8% (P<0.05) compared with SG3511 therapy.
|
||||
| In Vivo Model | Athymic CD1Nu/Nu female mice bearing human A375Pbeta6 xenograft tumours. | ||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Panc 04.03 PS1 cell | CVCL_1636 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
34.8 ± 4.6% (Day 28)
|
|||
| Administration Time | T hrice weekly for 4 consecutive weeks | ||||
| Administration Dosage | 10 µg/kg | ||||
| Evaluation Method | Tumor volume detection assay | ||||
| MOA of PDC |
Non-targeting SG3511 also produced a significant increase in apoptosis and DNA damage (P<0.05) again, probably as a result of the inherent lipophilicity of tesirine, but there was no significant change in tumor cell number, as measured by tumour cell area, CK-expression and confirmed with E-cadherin expression.
|
||||
| Description |
Tumors treated with SG3511 and SG3299 exhibited significant reductions in size (P<0.0001, 53.9±23.7% and 34.8±4.6% after 21 days respectively) but there was no significant difference in the effect of either treatment (P=0.24, after 30 days).
|
||||
| In Vivo Model | Athymic CD1Nu/Nu female mice bearing human A375Ppuro xenograft tumours. | ||||
| In Vitro Model | Amelanotic melanoma | A375P-puro cell | CVCL_5F66 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
56.9 ± 16.2% (Day 28)
|
|||
| Administration Time | T hrice weekly for 4 consecutive weeks | ||||
| Administration Dosage | 10 µg/kg | ||||
| Evaluation Method | Tumor volume detection assay | ||||
| MOA of PDC |
Non-targeting SG3511 also produced a significant increase in apoptosis and DNA damage (P<0.05) again, probably as a result of the inherent lipophilicity of tesirine, but there was no significant change in tumor cell number, as measured by tumour cell area, CK-expression and confirmed with E-cadherin expression.
|
||||
| Description |
In contrast, both SG3299 and SG3511 reduced A375P6 tumor growth compared with PBS treatment (79±7% and 56.9±16.2% respectively, P<0.0001) and SG3299 reduced growth by 2.3-fold more than SG3511 (P<0.0001).
|
||||
| In Vivo Model | Athymic CD1Nu/Nu female mice bearing human A375Pbeta6 xenograft tumours. | ||||
| In Vitro Model | Amelanotic melanoma | A375P-beta6 cell | CVCL_5F66 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
75% (Day 35)
|
|||
| Administration Time | T hrice weekly for 5 consecutive weeks | ||||
| Administration Dosage | 10 µg/kg | ||||
| Evaluation Method | Tumor volume detection assay | ||||
| MOA of PDC |
Non-targeting SG3511 also produced a significant increase in apoptosis and DNA damage (P<0.05) again, probably as a result of the inherent lipophilicity of tesirine, but there was no significant change in tumor cell number, as measured by tumour cell area, CK-expression and confirmed with E-cadherin expression.
|
||||
| Description |
Capan-1 xenografts responded in a similar manner to A375P6 xenografts upon 10 ug/kg tri-weekly treatment, with significant growth inhibition with SG3511 and SG3299 (P<0.0001). Again, SG3299 inhibited tumor growth significantly more than SG3511 (P<0.0001).
|
||||
| In Vivo Model | Athymic CD1Nu/Nu female mice bearing human Capan-1 xenograft tumours. | ||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Capan-1 cell | CVCL_0237 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
96.1 ± 3.4% (Day 28)
|
|||
| Administration Time | Bi-weekly for 4 weeks | ||||
| Administration Dosage | 25 µg/kg | ||||
| Evaluation Method | Tumor volume detection assay | ||||
| MOA of PDC |
Non-targeting SG3511 also produced a significant increase in apoptosis and DNA damage (P<0.05) again, probably as a result of the inherent lipophilicity of tesirine, but there was no significant change in tumor cell number, as measured by tumour cell area, CK-expression and confirmed with E-cadherin expression.
|
||||
| Description |
Again, SG3299 significantly reduced Capan-1 tumor growth achieving 97.7±2% (P<0.0001) and 96.1±3.4% (P<0.0001) reductions compared to PBS and SG3511, respectively.
|
||||
| In Vivo Model | Athymic CD1Nu/Nu female mice bearing human A375Pbeta6 xenograft tumours. | ||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Capan-1 cell | CVCL_0237 | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
223 nM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | WST-1 assay | ||||
| MOA of PDC |
The integrin vβ6 is expressed on ˜85% of pancreatic cancers with minimal expression in healthy tissues, and thus is a valid therapeutic target. We previously developed the A20FMDV2 peptide that binds with high-affinity to vβ6. SG3299 is a peptide-toxin conjugate that conjugates A20FMDV2 to a synthetic pyrrolobenzodiazepine (PBD) dimer cytotoxic warhead with a cathepsin B-cleavable valine-alanine linker. We have shown that SG3299 is highly effective in subcutaneous pancreatic cancer xenografts in immunodeficient models, with prolonged survival and tumour eliminations.
Click to Show/Hide
|
||||
| Description |
The relative specificity of SG3299 for v6, and the in vitro cytotoxic effect of SG3299 on v6-expressing murine cancer cells was confirmed with a growth inhibition assay performed on TB32043mb6s2 and TB32043 cells (high & negative v6 expression respectively). Cell viability was evaluated with a WST-1 assay following treatment with 0-105 nM of SG3299 or SG3511. The IC50 of v6-targeted SG3299 in TB32043mb6s2 was over 15-fold lower than in TB32043 cells (24 nM vs 418 nM, p < 0.001). There was no significant difference in the IC50 values for the non-targeted SG3511 between TB32043mb6s2 and TB32043 cells (223 vs 300 nM, p = 0.17).
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Pancreatic ductal adenocarcinoma cell TB32043mb6s2 | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
300 nM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | WST-1 assay | ||||
| MOA of PDC |
The integrin vβ6 is expressed on ˜85% of pancreatic cancers with minimal expression in healthy tissues, and thus is a valid therapeutic target. We previously developed the A20FMDV2 peptide that binds with high-affinity to vβ6. SG3299 is a peptide-toxin conjugate that conjugates A20FMDV2 to a synthetic pyrrolobenzodiazepine (PBD) dimer cytotoxic warhead with a cathepsin B-cleavable valine-alanine linker. We have shown that SG3299 is highly effective in subcutaneous pancreatic cancer xenografts in immunodeficient models, with prolonged survival and tumour eliminations.
Click to Show/Hide
|
||||
| Description |
The relative specificity of SG3299 for v6, and the in vitro cytotoxic effect of SG3299 on v6-expressing murine cancer cells was confirmed with a growth inhibition assay performed on TB32043mb6s2 and TB32043 cells (high & negative v6 expression respectively). Cell viability was evaluated with a WST-1 assay following treatment with 0-105 nM of SG3299 or SG3511. The IC50 of v6-targeted SG3299 in TB32043mb6s2 was over 15-fold lower than in TB32043 cells (24 nM vs 418 nM, p < 0.001). There was no significant difference in the IC50 values for the non-targeted SG3511 between TB32043mb6s2 and TB32043 cells (223 vs 300 nM, p = 0.17).
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Pancreatic ductal adenocarcinoma cell TB32043 | Homo sapiens | ||
References
