Drug Information
General Information of This Drug
| Drug ID | DRG00007 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Temozolomide | |||||
| Synonyms |
temozolomide; 85622-93-1; Methazolastone; Temodar; Temodal; 3-methyl-4-oxo-3,4-dihydroimidazo[5,1-d][1,2,3,5]tetrazine-8-carboxamide; Temozolamide; Sch 52365; CCRG-81045; Temozolomidum [Latin]; Temozolodida [Spanish]; Temozolomidum; CCRG 81045; Sch-52365; NSC 362856; M&B 39831; M&B-39831; NSC-362856; CCRIS 8996; MB 39831; 8-Carbamoyl-3-methylimidazo(5,1-d)-1,2,3,5-tetrazin-4(3H)-one; M & B 39831; BRN 5547136; M-39831; 3,4-Dihydro-3-methyl-4-oxoimidazo(5,1-d)-1,2,3,5-tetrazine-8-carboxamide; 3-Methyl-4-oxo-3,4-dihydroimidazo(5,1-d)(1,2,3,5)tetrazine-8-carboxamide; NSC362856; CHEMBL810; 3,4-Dihydro-3-methyl-4-oxoimidazo(5,1-d)-as-tetrazine-8-carboxamide; 3-methyl-4-oxo-3H,4H-imidazo[4,3-d][1,2,3,5]tetrazine-8-carboxamide; MLS002701861; YF1K15M17Y; DTXSID5043714; CHEBI:72564; Mk-7365; 3,4-dihydro-3-methyl-4-oxoimidazo[5,1-d]-1,2,3,5-tetrazine-8-carboxamide; Imidazo[5,1-d]-1,2,3,5-tetrazine-8-carboxamide, 3,4-dihydro-3-methyl-4-oxo-; MFCD00866492; TMZ; NCGC00167429-01; Temozolodida; 8-CARBAMOYL-3-METHYLIMIDAZO[5,1-D]-1,2,3,5-TETRAZIN-4(3H)-ONE; DTXCID3023714; C6H6N6O2; 3,4-Dihydro-3-methyl-4-oxoimidazo[5,1-d][1,2,3,5]tetrazine-8-carboxamide; 3-methyl-4-oxo-3,4-dihydroimidazo[5,1-d][1,2,3,5]tetrazine-8-carboxamide.; 3-Methyl-4-oxo-3,4-dihydro-imidazo[5,1-d][1,2,3,5]tetrazine-8-carboxylic acid amide; SMR000466338; Temodal (TN); Temodar (TN); CAS-85622-93-1; Temozolomide, VETRANAL(TM), analytical standard; SR-01000759347; temozolomida; UNII-YF1K15M17Y; Temozolomide (JAN/USAN/INN); Temozolomide [USAN:INN:BAN]; 3,4-Dihydro-3-methyl-4-oxoimidazo[5,1-d]-as-tetrazine-8-carboxamide; 3-methyl-4-oxoimidazo[5,1-d][1,2,3,5]tetrazine-8-carboxamide; IMIDAZO(5,1-D)-1,2,3,5-TETRAZINE-8-CARBOXAMIDE, 3,4-DIHYDRO-3-METHYL-4-OXO-; TEMOZOLIMIDE; Temozolomide- Bio-X; TEMOZOLOMIDE [MI]; TEMOZOLOMIDE [INN]; TEMOZOLOMIDE [JAN]; Temodar (TN) (Schering); TEMOZOLOMIDE [USAN]; SCHEMBL3739; 4-methyl-5-oxo-2,3,4,6,8-pentazabicyclo[4.3.0]nona-2,7,9-triene-9-carboxamide; TEMOZOLOMIDE [VANDF]; TEMOZOLOMIDE [MART.]; MLS000759447; MLS001424028; BIDD:GT0204; TEMOZOLOMIDE [USP-RS]; TEMOZOLOMIDE [WHO-DD]; GTPL7301; TEMOZOLOMIDE [EMA EPAR]; Temozolomide, >=98% (HPLC); GLXC-02560; HMS2051O12; HMS2090B08; HMS2232N13; HMS3264I14; HMS3269P05; HMS3372K13; HMS3393O12; HMS3413D06; HMS3654N05; HMS3677D06; HMS3713H16; Pharmakon1600-01502289; TEMOZOLOMIDE [ORANGE BOOK]; TEMOZOLOMIDE [EP MONOGRAPH]; 3-methyl-4-oxo-imidazo[5,1-d][1,2,3,5]tetrazine-8-carboxamide; ALBB-021358; BCP03692; TEMOZOLOMIDE [USP MONOGRAPH]; Tox21_112433; AC-758; BDBM50034562; DL-190; NSC759883; s1237; STK623541; 3-Methyl-4-oxo-3,4-dihydroimidazo-[5,1-d][1,2,3,5]tetrazine-8-carboxamide; 3-Methyl-4-oxo-3,4-dihydroimidazo[5,1-d][1,2,3,5]tetraazine-8-carboxamide; AKOS005557098; Tox21_112433_1; CCG-100870; CS-0943; DB00853; KS-1216; NC00120; NSC-759883; Imidazo(5,1-d)(1,2,3,5)tetrazine-8-carboxamide, 3,4-dihydro-3-methyl-4-oxo-; NCGC00167429-02; NCGC00167429-04; NCGC00167429-05; BP-25388; BT164447; HY-17364; NCI60_003316; BCP0726000154; Temozolomide 100 microg/mL in Acetonitrile; AM20110227; FT-0630936; FT-0674845; NS00004198; SW197500-4; T2744; D06067; EN300-122324; AB00639915-06; AB00639915-08; AB00639915-09; AB00639915_10; AB00639915_11; A841386; Q425088; Q-201786; SR-01000759347-4; SR-01000759347-5; BRD-K32107296-001-04-5; Z1201620684; 3-methyl-4-oxo-8-imidazo[5,1-d][1,2,3,5]tetrazinecarboxamide; Temozolomide, United States Pharmacopeia (USP) Reference Standard; 3-methyl-4-oxidanylidene-imidazo[5,1-d][1,2,3,5]tetrazine-8-carboxamide; 3-METHYL-4-OXO-8-IMIDAZOLO[5,1-D][1,2,3,5]TETRAZINECARBOXAMIDE; 3-Methyl-8-aminocarbonyl-imidazo[5,1-d]-1,2,3,5-tetrazin-4(3H)-one; Imidazo[5,2,3,5-tetrazine-8-carboxamide, 3,4-dihydro-3-methyl-4-oxo-; {Imidazo[5,1-d]-1,2,3,5-tetrazine-8-carboxamide,} 3, 4-dihydro-3-methyl-4-oxo-; Temozolomide, Pharmaceutical Secondary Standard; Certified Reference Material; 3-Methyl-4-oxo-3,4-dihydro-imidazo[5,1-d][1,2,3,5]tetrazine-8-carboxylic acid amide (Temozolomide); 3-Methyl-4-oxo-3,4-dihydro-imidazo[5,1-d][1,2,3,5]tetrazine-8-carboxylic acid amide(Temozolomide)
Click to Show/Hide
|
|||||
| Target(s) | Human Deoxyribonucleic acid (hDNA) | Target Info | ||||
| Structure |
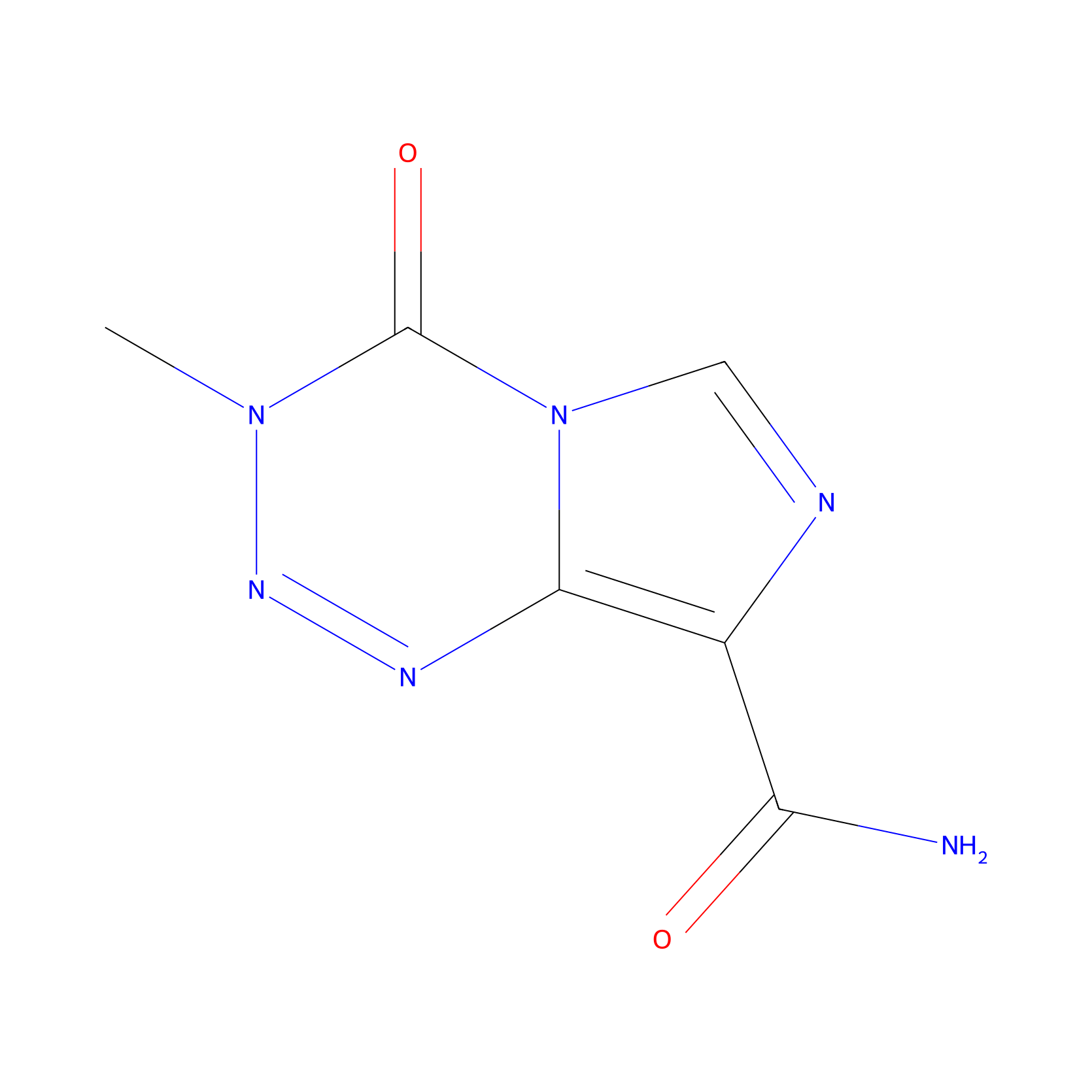
|
|||||
| Formula |
C6H6N6O2
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 194.15 | ||||
| Lipid-water partition coefficient (xlogp) | -1.1 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | |||||
| Rotatable Bond Count (rotbonds) | 1 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CN1C(=O)N2C=NC(=C2N=N1)C(=O)N
|
|||||
| InChI |
InChI=1S/C6H6N6O2/c1-11-6(14)12-2-8-3(4(7)13)5(12)9-10-11/h2H,1H3,(H2,7,13)
|
|||||
| InChIKey |
BPEGJWRSRHCHSN-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
3-methyl-4-oxoimidazo[5,1-d][1,2,3,5]tetrazine-8-carboxamide
|
|||||
The activity data of This Drug
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
67Ga-MMC(TMZ)-TOC [Investigative]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic serotonin-producing neuroendocrine tumor | ||||
| Efficacy Data | Surviving fraction | 80% | |||
| Evaluation Method | Colony-forming assay | ||||
| Administration Time | 5 days | ||||
| Administration Dosage | 2 μmol/L | ||||
| MOA of PDC |
In this study, we hypothesized that the clinically utilized radiopharmaceutical 68Ga-DOTA-TOC could be converted to a radiolabeled peptide-drug conjugate (PDC) for selective delivery of TMZ to SSTR2-positive tumor cells. We used the intrinsic radiolabeling utility of the PDC to characterize receptor-binding properties, pharmacokinetics (PK), and tissue biodistribution in cells and animal studies. Receptor-targeted DNA-damaging properties and cytotoxic effects of the PDC were examined in different cell lines, and the effects of accumulated doses on MGMT content were evaluated in MGMT-positive cells.
Click to Show/Hide
|
||||
| Description |
TMZ is an alkylating agent that causes cell death through DNA-damaging mechanisms.?To determine whether MMC(TMZ)-TOC induces the same cytotoxic effects through a receptor-dependent process, we first studied its DNA-damaging properties in IMR-32 cells that endogenously express SSTR2. We performed an alkaline comet assay to evaluate the ability of our drug conjugate to produce single- and double-stranded DNA breaks. As shown in?Figure 3A,B, the targeted agent-induced DNA breaks that were similar to free TMZ, resulting in migrating DNA fragments (comet tail) from the nucleoid (comet head). The damaging effects of MMC(TMZ)-TOC were significantly reduced (P?< 0.0001) when cells were preincubated with blocking doses of the high-affinity SSTR2 antagonist JR11, indicating receptor-dependent cytotoxicity. Next, we used a colony formation assay to assess the long-term survival and reproductive capability of the cells after treatment with the PDC. We treated BON1 (receptor-negative) and BON1-SSTR2 (receptor-positive) cells with 2 mu;mol/L MMC(TMZ)-TOC or 10 mu;mol/L TMZ for 5 consecutive days, seeded the cells at a low-density number, and allowed them to grow and form colonies for 14 days. The clonogenic survival data showed that MMC(TMZ)-TOC produced cytotoxic effects in the receptor-positive BON1-SSTR2 cells (P?< 0.01) similar to clinically relevant doses of TMZ. Conversely, no cytotoxicity was observed in receptor-negative BON1 cells following treatment with the drug conjugate, further illustrating the receptor-dependent effects of the PDC.
Click to Show/Hide
|
||||
| In Vivo Model | Dually implanted HCT116-WT/SSTR2 mice xenograft models. | ||||
| In Vitro Model | Pancreatic serotonin-producing neuroendocrine tumor | BON-1 cell | CVCL_3985 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic serotonin-producing neuroendocrine tumor | ||||
| Efficacy Data | Surviving fraction | 100% | |||
| Evaluation Method | Colony-forming assay | ||||
| Administration Time | 5 days | ||||
| Administration Dosage | 2 μmol/L | ||||
| MOA of PDC |
In this study, we hypothesized that the clinically utilized radiopharmaceutical 68Ga-DOTA-TOC could be converted to a radiolabeled peptide-drug conjugate (PDC) for selective delivery of TMZ to SSTR2-positive tumor cells. We used the intrinsic radiolabeling utility of the PDC to characterize receptor-binding properties, pharmacokinetics (PK), and tissue biodistribution in cells and animal studies. Receptor-targeted DNA-damaging properties and cytotoxic effects of the PDC were examined in different cell lines, and the effects of accumulated doses on MGMT content were evaluated in MGMT-positive cells.
Click to Show/Hide
|
||||
| Description |
TMZ is an alkylating agent that causes cell death through DNA-damaging mechanisms.?To determine whether MMC(TMZ)-TOC induces the same cytotoxic effects through a receptor-dependent process, we first studied its DNA-damaging properties in IMR-32 cells that endogenously express SSTR2. We performed an alkaline comet assay to evaluate the ability of our drug conjugate to produce single- and double-stranded DNA breaks. As shown in?Figure 3A,B, the targeted agent-induced DNA breaks that were similar to free TMZ, resulting in migrating DNA fragments (comet tail) from the nucleoid (comet head). The damaging effects of MMC(TMZ)-TOC were significantly reduced (P?< 0.0001) when cells were preincubated with blocking doses of the high-affinity SSTR2 antagonist JR11, indicating receptor-dependent cytotoxicity. Next, we used a colony formation assay to assess the long-term survival and reproductive capability of the cells after treatment with the PDC. We treated BON1 (receptor-negative) and BON1-SSTR2 (receptor-positive) cells with 2 mu;mol/L MMC(TMZ)-TOC or 10 mu;mol/L TMZ for 5 consecutive days, seeded the cells at a low-density number, and allowed them to grow and form colonies for 14 days. The clonogenic survival data showed that MMC(TMZ)-TOC produced cytotoxic effects in the receptor-positive BON1-SSTR2 cells (P?< 0.01) similar to clinically relevant doses of TMZ. Conversely, no cytotoxicity was observed in receptor-negative BON1 cells following treatment with the drug conjugate, further illustrating the receptor-dependent effects of the PDC.
Click to Show/Hide
|
||||
| In Vivo Model | Dually implanted HCT116-WT/SSTR2 mice xenograft models. | ||||
| In Vitro Model | Pancreatic serotonin-producing neuroendocrine tumor | BON-1 cell | CVCL_3985 | ||
MMC(TMZ)-TOC [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Neuroendocrine tumour | ||||
| Efficacy Data | Survival rate | 80% | |||
| Evaluation Method | Colony-forming assay | ||||
| Administration Time | 4-6 h/day for 5 consecutive days | ||||
| Administration Dosage | 2 µmol/L | ||||
| MOA of PDC |
In this study, we hypothesized that the clinically utilized radiopharmaceutical 68Ga-DOTA-TOC could be converted to a radiolabeled peptide-drug conjugate (PDC) for selective delivery of TMZ to SSTR2-positive tumor cells. We used the intrinsic radiolabeling utility of the PDC to characterize receptor-binding properties, pharmacokinetics (PK), and tissue biodistribution in cells and animal studies. Receptor-targeted DNA-damaging properties and cytotoxic effects of the PDC were examined in different cell lines, and the effects of accumulated doses on MGMT content were evaluated in MGMT-positive cells.
Click to Show/Hide
|
||||
| Description |
TMZ is an alkylating agent that causes cell death through DNA-damaging mechanisms. To determine whether MMC(TMZ)-TOC induces the same cytotoxic effects through a receptor-dependent process, we first studied its DNA-damaging properties in IMR-32 cells that endogenously express SSTR2. We performed an alkaline comet assay to evaluate the ability of our drug conjugate to produce single- and double-stranded DNA breaks. As shown in Figure 3A,B, the targeted agent-induced DNA breaks that were similar to free TMZ, resulting in migrating DNA fragments (comet tail) from the nucleoid (comet head). The damaging effects of MMC(TMZ)-TOC were significantly reduced (P < 0.0001) when cells were preincubated with blocking doses of the high-affinity SSTR2 antagonist JR11, indicating receptor-dependent cytotoxicity. Next, we used a colony formation assay to assess the long-term survival and reproductive capability of the cells after treatment with the PDC. We treated BON1 (receptor-negative) and BON1-SSTR2 (receptor-positive) cells with 2 umol/L MMC(TMZ)-TOC or 10 umol/L TMZ for 5 consecutive days, seeded the cells at a low-density number, and allowed them to grow and form colonies for 14 days. The clonogenic survival data showed that MMC(TMZ)-TOC produced cytotoxic effects in the receptor-positive BON1-SSTR2 cells (P < 0.01) similar to clinically relevant doses of TMZ. Conversely, no cytotoxicity was observed in receptor-negative BON1 cells following treatment with the drug conjugate, further illustrating the receptor-dependent effects of the PDC.
Click to Show/Hide
|
||||
| In Vitro Model | SSR2 positive neuroendocrine tumour | BON1-SSTR2 cell | CVCL_3985 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Neuroendocrine tumour | ||||
| Efficacy Data | Survival rate | 100% | |||
| Evaluation Method | Colony-forming assay | ||||
| Administration Time | 4-6 h/day for 5 consecutive days | ||||
| Administration Dosage | 2 µmol/L | ||||
| MOA of PDC |
In this study, we hypothesized that the clinically utilized radiopharmaceutical 68Ga-DOTA-TOC could be converted to a radiolabeled peptide-drug conjugate (PDC) for selective delivery of TMZ to SSTR2-positive tumor cells. We used the intrinsic radiolabeling utility of the PDC to characterize receptor-binding properties, pharmacokinetics (PK), and tissue biodistribution in cells and animal studies. Receptor-targeted DNA-damaging properties and cytotoxic effects of the PDC were examined in different cell lines, and the effects of accumulated doses on MGMT content were evaluated in MGMT-positive cells.
Click to Show/Hide
|
||||
| Description |
TMZ is an alkylating agent that causes cell death through DNA-damaging mechanisms. To determine whether MMC(TMZ)-TOC induces the same cytotoxic effects through a receptor-dependent process, we first studied its DNA-damaging properties in IMR-32 cells that endogenously express SSTR2. We performed an alkaline comet assay to evaluate the ability of our drug conjugate to produce single- and double-stranded DNA breaks. As shown in Figure 3A,B, the targeted agent-induced DNA breaks that were similar to free TMZ, resulting in migrating DNA fragments (comet tail) from the nucleoid (comet head). The damaging effects of MMC(TMZ)-TOC were significantly reduced (P < 0.0001) when cells were preincubated with blocking doses of the high-affinity SSTR2 antagonist JR11, indicating receptor-dependent cytotoxicity. Next, we used a colony formation assay to assess the long-term survival and reproductive capability of the cells after treatment with the PDC. We treated BON1 (receptor-negative) and BON1-SSTR2 (receptor-positive) cells with 2 umol/L MMC(TMZ)-TOC or 10 umol/L TMZ for 5 consecutive days, seeded the cells at a low-density number, and allowed them to grow and form colonies for 14 days. The clonogenic survival data showed that MMC(TMZ)-TOC produced cytotoxic effects in the receptor-positive BON1-SSTR2 cells (P < 0.01) similar to clinically relevant doses of TMZ. Conversely, no cytotoxicity was observed in receptor-negative BON1 cells following treatment with the drug conjugate, further illustrating the receptor-dependent effects of the PDC.
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic serotonin-producing neuroendocrine tumor | BON-1 cell | CVCL_3985 | ||
References
