Peptide Information
General Information of This Peptide
| Peptide ID |
PEP01103
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
AHNP
|
|||||
| Structure |
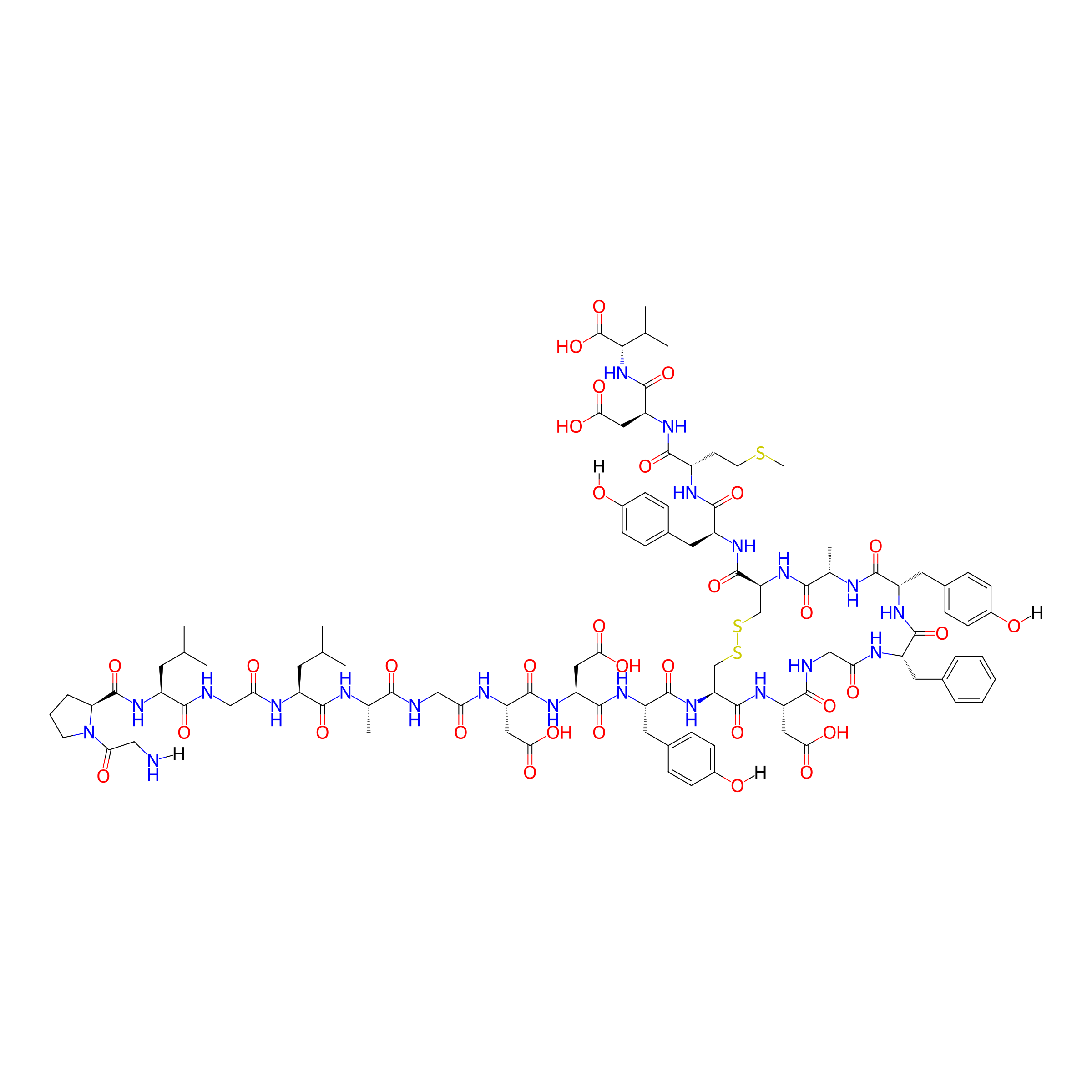
|
|||||
| Sequence |
GPLGLAGDDYCDGFYACYMDV
|
|||||
| Peptide Type |
Linear
|
|||||
| Receptor Name |
Receptor tyrosine-protein kinase erbB-2 (ERBB2)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C99H135N21O33S3
|
|||||
| Isosmiles |
[H]NCC(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)NCC(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](Cc1ccc(O[H])cc1)C(=O)N[C@H]1CSSC[C@@H](C(=O)N[C@@H](Cc2ccc(O[H])cc2)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(=O)O)C(=O)N[C@H](C(=O)O)C(C)C)NC(=O)[C@H](C)NC(=O)[C@H](Cc2ccc(O[H])cc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)CNC(=O)[C@H](CC(=O)O)NC1=O
|
|||||
| InChI |
InChI=1S/C99H135N21O33S3/c1-48(2)32-61(116-98(151)73-16-13-30-120(73)77(127)42-100)85(138)102-44-75(125)106-62(33-49(3)4)88(141)104-51(7)83(136)101-43-74(124)108-68(39-79(130)131)93(146)115-69(40-80(132)133)94(147)111-66(37-56-21-27-59(123)28-22-56)92(145)118-72-47-156-155-46-71(96(149)112-65(36-55-19-25-58(122)26-20-55)91(144)109-60(29-31-154-9)87(140)114-70(41-81(134)135)95(148)119-82(50(5)6)99(152)153)117-84(137)52(8)105-89(142)64(35-54-17-23-57(121)24-18-54)110-90(143)63(34-53-14-11-10-12-15-53)107-76(126)45-103-86(139)67(38-78(128)129)113-97(72)150/h10-12,14-15,17-28,48-52,60-73,82,121-123H,13,16,29-47,100H2,1-9H3,(H,101,136)(H,102,138)(H,103,139)(H,104,141)(H,105,142)(H,106,125)(H,107,126)(H,108,124)(H,109,144)(H,110,143)(H,111,147)(H,112,149)(H,113,150)(H,114,140)(H,115,146)(H,116,151)(H,117,137)(H,118,145)(H,119,148)(H,128,129)(H,130,131)(H,132,133)(H,134,135)(H,152,153)/t51-,52-,60-,61-,62-,63-,64-,65-,66-,67-,68-,69-,70-,71-,72-,73-,82-/m0/s1
|
|||||
| InChIKey |
LSCLRSGVJCEZPD-NQLCMFIOSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
2243.484
|
Polar area
|
846.42
|
||
|
Complexity
|
2241.869329
|
xlogp Value
|
-5.9514
|
|||
|
Heavy Count
|
156
|
Rot Bonds
|
56
|
|||
|
Hbond acc
|
32
|
Hbond Donor
|
28
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
MAHNP-Dox conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
110.1 ± 12.7 nM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
You et al. developed a unique approach for the treatment of human epidermal growth factor receptor 2 (HER2)-positive breast cancer. A peptide fragment from the heavy chain 3 of the full-length antibody trastuzumab was obtained and termed AHNP. The 12-mer AHNP binds the extracellular domain of HER2 with high affinity and displays similar potency as trastuzumab. The peptide mimetic AHNP was then conjugated via an MMP-2 sensitive linker with doxorubicin (Dox) to form MAHNP-Dox conjugate essentially using ester/amide bonds.
Click to Show/Hide
|
||||
| Description |
Conjugate MAHNP-Dox was compared to Dox using HER2 positive breast cancer cell lines BT474 and SKBR3. Cellular toxicity analysis showed that MAHNP-Dox was more potent than free Dox, as indicated by lower IC50 values for both cell lines. The BT474 and SKBR3 cell lines had an IC50 of 746.8 ± 81.5 nM and 110.1 ± 12.7 nM for MAHNP-DOX and 2075.0 ± 368.0 nM and 172.9 ± 19.2 nM for Dox, respectively.
Click to Show/Hide
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
746.8 ± 81.5 nM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
You et al. developed a unique approach for the treatment of human epidermal growth factor receptor 2 (HER2)-positive breast cancer. A peptide fragment from the heavy chain 3 of the full-length antibody trastuzumab was obtained and termed AHNP. The 12-mer AHNP binds the extracellular domain of HER2 with high affinity and displays similar potency as trastuzumab. The peptide mimetic AHNP was then conjugated via an MMP-2 sensitive linker with doxorubicin (Dox) to form MAHNP-Dox conjugate essentially using ester/amide bonds.
Click to Show/Hide
|
||||
| Description |
Conjugate MAHNP-Dox was compared to Dox using HER2 positive breast cancer cell lines BT474 and SKBR3. Cellular toxicity analysis showed that MAHNP-Dox was more potent than free Dox, as indicated by lower IC50 values for both cell lines. The BT474 and SKBR3 cell lines had an IC50 of 746.8 ± 81.5 nM and 110.1 ± 12.7 nM for MAHNP-DOX and 2075.0 ± 368.0 nM and 172.9 ± 19.2 nM for Dox, respectively.
Click to Show/Hide
|
||||
References
