Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00147
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
IDR-1018
|
|||||
| Structure |
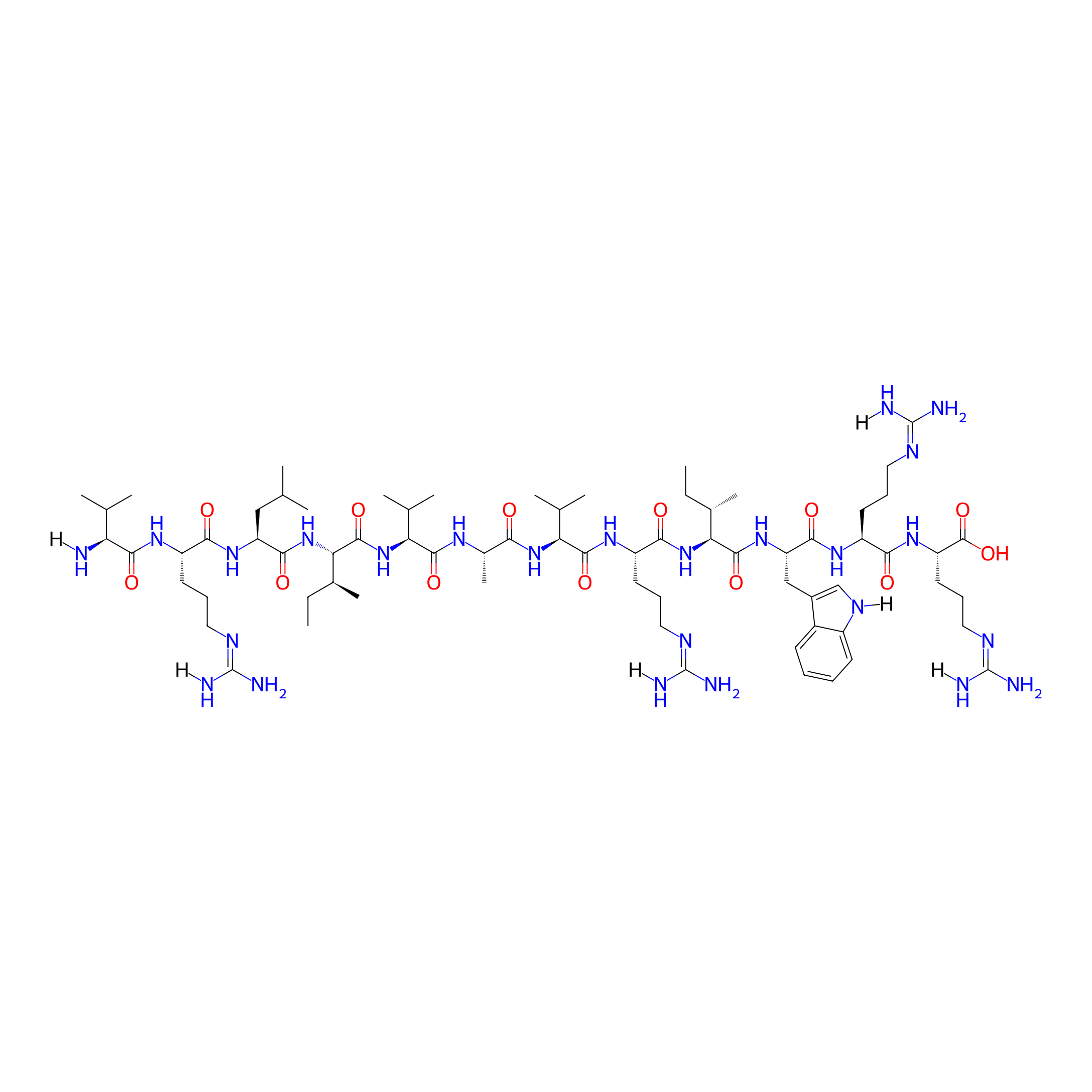
|
|||||
| Sequence |
VRLIVAVRIWRR
|
|||||
| Peptide Type |
Linear
|
|||||
| PDC Transmembrane Types | Cell-penetrating peptides (CPPs) | |||||
| Formula |
C71H125N25O13
|
|||||
| Isosmiles |
[H]N/C(N)=N\CCC[C@H](NC(=O)[C@H](CCC/N=C(/N)N[H])NC(=O)[C@H](Cc1cn([H])c2ccccc12)NC(=O)[C@@]([H])(NC(=O)[C@H](CCC/N=C(/N)N[H])NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@@]([H])(NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCC/N=C(/N)N[H])NC(=O)[C@@H](N[H])C(C)C)[C@@H](C)CC)C(C)C)C(C)C)[C@@H](C)CC)C(=O)O
|
|||||
| InChI |
InChI=1S/C71H125N25O13/c1-14-39(11)54(65(106)92-50(33-42-34-85-44-23-17-16-22-43(42)44)60(101)87-45(24-18-28-81-68(73)74)57(98)90-48(67(108)109)27-21-31-84-71(79)80)95-59(100)47(26-20-30-83-70(77)78)89-64(105)53(38(9)10)93-56(97)41(13)86-63(104)52(37(7)8)94-66(107)55(40(12)15-2)96-61(102)49(32-35(3)4)91-58(99)46(25-19-29-82-69(75)76)88-62(103)51(72)36(5)6/h16-17,22-23,34-41,45-55,85H,14-15,18-21,24-33,72H2,1-13H3,(H,86,104)(H,87,101)(H,88,103)(H,89,105)(H,90,98)(H,91,99)(H,92,106)(H,93,97)(H,94,107)(H,95,100)(H,96,102)(H,108,109)(H4,73,74,81)(H4,75,76,82)(H4,77,78,83)(H4,79,80,84)/t39-,40-,41-,45-,46-,47-,48-,49-,50-,51-,52-,53-,54-,55-/m0/s1
|
|||||
| InChIKey |
WGSNSTOKOBNSSK-MUOSHHONSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
1536.943
|
Polar area
|
656.81
|
||
|
Complexity
|
1535.988869
|
xlogp Value
|
-3.2043
|
|||
|
Heavy Count
|
109
|
Rot Bonds
|
51
|
|||
|
Hbond acc
|
17
|
Hbond Donor
|
22
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
Cq-C4-IDR-1018 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 10 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| Description |
Only three of the Cq-C4-CPP conjugates, namely, 5a, 5b, and 5g, displayed IC50 values below 10 μM, with TP10- and Transportan-derived conjugates 5a (IC50 = 1.52 μM) and 5b (IC50 = 5.20 μM) being the most active.
|
||||
| In Vivo Model | Plasmodium falciparum W2. | ||||
References
