Peptide Information
General Information of This Peptide
| Peptide ID |
PEP01092
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
Peptide 18-4 analog
|
|||||
| Structure |
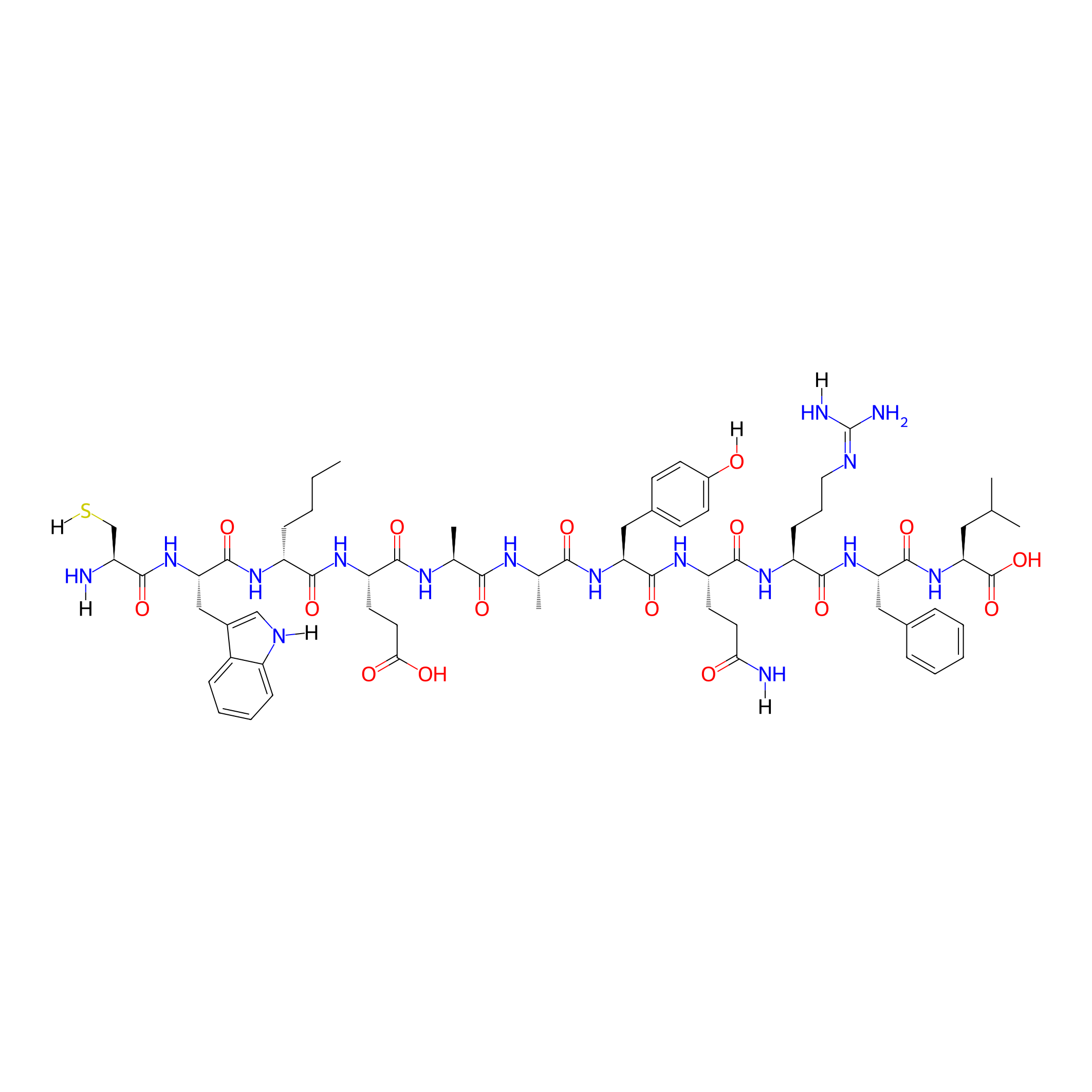
|
|||||
| Sequence |
CWXEAAYQRFL
|
|||||
| Peptide Type |
Linear
|
|||||
| Receptor Name |
Keratin, type II cytoskeletal 1 (KRT1)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C66H94N16O16S
|
|||||
| Isosmiles |
[H]NC(=O)CC[C@H](NC(=O)[C@H](Cc1ccc(O[H])cc1)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(=O)O)NC(=O)[C@@H](CCCC)NC(=O)[C@H](Cc1cn([H])c2ccccc12)NC(=O)[C@H](CS[H])N[H])C(=O)N[C@@H](CCC/N=C(/N)N[H])C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CC(C)C)C(=O)O
|
|||||
| InChI |
InChI=1S/C66H94N16O16S/c1-6-7-17-45(76-64(96)51(80-57(89)43(67)34-99)32-40-33-72-44-18-12-11-16-42(40)44)59(91)77-48(25-27-54(85)86)58(90)74-36(4)55(87)73-37(5)56(88)79-49(31-39-20-22-41(83)23-21-39)62(94)78-47(24-26-53(68)84)61(93)75-46(19-13-28-71-66(69)70)60(92)81-50(30-38-14-9-8-10-15-38)63(95)82-52(65(97)98)29-35(2)3/h8-12,14-16,18,20-23,33,35-37,43,45-52,72,83,99H,6-7,13,17,19,24-32,34,67H2,1-5H3,(H2,68,84)(H,73,87)(H,74,90)(H,75,93)(H,76,96)(H,77,91)(H,78,94)(H,79,88)(H,80,89)(H,81,92)(H,82,95)(H,85,86)(H,97,98)(H4,69,70,71)/t36-,37-,43-,45+,46-,47-,48-,49-,50-,51-,52-/m0/s1
|
|||||
| InChIKey |
GTDNMWZARMRSJA-HEFKSMCPSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
1399.641
|
Polar area
|
535.13
|
||
|
Complexity
|
1398.675442
|
xlogp Value
|
-1.4489
|
|||
|
Heavy Count
|
99
|
Rot Bonds
|
46
|
|||
|
Hbond acc
|
17
|
Hbond Donor
|
19
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
Peptide 18-4 analog doxorubicin conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
75%
|
|||
| Administration Time | 30 days | ||||
| Administration Dosage | 2.5 mg DOX equivalent/kg | ||||
| MOA of PDC |
Keratin 1 (K1) is a novel receptor, present on the surface of cancer cells (breast and neuroblastoma) and cells that have undergone oxidative stress, that is being used for targeted drug delivery. We showed that K1 is present on the surface of MCF-7 breast cancer cells, and a comparison of the total K1 levels in cell lysates using Western blot showed that cancer cells (MCF-7 and MDA-MB-435) have a much higher expression of K1 compared to non-cancerous breast tissue derived epithelial (MCF-10A) cells. We engineered peptides, such as linear 18-4 and cyclic analogues, for specific uptake by breast cancer cells (MCF-7 and MDA-MB-231) via cell surface K1 mediated endocytosis. Further, K1 targeting linear peptide 18-4 was used to synthesize four peptide-doxorubicin conjugates with different linker chemistries, such as ester, amide, succinimidyl thioether, and hydrazone. We showed specific uptake of the targeted PDCs via receptor mediated endocytosis in MCF-7 and MDA-MB-435-MDR cancer cells. The PDCs with K1 targeting peptide 18-4 were more cytotoxic to TNBC cells (IC50 1.2-4.7 μM) compared to non-cancerous human mammary epithelial MCF-10A cells (IC50 15.1-38.6 μM), while free drug (doxorubicin) was equally cytotoxic to both cancer and non-cancerous cells (IC50 0.24-1.5 μM). To explore the in vivo efficacy and evaluate the potential of K1 targeting PDC for TNBC treatment, we report here the antitumor activity of one of these peptide-doxorubicin conjugates, where the peptide (18-4) is conjugated to Dox via an acid-sensitive N-acyl hydrazone linker in a mouse model for TNBC. TNBC MDA-MB-231 cells were subcutaneously injected into female NOD/SCID mice to generate TNBC cell-derived xenograft models. Mice treated with the conjugate showed better efficacy, pharmacokinetics, and safety profile compared to the Dox treated mice, supporting the future clinical development of K1 targeted PDCs for treatment of TNBC.
Click to Show/Hide
|
||||
| Description |
After the tumor xenografts reached a volume of around 100150 mm3, mice were randomized into three groups (n = 7), namely, saline (negative control), free doxorubicin (positive control), and hydrazone PDC. A low dose of 2.5 mg/kg Dox or 2.5 mg/kg Dox equivalent for PDC was chosen to study the antitumor efficacy in vivo. Mice were intravenously administered treatment by tail vein every seventh day for six doses. Compared to the saline group, the PDC reduced tumor growth significantly (3.8 times) on day 35 after treatment, whereas the reduction of tumor growth after free Dox treatment was 2.5 times, suggesting the PDC, at the same equivalent dose, was more potent than the free Dox. In addition, the mice treated with PDC remained in overall good health condition, as evidenced by the general appearance, behavior, diet consumption, and body weight. On day 32 during the treatment period, there were no significant differences observed between the PDC and saline groups in the average body weight (p > 0.05). However, the mice treated with Dox showed significant body weight loss (reduced by 11.2%) compared to the PDC group. Twenty-four hours after the last treatment with PDC or free Dox, mice were euthanized. Mice treated with saline were euthanized on day 32 because of the tumor size per IACUC policy and the conditions for euthanasia. Tumor and other major tissues were collected and weighted for further analysis. The mice with PDC treatment exhibited a greater reduction (three times reduction compared to saline) of tumor weight compared to that of free Dox treated (two times reduction compared to saline).
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.2 µM
|
|||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that the cytotoxicity of both conjugates 1 and 2 (IC50 = 1.3 and 2.2 uM, respectively), as well as the free Dox (IC50 = 1.5 uM) on MDA-MB-231 breast cancer cell line, were in the low micromolar range (Figure 4). For the breast cancer cell line MDA-MB-468, the free Dox (IC50 = 0.35 uM) was slightly more toxic compared to conjugates 1 (4.7 uM) and 2 (1.2 uM). For the non-cancerous cell line MCF 10A, the free Dox was highly toxic (IC50 = 0.24 uM) whereas conjugates 1 and 2 displayed much-reduced toxicity (IC50 = 38.6 and 15.1 uM, respectively).
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-468 cell | CVCL_0419 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
2.2 µM
|
|||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that the cytotoxicity of both conjugates 1 and 2 (IC50 = 1.3 and 2.2 uM, respectively), as well as the free Dox (IC50 = 1.5 uM) on MDA-MB-231 breast cancer cell line, were in the low micromolar range (Figure 4). For the breast cancer cell line MDA-MB-468, the free Dox (IC50 = 0.35 uM) was slightly more toxic compared to conjugates 1 (4.7 uM) and 2 (1.2 uM). For the non-cancerous cell line MCF 10A, the free Dox was highly toxic (IC50 = 0.24 uM) whereas conjugates 1 and 2 displayed much-reduced toxicity (IC50 = 38.6 and 15.1 uM, respectively).
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
15.1 µM
|
|||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that the cytotoxicity of both conjugates 1 and 2 (IC50 = 1.3 and 2.2 uM, respectively), as well as the free Dox (IC50 = 1.5 uM) on MDA-MB-231 breast cancer cell line, were in the low micromolar range (Figure 4). For the breast cancer cell line MDA-MB-468, the free Dox (IC50 = 0.35 uM) was slightly more toxic compared to conjugates 1 (4.7 uM) and 2 (1.2 uM). For the non-cancerous cell line MCF 10A, the free Dox was highly toxic (IC50 = 0.24 uM) whereas conjugates 1 and 2 displayed much-reduced toxicity (IC50 = 38.6 and 15.1 uM, respectively).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MCF-10A cell | CVCL_0598 | ||
References
