Peptide Information
General Information of This Peptide
| Peptide ID |
PEP01104
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
Potent somatostatin analog (SSA)
|
|||||
| Structure |
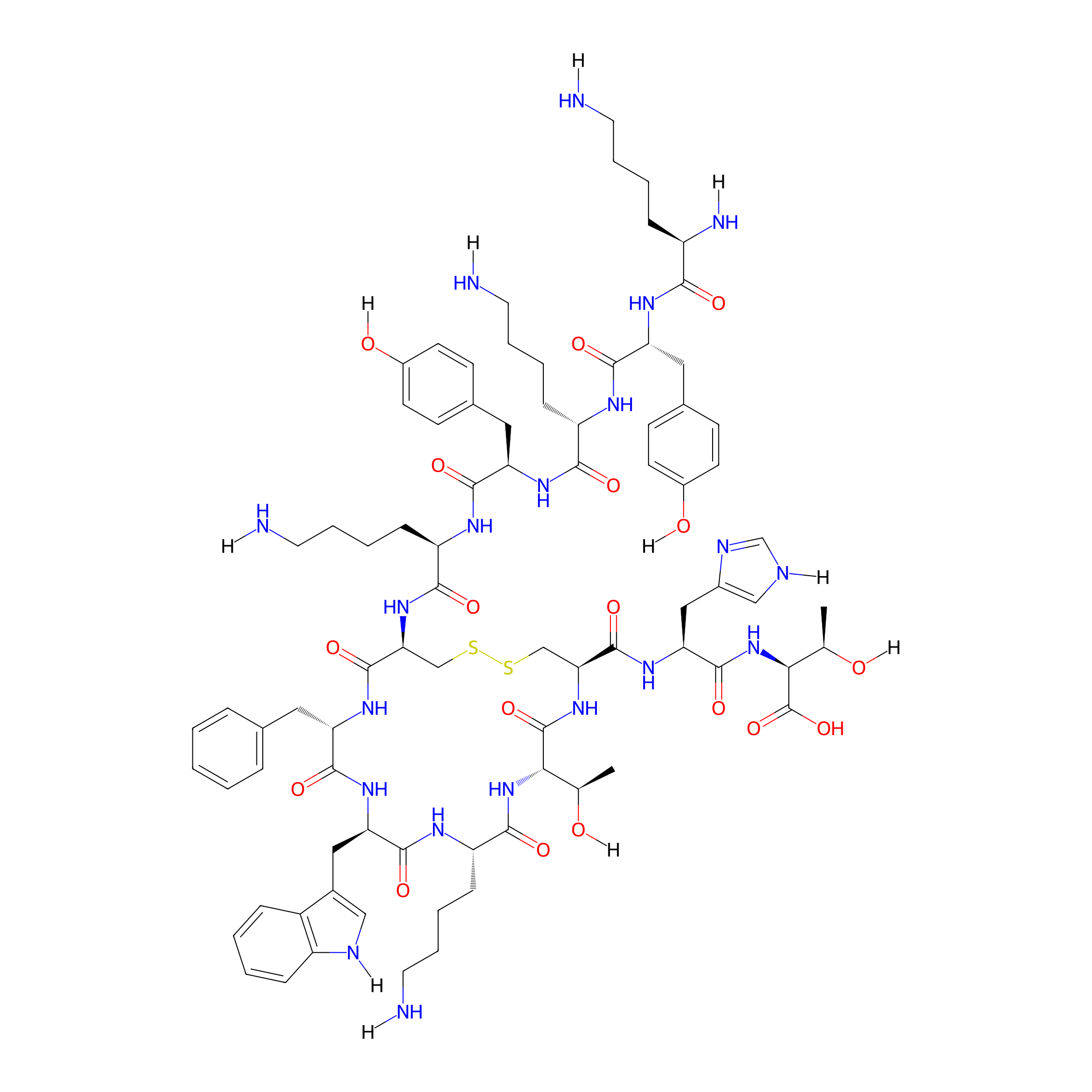
|
|||||
| Sequence |
KYKYKCFWKTCHT
|
|||||
| Peptide Type |
Cyclic
|
|||||
| Receptor Name |
Somatostatin receptor type 2 (SSTR2)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C82H116N20O18S2
|
|||||
| Isosmiles |
[H]NCCCC[C@@H]1NC(=O)[C@@H](Cc2cn([H])c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@@H](CCCCN[H])NC(=O)[C@@H](Cc2ccc(O[H])cc2)NC(=O)[C@H](CCCCN[H])NC(=O)[C@@H](Cc2ccc(O[H])cc2)NC(=O)[C@@H](CCCCN[H])N[H])CSSC[C@@H](C(=O)N[C@@H](Cc2cn([H])cn2)C(=O)N[C@]([H])(C(=O)O)[C@@H](C)O[H])NC(=O)[C@]([H])([C@@H](C)O[H])NC1=O
|
|||||
| InChI |
InChI=1S/C82H116N20O18S2/c1-46(103)68-81(118)100-67(80(117)98-65(40-52-42-88-45-90-52)78(115)102-69(47(2)104)82(119)120)44-122-121-43-66(79(116)96-62(36-48-16-4-3-5-17-48)76(113)97-64(39-51-41-89-57-20-7-6-18-55(51)57)77(114)93-60(73(110)101-68)23-11-15-35-86)99-72(109)59(22-10-14-34-85)92-75(112)63(38-50-26-30-54(106)31-27-50)95-71(108)58(21-9-13-33-84)91-74(111)61(37-49-24-28-53(105)29-25-49)94-70(107)56(87)19-8-12-32-83/h3-7,16-18,20,24-31,41-42,45-47,56,58-69,89,103-106H,8-15,19,21-23,32-40,43-44,83-87H2,1-2H3,(H,88,90)(H,91,111)(H,92,112)(H,93,114)(H,94,107)(H,95,108)(H,96,116)(H,97,113)(H,98,117)(H,99,109)(H,100,118)(H,101,110)(H,102,115)(H,119,120)/t46-,47-,56-,58+,59-,60+,61-,62+,63-,64-,65+,66+,67+,68+,69+/m1/s1
|
|||||
| InChIKey |
PVRQZZUGMBLRML-DQQUOFAQSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
1734.086
|
Polar area
|
641.99
|
||
|
Complexity
|
1732.821789
|
xlogp Value
|
-2.2824
|
|||
|
Heavy Count
|
122
|
Rot Bonds
|
52
|
|||
|
Hbond acc
|
25
|
Hbond Donor
|
24
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
JF-10-71 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1], [2] | ||||
| Indication | Neuroblastoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
1363 nM
|
|||
| Administration Time | 3 days | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In a previous study, we used a potent somatostatin analog (SSA) with high affinity for SSTR2 for attachment to an antisense peptide nucleic acid (PNA) directed against the n-myc oncogene and showed that PNA-SSA conjugates produced receptor-specific inhibition of cell growth. Cleavable-linker chemistry was then developed that allowed this approach to be extended to well-known cytotoxic compounds such as camptothecin and combretastatin. In the present report two CPT-SSA conjugates, JF-10-71 and JF-10-81, displaying differing stabilities were chosen to potentially treat SSTR2-positive rat pancreatic CA20948 tumors in Lewis rats and SCLC NCI-H69 tumors in athymic nude mice.
Click to Show/Hide
|
||||
| Description |
Previously, a series of CPT-SSA conjugates were tested for their stability in phosphate buffer/rat plasma and in vitro inhibitory activity in tumor cell growth (data not shown). Two of these conjugates, JF-10-71 and JF-10-81, displaying higher and lower stability, respectively, were chosen for further experiments in serial tumor cell lines. Also, free CPT and SSA itself were tested as controls. The results (Table 1) show that the IC50 values increased compared with CPT to JF-10-81 and further to JF-10-71 with the exception of the CA20948 cells. CPT and both conjugates were particularly effective in SSTR2-overexpressing IMR32 cells displaying IC50 values 3.1, 64.13, and 282.50 nM, respectively. SSTR2-overexpressing CA20948 cells were poorly responsive to CPT itself (IC50, 3077 nM); however, its somatostatin conjugates were actually more potent (IC50: JF-10-81, 1790 nM; JF-10-71, 1363 nM). SSA itself exhibited little effect on growth of all tested cell lines even at doses up to 10-5 M.
Click to Show/Hide
|
||||
JF-10-81 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1], [2] | ||||
| Indication | Neuroblastoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
1790 nM
|
|||
| Administration Time | 3 days | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In a previous study, we used a potent somatostatin analog (SSA) with high affinity for SSTR2 for attachment to an antisense peptide nucleic acid (PNA) directed against the n-myc oncogene and showed that PNA-SSA conjugates produced receptor-specific inhibition of cell growth. Cleavable-linker chemistry was then developed that allowed this approach to be extended to well-known cytotoxic compounds such as camptothecin and combretastatin. In the present report two CPT-SSA conjugates, JF-10-71 and JF-10-81, displaying differing stabilities were chosen to potentially treat SSTR2-positive rat pancreatic CA20948 tumors in Lewis rats and SCLC NCI-H69 tumors in athymic nude mice.
Click to Show/Hide
|
||||
| Description |
Previously, a series of CPT-SSA conjugates were tested for their stability in phosphate buffer/rat plasma and in vitro inhibitory activity in tumor cell growth (data not shown). Two of these conjugates, JF-10-71 and JF-10-81, displaying higher and lower stability, respectively, were chosen for further experiments in serial tumor cell lines. Also, free CPT and SSA itself were tested as controls. The results (Table 1) show that the IC50 values increased compared with CPT to JF-10-81 and further to JF-10-71 with the exception of the CA20948 cells. CPT and both conjugates were particularly effective in SSTR2-overexpressing IMR32 cells displaying IC50 values 3.1, 64.13, and 282.50 nM, respectively. SSTR2-overexpressing CA20948 cells were poorly responsive to CPT itself (IC50, 3077 nM); however, its somatostatin conjugates were actually more potent (IC50: JF-10-81, 1790 nM; JF-10-71, 1363 nM). SSA itself exhibited little effect on growth of all tested cell lines even at doses up to 10-5 M.
Click to Show/Hide
|
||||
References
