Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00036
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
67Ga-MMC(TMZ)-TOC
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
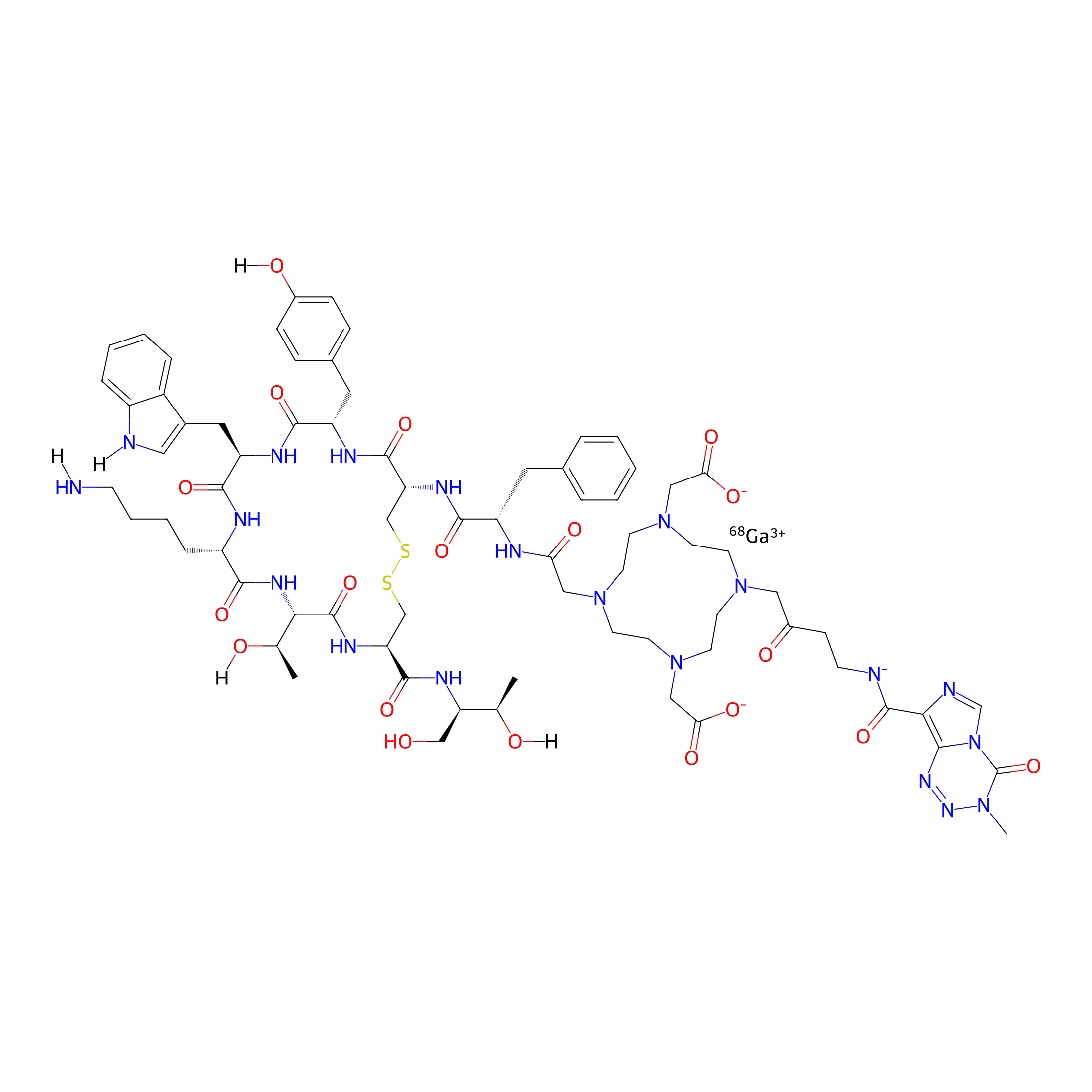
|
|||||
| Peptide Name |
3-Tyr-Octreotide
|
Peptide Info | ||||
| Receptor Name |
Somatostatin receptor type 1 (SSTR1)
|
Receptor Info | ||||
| Drug Name |
Temozolomide
|
Drug Info | ||||
| Therapeutic Target |
Human Deoxyribonucleic acid (hDNA)
|
Target Info | ||||
| Linker Name |
Amide bond
|
Linker Info | ||||
| Peptide Modified Type |
Amino acid modifications; Cyclization modification
|
|||||
| Modified Segment |
D-amino acids
|
|||||
| Ternimal Modification |
N-terminal modification
|
|||||
| Formula |
C73H97GaN20O19S2
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 1690.76198 | ||||
| Lipid-water partition coefficient (xlogp) | -7.292 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 14 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 31 | |||||
| Rotatable Bond Count (rotbonds) | 34 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic serotonin-producing neuroendocrine tumor | ||||
| Efficacy Data | Surviving fraction |
80%
|
|||
| Administration Time | 5 days | ||||
| Administration Dosage | 2 μmol/L | ||||
| Evaluation Method | Colony-forming assay | ||||
| MOA of PDC |
In this study, we hypothesized that the clinically utilized radiopharmaceutical 68Ga-DOTA-TOC could be converted to a radiolabeled peptide-drug conjugate (PDC) for selective delivery of TMZ to SSTR2-positive tumor cells. We used the intrinsic radiolabeling utility of the PDC to characterize receptor-binding properties, pharmacokinetics (PK), and tissue biodistribution in cells and animal studies. Receptor-targeted DNA-damaging properties and cytotoxic effects of the PDC were examined in different cell lines, and the effects of accumulated doses on MGMT content were evaluated in MGMT-positive cells.
Click to Show/Hide
|
||||
| Description |
TMZ is an alkylating agent that causes cell death through DNA-damaging mechanisms.?To determine whether MMC(TMZ)-TOC induces the same cytotoxic effects through a receptor-dependent process, we first studied its DNA-damaging properties in IMR-32 cells that endogenously express SSTR2. We performed an alkaline comet assay to evaluate the ability of our drug conjugate to produce single- and double-stranded DNA breaks. As shown in?Figure 3A,B, the targeted agent-induced DNA breaks that were similar to free TMZ, resulting in migrating DNA fragments (comet tail) from the nucleoid (comet head). The damaging effects of MMC(TMZ)-TOC were significantly reduced (P?< 0.0001) when cells were preincubated with blocking doses of the high-affinity SSTR2 antagonist JR11, indicating receptor-dependent cytotoxicity. Next, we used a colony formation assay to assess the long-term survival and reproductive capability of the cells after treatment with the PDC. We treated BON1 (receptor-negative) and BON1-SSTR2 (receptor-positive) cells with 2 mu;mol/L MMC(TMZ)-TOC or 10 mu;mol/L TMZ for 5 consecutive days, seeded the cells at a low-density number, and allowed them to grow and form colonies for 14 days. The clonogenic survival data showed that MMC(TMZ)-TOC produced cytotoxic effects in the receptor-positive BON1-SSTR2 cells (P?< 0.01) similar to clinically relevant doses of TMZ. Conversely, no cytotoxicity was observed in receptor-negative BON1 cells following treatment with the drug conjugate, further illustrating the receptor-dependent effects of the PDC.
Click to Show/Hide
|
||||
| In Vivo Model | Dually implanted HCT116-WT/SSTR2 mice xenograft models. | ||||
| In Vitro Model | Pancreatic serotonin-producing neuroendocrine tumor | BON-1 cell | CVCL_3985 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic serotonin-producing neuroendocrine tumor | ||||
| Efficacy Data | Surviving fraction |
100%
|
|||
| Administration Time | 5 days | ||||
| Administration Dosage | 2 μmol/L | ||||
| Evaluation Method | Colony-forming assay | ||||
| MOA of PDC |
In this study, we hypothesized that the clinically utilized radiopharmaceutical 68Ga-DOTA-TOC could be converted to a radiolabeled peptide-drug conjugate (PDC) for selective delivery of TMZ to SSTR2-positive tumor cells. We used the intrinsic radiolabeling utility of the PDC to characterize receptor-binding properties, pharmacokinetics (PK), and tissue biodistribution in cells and animal studies. Receptor-targeted DNA-damaging properties and cytotoxic effects of the PDC were examined in different cell lines, and the effects of accumulated doses on MGMT content were evaluated in MGMT-positive cells.
Click to Show/Hide
|
||||
| Description |
TMZ is an alkylating agent that causes cell death through DNA-damaging mechanisms.?To determine whether MMC(TMZ)-TOC induces the same cytotoxic effects through a receptor-dependent process, we first studied its DNA-damaging properties in IMR-32 cells that endogenously express SSTR2. We performed an alkaline comet assay to evaluate the ability of our drug conjugate to produce single- and double-stranded DNA breaks. As shown in?Figure 3A,B, the targeted agent-induced DNA breaks that were similar to free TMZ, resulting in migrating DNA fragments (comet tail) from the nucleoid (comet head). The damaging effects of MMC(TMZ)-TOC were significantly reduced (P?< 0.0001) when cells were preincubated with blocking doses of the high-affinity SSTR2 antagonist JR11, indicating receptor-dependent cytotoxicity. Next, we used a colony formation assay to assess the long-term survival and reproductive capability of the cells after treatment with the PDC. We treated BON1 (receptor-negative) and BON1-SSTR2 (receptor-positive) cells with 2 mu;mol/L MMC(TMZ)-TOC or 10 mu;mol/L TMZ for 5 consecutive days, seeded the cells at a low-density number, and allowed them to grow and form colonies for 14 days. The clonogenic survival data showed that MMC(TMZ)-TOC produced cytotoxic effects in the receptor-positive BON1-SSTR2 cells (P?< 0.01) similar to clinically relevant doses of TMZ. Conversely, no cytotoxicity was observed in receptor-negative BON1 cells following treatment with the drug conjugate, further illustrating the receptor-dependent effects of the PDC.
Click to Show/Hide
|
||||
| In Vivo Model | Dually implanted HCT116-WT/SSTR2 mice xenograft models. | ||||
| In Vitro Model | Pancreatic serotonin-producing neuroendocrine tumor | BON-1 cell | CVCL_3985 | ||
References
