Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00119
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
cLABL
|
|||||
| Structure |
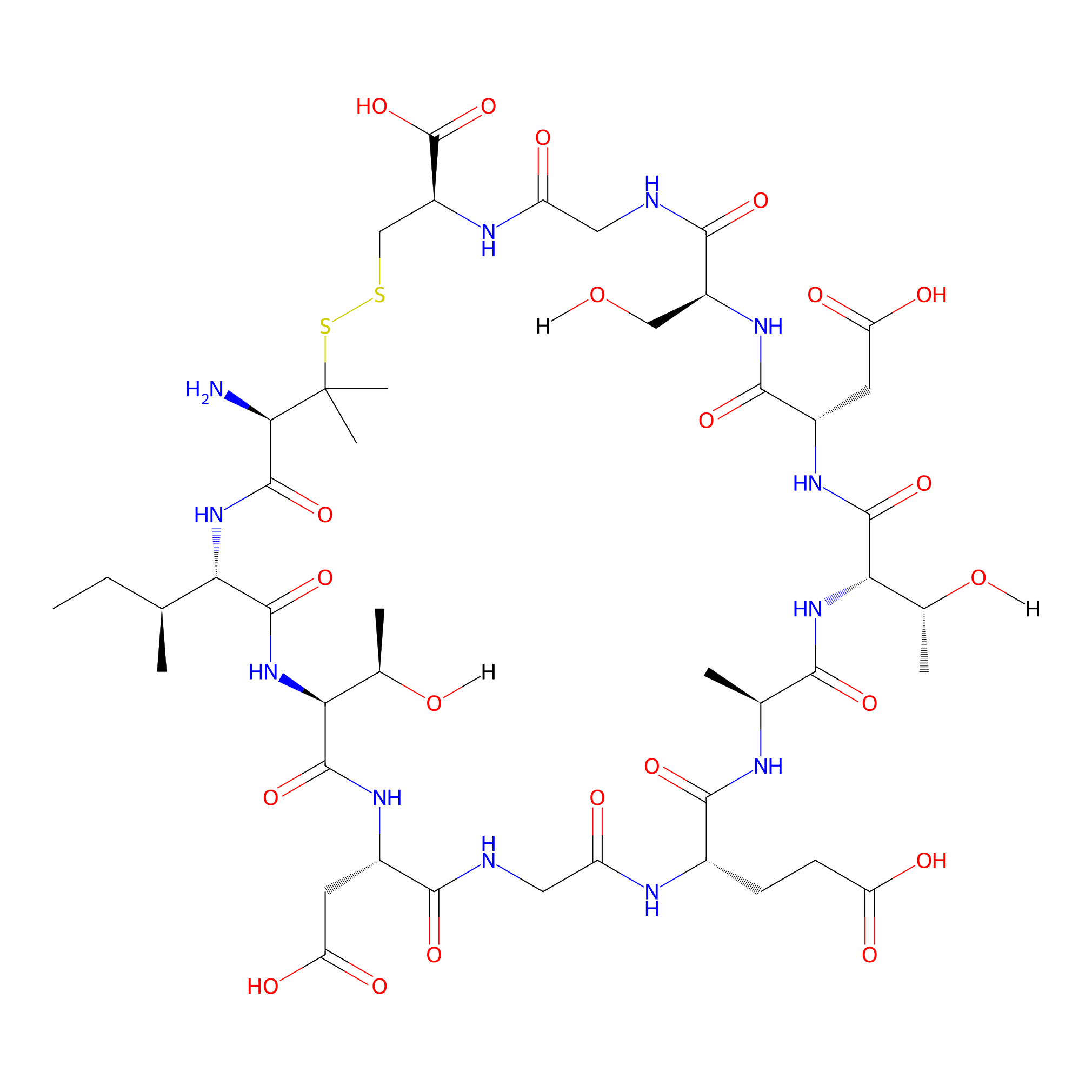
|
|||||
| Sequence |
Penicillamine-ITDGEATDSGC
|
|||||
| Peptide Type |
Cyclic
|
|||||
| Receptor Name |
Intercellular adhesion molecule 1 (ICAM1)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C45H72N12O22S2
|
|||||
| Isosmiles |
[H]OC[C@@H]1NC(=O)[C@H](CC(=O)O)NC(=O)[C@]([H])([C@@H](C)O[H])NC(=O)[C@H](C)NC(=O)[C@H](CCC(=O)O)NC(=O)CNC(=O)[C@H](CC(=O)O)NC(=O)[C@]([H])([C@@H](C)O[H])NC(=O)[C@]([H])([C@@H](C)CC)NC(=O)[C@@H](N)C(C)(C)SSC[C@@H](C(=O)O)NC(=O)CNC1=O
|
|||||
| InChI |
InChI=1S/C45H72N12O22S2/c1-8-17(2)31-40(74)57-33(20(5)60)42(76)52-22(11-29(65)66)36(70)47-13-26(61)50-21(9-10-28(63)64)38(72)49-18(3)35(69)56-32(19(4)59)41(75)53-23(12-30(67)68)39(73)54-24(15-58)37(71)48-14-27(62)51-25(44(78)79)16-80-81-45(6,7)34(46)43(77)55-31/h17-25,31-34,58-60H,8-16,46H2,1-7H3,(H,47,70)(H,48,71)(H,49,72)(H,50,61)(H,51,62)(H,52,76)(H,53,75)(H,54,73)(H,55,77)(H,56,69)(H,57,74)(H,63,64)(H,65,66)(H,67,68)(H,78,79)/t17-,18-,19+,20+,21-,22-,23-,24-,25-,31-,32-,33-,34+/m0/s1
|
|||||
| InChIKey |
RNJRLNAFZZTPJM-QMUSQRDESA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
1197.267
|
Polar area
|
556.01
|
||
|
Complexity
|
1196.432554
|
xlogp Value
|
-8.1984
|
|||
|
Heavy Count
|
81
|
Rot Bonds
|
16
|
|||
|
Hbond acc
|
21
|
Hbond Donor
|
19
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
MTX-cLABL [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Inflammation | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 1 h | ||||
| Evaluation Method | Propidium iodide (PI) assay | ||||
| MOA of PDC |
In this study, cLABL and cLBEL peptides were linked to methotrexate (MTX) to produce MTX-cLABL and MTX-cLBEL conjugates. The goal was to target MTX to human coronary artery endothelial cells (HCAEC) via the ICAM-1 receptor to lower MTX toxicity and side effects. The biological abilities of MTX-cLABL, MTX-cLBEL, cLABL, cLBEL, and MTX were compared by their activities to inhibit binding of anti-ICAM-1 mAb to ICAM-1 on the surface of HCAEC. In addition, these molecules were compared in inhibiting T cell adhesion to HCAEC monolayers. Finally, their activities in suppressing IL-6 and IL-8 production as inflammatory cytokines were determined. The toxicities of MTX-cLABL and MTX-cLBEL conjugates were also determined relative to MTX alone as well as cLABL and cLBEL peptides.
Click to Show/Hide
|
||||
| Description |
We next determined whether treatment of HCAEC and Molt-3 T cells with peptides, MTX, and MTX-peptide conjugates resulted in inhibition of cell proliferation. Both HCAEC and Molt-3 T cells were affected by test compound in different levels. None of the molecules caused growth stimulation or total culture extinction. A net cell killing of HCAEC was observed upon treatment with MTX at all test concentrations while MTX affected net killing at ≥1.0 uM in Molt-3 T cells. The MTX-peptide conjugates were less toxic than MTX. In HCAEC, the net cell killing was at lower concentration for MTX at ≥0.1 uM compared to MTX-peptide conjugates at ≥500 uM. The net cell killing of Molt-3 T cells was found at ≥1.0 uM for MTX and ≥50 uM for MTX-peptide conjugates. For all test concentrations, the conjugates only resulted in HCAEC partial growth inhibition. For Molt-3 T cells, a total growth inhibition emerged at 100 uM for cLABL and cLBEL; however, 500 uM cLABL and cLBEL did not cause total cell killing for T cells.
Click to Show/Hide
|
||||
| In Vitro Model | human coronary artery endothelial cell | Human coronary artery endothelial cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Inflammation | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 500 µM | |||
| Administration Time | 1 h | ||||
| Evaluation Method | Propidium iodide (PI) assay | ||||
| MOA of PDC |
In this study, cLABL and cLBEL peptides were linked to methotrexate (MTX) to produce MTX-cLABL and MTX-cLBEL conjugates. The goal was to target MTX to human coronary artery endothelial cells (HCAEC) via the ICAM-1 receptor to lower MTX toxicity and side effects. The biological abilities of MTX-cLABL, MTX-cLBEL, cLABL, cLBEL, and MTX were compared by their activities to inhibit binding of anti-ICAM-1 mAb to ICAM-1 on the surface of HCAEC. In addition, these molecules were compared in inhibiting T cell adhesion to HCAEC monolayers. Finally, their activities in suppressing IL-6 and IL-8 production as inflammatory cytokines were determined. The toxicities of MTX-cLABL and MTX-cLBEL conjugates were also determined relative to MTX alone as well as cLABL and cLBEL peptides.
Click to Show/Hide
|
||||
| Description |
We next determined whether treatment of HCAEC and Molt-3 T cells with peptides, MTX, and MTX-peptide conjugates resulted in inhibition of cell proliferation. Both HCAEC and Molt-3 T cells were affected by test compound in different levels. None of the molecules caused growth stimulation or total culture extinction. A net cell killing of HCAEC was observed upon treatment with MTX at all test concentrations while MTX affected net killing at ≥1.0 uM in Molt-3 T cells. The MTX-peptide conjugates were less toxic than MTX. In HCAEC, the net cell killing was at lower concentration for MTX at ≥0.1 uM compared to MTX-peptide conjugates at ≥500 uM. The net cell killing of Molt-3 T cells was found at ≥1.0 uM for MTX and ≥50 uM for MTX-peptide conjugates. For all test concentrations, the conjugates only resulted in HCAEC partial growth inhibition. For Molt-3 T cells, a total growth inhibition emerged at 100 uM for cLABL and cLBEL; however, 500 uM cLABL and cLBEL did not cause total cell killing for T cells.
Click to Show/Hide
|
||||
| In Vitro Model | Adult T acute lymphoblastic leukemia | Molt-3 T cell | CVCL_0624 | ||
References
