Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00148
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
rL-A9
|
|||||
| Structure |
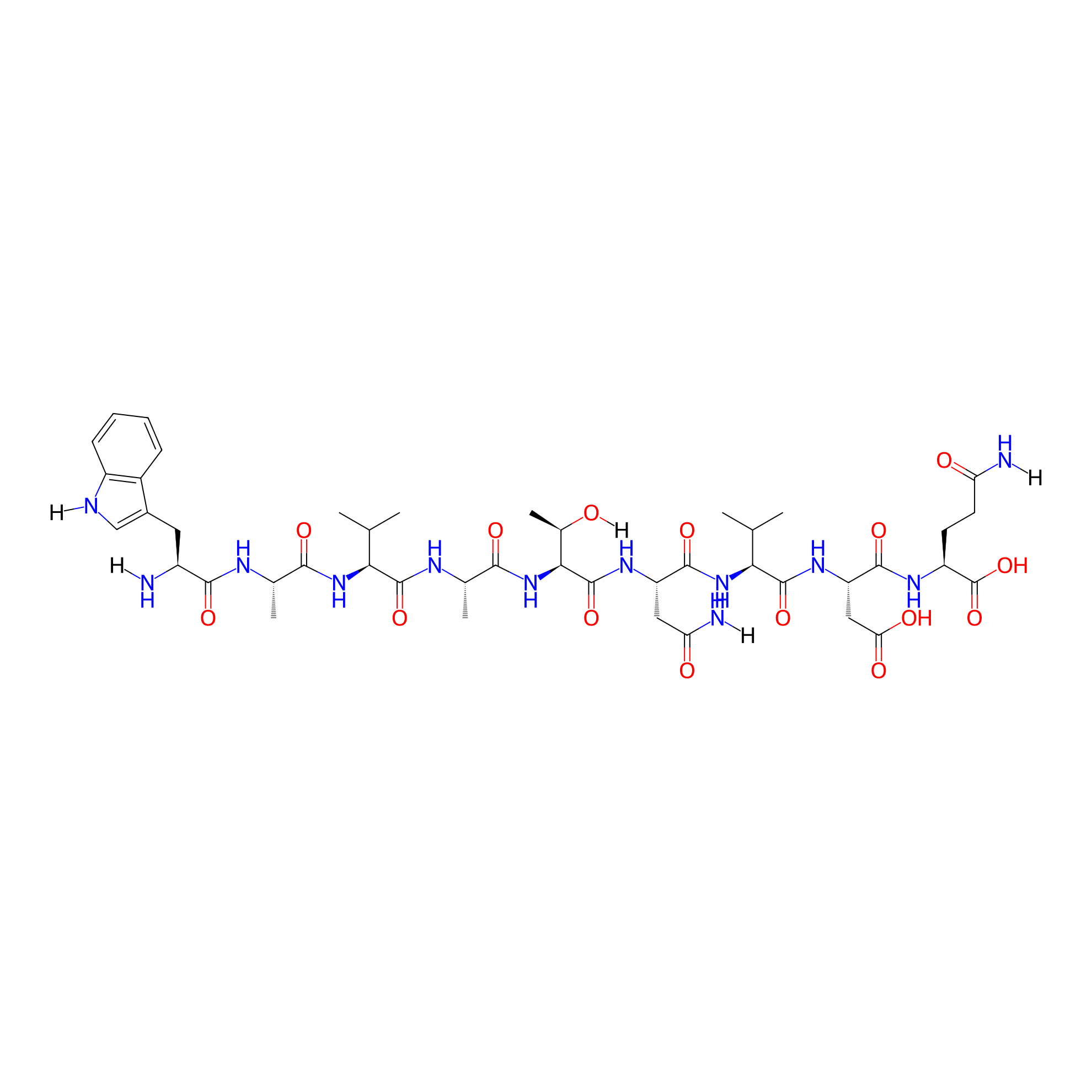
|
|||||
| Sequence |
WAVATNVDQ-NH2
|
|||||
| Peptide Type |
Linear
|
|||||
| Receptor Name |
Receptor tyrosine-protein kinase erbB-2 (ERBB2)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C44H66N12O15
|
|||||
| Isosmiles |
[H]NC(=O)CC[C@H](NC(=O)[C@H](CC(=O)O)NC(=O)[C@@H](NC(=O)[C@H](CC(=O)N[H])NC(=O)[C@@]([H])(NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](Cc1cn([H])c2ccccc12)N[H])C(C)C)[C@@H](C)O[H])C(C)C)C(=O)O
|
|||||
| InChI |
InChI=1S/C44H66N12O15/c1-18(2)33(54-36(62)20(5)49-38(64)25(45)14-23-17-48-26-11-9-8-10-24(23)26)41(67)50-21(6)37(63)56-35(22(7)57)43(69)52-28(15-31(47)59)40(66)55-34(19(3)4)42(68)53-29(16-32(60)61)39(65)51-27(44(70)71)12-13-30(46)58/h8-11,17-22,25,27-29,33-35,48,57H,12-16,45H2,1-7H3,(H2,46,58)(H2,47,59)(H,49,64)(H,50,67)(H,51,65)(H,52,69)(H,53,68)(H,54,62)(H,55,66)(H,56,63)(H,60,61)(H,70,71)/t20-,21-,22+,25-,27-,28-,29-,33-,34-,35-/m0/s1
|
|||||
| InChIKey |
KFEVXBPTHGZMLQ-OZKZYCRBSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
1003.081
|
Polar area
|
455.62
|
||
|
Complexity
|
1002.477059
|
xlogp Value
|
-4.6511
|
|||
|
Heavy Count
|
71
|
Rot Bonds
|
31
|
|||
|
Hbond acc
|
14
|
Hbond Donor
|
15
|
|||
The Activity Data of This Peptide
| Peptide Activity Information 1 | [1] | |||||
| Binding free energy | -22.2 kJ/mol | |||||
|---|---|---|---|---|---|---|
| Binding Affinity Assay |
The peptides were prepared and energy minimized using the ACD ChemSketch suite (ACD/ChemSketch for Academic and Personal Use: ACD/Labs.com, 2018). The protein was prepared by adding hydrogens using AutoDockTools 4. (27) The active site cavity was selected and used a grid box of 20 20 20 for docking, spanning the peptide binding region on the HER2 surface. Molecular docking studies were performed using AutoDock Vina 1.1 with its scoring functions. (28) The top 10 peptide poses were generated after docking. The binding free energies of the best 10 ligand poses bound to the receptor were obtained, and these poses were analyzed.
Click to Show/Hide
|
|||||
| Experimental Condition | The model of HER2-DIVMP/peptide complexes | |||||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
rL-A9-DOX [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
7%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 15 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
13%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 15 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
26%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 7.5 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
33%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 7.5 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
49%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 3.75 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
67%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 1.87 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
87%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 3.75 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
91%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.94 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
94%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 1.87 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
95%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.47 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
96%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.94 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
97%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.47 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
References
