Linker Information
General Information of This Linker
| Linker ID |
LIN00026
|
|||||
|---|---|---|---|---|---|---|
| Linker Name |
Aminocaproic acid
|
|||||
| Structure |
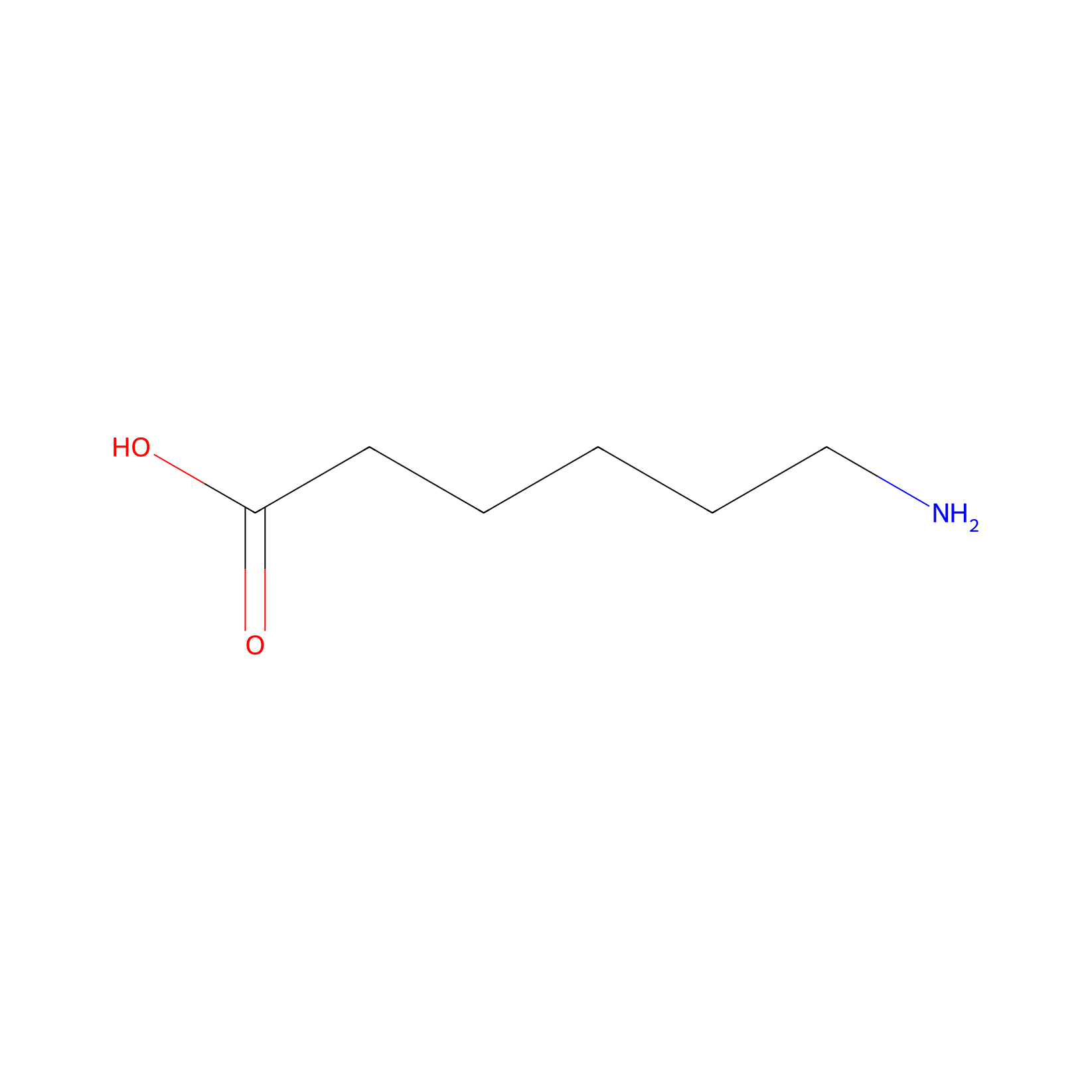
|
|||||
| Formula |
C6H13NO2
|
|||||
| #Ro5 Violations (Lipinski): 1 | Molecular Weight (mw) | 131.17 | ||||
| Lipid-water partition coefficient (xlogp) | -3 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 3 | |||||
| Rotatable Bond Count (rotbonds) | 5 | |||||
| Chemble ID | ||||||
| Chemble ID | ||||||
| Chemble ID | ||||||
| PubChem CID | ||||||
| Canonical smiles |
C(CCC(=O)O)CCN
|
|||||
| InChI |
InChI=1S/C6H13NO2/c7-5-3-1-2-4-6(8)9/h1-5,7H2,(H,8,9)
|
|||||
| InChIKey |
SLXKOJJOQWFEFD-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
6-aminohexanoic acid
|
|||||
Each Peptide-drug Conjugate Related to This Linker
Full Information of The Activity Data of The PDC(s) Related to This Linker
68Ga-PSMA HBED-CC [Investigative]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Stable disease (SD) |
31.60%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
A PSA response was seen in 18 patients (47.4%), stable disease in 12 (31.6%) and progressive disease in 8 (21.1%).
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Response Evaluation Criteria in Solid Tumours (RECIST) |
11.30%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | PSA response |
47.40%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
A PSA response was seen in 18 patients (47.4%), stable disease in 12 (31.6%) and progressive disease in 8 (21.1%).
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Progressive Disease (PD) |
21.10%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
A PSA response was seen in 18 patients (47.4%), stable disease in 12 (31.6%) and progressive disease in 8 (21.1%).
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Mean standardized uptake values (SUVmean) |
26%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Decrease of total tumor volumes (TTV) |
45%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Decrease of median PSA level |
44%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
Sinomenine-synovial homing peptide conjugate (L) [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Rheumatoid arthritis | ||||
| Efficacy Data | Swelling inhibition value |
41.00%
|
|||
| Administration Time | 4 h | ||||
| Administration Dosage | 90.96 mg/kg | ||||
| MOA of PDC |
In this study, we utilized a synovial endothelium targeting peptide (CKSTHDRLC), which was identified and shown to preferentially locate at synovial xenografts in a human/SCID chimeric model of RA using ex vivo and in vivo screening of a phage peptide-display library. This peptide was subsequently developed as a targeting carrier for selective delivery of recombinant antibodies, cytokines, and nanoparticles. In this work, this target peptide was conjugated to SIN with a 6-aminocaproic acid linker to construct peptide-drug conjugates, and their characterization, stability, drugs released in vitro, therapeutic efficacy and in vivo bio-distribution are reported.
Click to Show/Hide
|
||||
| Description |
To assess the anti-inflammatory efficacy of the conjugates, a carrageenan-induced acute inflammation model was developed in Balb/c male mice. Experimental mice were treated by injection of saline, free SIN, conjugate (L) or conjugate (C) in the tail vein at 1 h prior to the induction of paw edema. Photographs of the right hind paws indicated that swelling was milder in the mice treated with conjugate (C) and conjugate (L) than in the model group. Data for hind paw volumes suggested that conjugate (C) had the strongest therapeutic efficacy compared to that of the free drug SIN at 20.96 mg/kg and the model group. As previously reported, effective doses of SIN against experimental arthritis in vivo range from 15 mg/kg to 300 mg/kg body weight. Therefore, the dose of SIN was considered since the administration of 20.96 mg/kg is widely identified in the literature as a low dose for treating animals in arthritis mouse models without inducing side effects. The benefit of IV injections of conjugates at 90.96 mg/kg (eq. SIN) was clearly demonstrated by comparison with free SIN at the same molar dose. To verify the therapeutic efficacies of the conjugates, histological structures of paws were evaluated. Histological examination of the right hind paws revealed that the pedis skins contained massive lymphocyte, plasma cell and neutrophil infiltration in the paws of animals treated with saline. Animals treated with conjugates and free SIN showed less inflammatory cell infiltration, and conjugate (C) showed the mildest inflammation compared to the same dose of free SIN. To characterize the therapeutic efficacies of the conjugates more clearly, Image J software was used to statistically analyze the inflammatory cells in pathological sections (n = 7), and the results are shown in Fig. 6D. There were significant differences between the treatment groups and the model group (*p < 0.05, **p < 0.01, ***p < 0.001).
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c male mice carrageenan-induced acute inflammation model. | ||||
| Half life period | 24.866 h | ||||
Sinomenine-synovial homing peptide conjugate (C) [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Rheumatoid arthritis | ||||
| Efficacy Data | Swelling inhibition value |
58.30%
|
|||
| Administration Time | 4 h | ||||
| Administration Dosage | 90.96 mg/kg | ||||
| MOA of PDC |
In this study, we utilized a synovial endothelium targeting peptide (CKSTHDRLC), which was identified and shown to preferentially locate at synovial xenografts in a human/SCID chimeric model of RA using ex vivo and in vivo screening of a phage peptide-display library. This peptide was subsequently developed as a targeting carrier for selective delivery of recombinant antibodies, cytokines, and nanoparticles. In this work, this target peptide was conjugated to SIN with a 6-aminocaproic acid linker to construct peptide-drug conjugates, and their characterization, stability, drugs released in vitro, therapeutic efficacy and in vivo bio-distribution are reported.
Click to Show/Hide
|
||||
| Description |
To assess the anti-inflammatory efficacy of the conjugates, a carrageenan-induced acute inflammation model was developed in Balb/c male mice. Experimental mice were treated by injection of saline, free SIN, conjugate (L) or conjugate (C) in the tail vein at 1 h prior to the induction of paw edema. Photographs of the right hind paws indicated that swelling was milder in the mice treated with conjugate (C) and conjugate (L) than in the model group. Data for hind paw volumes suggested that conjugate (C) had the strongest therapeutic efficacy compared to that of the free drug SIN at 20.96 mg/kg and the model group. As previously reported, effective doses of SIN against experimental arthritis in vivo range from 15 mg/kg to 300 mg/kg body weight. Therefore, the dose of SIN was considered since the administration of 20.96 mg/kg is widely identified in the literature as a low dose for treating animals in arthritis mouse models without inducing side effects. The benefit of IV injections of conjugates at 90.96 mg/kg (eq. SIN) was clearly demonstrated by comparison with free SIN at the same molar dose. To verify the therapeutic efficacies of the conjugates, histological structures of paws were evaluated. Histological examination of the right hind paws revealed that the pedis skins contained massive lymphocyte, plasma cell and neutrophil infiltration in the paws of animals treated with saline. Animals treated with conjugates and free SIN showed less inflammatory cell infiltration, and conjugate (C) showed the mildest inflammation compared to the same dose of free SIN. To characterize the therapeutic efficacies of the conjugates more clearly, Image J software was used to statistically analyze the inflammatory cells in pathological sections (n = 7), and the results are shown in Fig. 6D. There were significant differences between the treatment groups and the model group (*p < 0.05, **p < 0.01, ***p < 0.001).
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c male mice carrageenan-induced acute inflammation model. | ||||
| Half life period | 24.347 h | ||||
GA-TAT [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Bladder cancer | ||||
| Efficacy Data | Inhibition rate |
46.4% ± 4.86%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 1.0 µM | ||||
| MOA of PDC |
These findings suggest that GA-TAT-induced EJ cell apoptosis is mediated via ROS production. Our study demonstrated that GA-TAT enhanced the pro-apoptotic effect via increasing caspase-3 and caspase-9 processing and activities and decreasing the Bcl-2/Bax ratio, which were regulated by intracellular ROS.
|
||||
| Description |
However, after treatment with GA-TAT, the percentage of apoptotic cells was greatly increased to 46.4%±4.86%.
|
||||
| In Vitro Model | Bladder carcinoma | EJ-1 cell | CVCL_2893 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Bladder cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.24 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
These findings suggest that GA-TAT-induced EJ cell apoptosis is mediated via ROS production. Our study demonstrated that GA-TAT enhanced the pro-apoptotic effect via increasing caspase-3 and caspase-9 processing and activities and decreasing the Bcl-2/Bax ratio, which were regulated by intracellular ROS.
|
||||
| Description |
The 50% inhibitory concentration (IC50) of GA-TAT at 24 h was 1.24 uM
|
||||
| In Vitro Model | Bladder cancer | Bladder cancer cell | Homo sapiens | ||
PDC-Z1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
4.96 ± 0.98 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In the previous study, we focused on the discovery of novel cell targeting peptides, leading to the identification of the preferred HER2-targeting peptide P1 (NPNWGRSWYNQRFK) derived from pertuzumab, with the Kd of 5.70 10-6 M. Here, we designed and synthesized a series of PDCs choosing Camptothecin (CPT) as the payload and P1 as the peptide carrier. We aimed to discover novel PDC molecules with potent inhibitory activity and selectivity targeting HER2 for effective cancer treatment. Conjugate Z8, which balanced the activity and the biosafety, showed good antiproliferative activity (IC50 = 1.91 ± 0.71 M), comparable to that of CPT (IC50 = 2.10 ± 1.34 M), against NCI-N87 cells. Moreover, subsequent verification tests indicated that Z8 operated through a mechanism similar to CPT, but the results from in vivo anti-tumor activity assay suggested that Z8 exhibited greater safety compared to CPT to some extent. These findings highlight Z8 as a promising candidate for further investigation in the treatment of HER2-positive tumors.
Click to Show/Hide
|
||||
| Description |
To evaluated the in vitro anti-tumor activity of the conjugates, HER2-positive SK-BR-3, NCI-N87, HER2-negative MDA-MB-231 and normal MCF-10A cells were selected. As shown in Table 2, nearly all conjugates exhibited obvious anti-proliferation activity against SK-BR-3 cells, while most of the conjugates, except for Z6 and Z9, demonstrated different degrees of inhibitory activity on NCI-N87 cells. Taken together, Z8 and Z11 exhibited superior anti-tumor activity. The antiproliferative activity of Z8 against NCI-N87 cells (IC50 = 1.91 ± 0.71 uM) was comparable to that of CPT (IC50 = 2.10 ± 1.34 uM), while the antiproliferative activity of Z11 against SK-BR-3 cells (IC50 = 0.41 ± 0.13 uM) was comparable to that of CPT (IC50 = 0.26 ± 0.06 uM). Additionally, the IC50 values of these two conjugates against MDA-MB-231 and MCF-10A cells were significantly increased, showing their ability to selectively inhibit tumor cells. Based on the above results, Z8 and Z11, which exhibited better activity and apparent selectivity, were selected for subsequent studies.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | SK-BR-3 cell | CVCL_0033 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Gastric cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
17.94 ± 2.78 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In the previous study, we focused on the discovery of novel cell targeting peptides, leading to the identification of the preferred HER2-targeting peptide P1 (NPNWGRSWYNQRFK) derived from pertuzumab, with the Kd of 5.70 10-6 M. Here, we designed and synthesized a series of PDCs choosing Camptothecin (CPT) as the payload and P1 as the peptide carrier. We aimed to discover novel PDC molecules with potent inhibitory activity and selectivity targeting HER2 for effective cancer treatment. Conjugate Z8, which balanced the activity and the biosafety, showed good antiproliferative activity (IC50 = 1.91 ± 0.71 M), comparable to that of CPT (IC50 = 2.10 ± 1.34 M), against NCI-N87 cells. Moreover, subsequent verification tests indicated that Z8 operated through a mechanism similar to CPT, but the results from in vivo anti-tumor activity assay suggested that Z8 exhibited greater safety compared to CPT to some extent. These findings highlight Z8 as a promising candidate for further investigation in the treatment of HER2-positive tumors.
Click to Show/Hide
|
||||
| Description |
To evaluated the in vitro anti-tumor activity of the conjugates, HER2-positive SK-BR-3, NCI-N87, HER2-negative MDA-MB-231 and normal MCF-10A cells were selected. As shown in Table 2, nearly all conjugates exhibited obvious anti-proliferation activity against SK-BR-3 cells, while most of the conjugates, except for Z6 and Z9, demonstrated different degrees of inhibitory activity on NCI-N87 cells. Taken together, Z8 and Z11 exhibited superior anti-tumor activity. The antiproliferative activity of Z8 against NCI-N87 cells (IC50 = 1.91 ± 0.71 uM) was comparable to that of CPT (IC50 = 2.10 ± 1.34 uM), while the antiproliferative activity of Z11 against SK-BR-3 cells (IC50 = 0.41 ± 0.13 uM) was comparable to that of CPT (IC50 = 0.26 ± 0.06 uM). Additionally, the IC50 values of these two conjugates against MDA-MB-231 and MCF-10A cells were significantly increased, showing their ability to selectively inhibit tumor cells. Based on the above results, Z8 and Z11, which exhibited better activity and apparent selectivity, were selected for subsequent studies.
Click to Show/Hide
|
||||
| In Vitro Model | Gastric tubular adenocarcinoma | NCI-N87 cell | CVCL_1603 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
27.24 ± 1.73 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In the previous study, we focused on the discovery of novel cell targeting peptides, leading to the identification of the preferred HER2-targeting peptide P1 (NPNWGRSWYNQRFK) derived from pertuzumab, with the Kd of 5.70 10-6 M. Here, we designed and synthesized a series of PDCs choosing Camptothecin (CPT) as the payload and P1 as the peptide carrier. We aimed to discover novel PDC molecules with potent inhibitory activity and selectivity targeting HER2 for effective cancer treatment. Conjugate Z8, which balanced the activity and the biosafety, showed good antiproliferative activity (IC50 = 1.91 ± 0.71 M), comparable to that of CPT (IC50 = 2.10 ± 1.34 M), against NCI-N87 cells. Moreover, subsequent verification tests indicated that Z8 operated through a mechanism similar to CPT, but the results from in vivo anti-tumor activity assay suggested that Z8 exhibited greater safety compared to CPT to some extent. These findings highlight Z8 as a promising candidate for further investigation in the treatment of HER2-positive tumors.
Click to Show/Hide
|
||||
| Description |
To evaluated the in vitro anti-tumor activity of the conjugates, HER2-positive SK-BR-3, NCI-N87, HER2-negative MDA-MB-231 and normal MCF-10A cells were selected. As shown in Table 2, nearly all conjugates exhibited obvious anti-proliferation activity against SK-BR-3 cells, while most of the conjugates, except for Z6 and Z9, demonstrated different degrees of inhibitory activity on NCI-N87 cells. Taken together, Z8 and Z11 exhibited superior anti-tumor activity. The antiproliferative activity of Z8 against NCI-N87 cells (IC50 = 1.91 ± 0.71 uM) was comparable to that of CPT (IC50 = 2.10 ± 1.34 uM), while the antiproliferative activity of Z11 against SK-BR-3 cells (IC50 = 0.41 ± 0.13 uM) was comparable to that of CPT (IC50 = 0.26 ± 0.06 uM). Additionally, the IC50 values of these two conjugates against MDA-MB-231 and MCF-10A cells were significantly increased, showing their ability to selectively inhibit tumor cells. Based on the above results, Z8 and Z11, which exhibited better activity and apparent selectivity, were selected for subsequent studies.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MCF-10A cell | CVCL_0598 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
38.72 ± 4.62 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In the previous study, we focused on the discovery of novel cell targeting peptides, leading to the identification of the preferred HER2-targeting peptide P1 (NPNWGRSWYNQRFK) derived from pertuzumab, with the Kd of 5.70 10-6 M. Here, we designed and synthesized a series of PDCs choosing Camptothecin (CPT) as the payload and P1 as the peptide carrier. We aimed to discover novel PDC molecules with potent inhibitory activity and selectivity targeting HER2 for effective cancer treatment. Conjugate Z8, which balanced the activity and the biosafety, showed good antiproliferative activity (IC50 = 1.91 ± 0.71 M), comparable to that of CPT (IC50 = 2.10 ± 1.34 M), against NCI-N87 cells. Moreover, subsequent verification tests indicated that Z8 operated through a mechanism similar to CPT, but the results from in vivo anti-tumor activity assay suggested that Z8 exhibited greater safety compared to CPT to some extent. These findings highlight Z8 as a promising candidate for further investigation in the treatment of HER2-positive tumors.
Click to Show/Hide
|
||||
| Description |
To evaluated the in vitro anti-tumor activity of the conjugates, HER2-positive SK-BR-3, NCI-N87, HER2-negative MDA-MB-231 and normal MCF-10A cells were selected. As shown in Table 2, nearly all conjugates exhibited obvious anti-proliferation activity against SK-BR-3 cells, while most of the conjugates, except for Z6 and Z9, demonstrated different degrees of inhibitory activity on NCI-N87 cells. Taken together, Z8 and Z11 exhibited superior anti-tumor activity. The antiproliferative activity of Z8 against NCI-N87 cells (IC50 = 1.91 ± 0.71 uM) was comparable to that of CPT (IC50 = 2.10 ± 1.34 uM), while the antiproliferative activity of Z11 against SK-BR-3 cells (IC50 = 0.41 ± 0.13 uM) was comparable to that of CPT (IC50 = 0.26 ± 0.06 uM). Additionally, the IC50 values of these two conjugates against MDA-MB-231 and MCF-10A cells were significantly increased, showing their ability to selectively inhibit tumor cells. Based on the above results, Z8 and Z11, which exhibited better activity and apparent selectivity, were selected for subsequent studies.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
QR-KLU [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
7.3 ± 0.74 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | CCK8 assay | ||||
| MOA of PDC |
In this paper, we designed a novel PDC which was a conjugation of VEGFR targeting peptide VEGF125-136 and a lytic peptide. This novel peptide conjugate may not only target VEGFR expressed on endothelial cells and inhibit angiogenesis, but also potently inhibit cancer cell proliferation through destroying cell membrane. As its different mechanism from chemotherapeutics, this PDC has a good potential for chemo-resistance cancer therapy. We recognized this peptide could be a potential drug candidate delivered through TAE for HCC therapy. As we know, there has not been a peptide inhibitor used in combination with TACE for HCC therapy. So we developed a VX2 rabbit tumor model and applied this peptide conjugate to TAE for liver cancer therapy, in which this peptide demonstrated better in vivo anti-tumor and anti-angiogenesis effect than conventional TACE. This work may provide an alternative option in combination with TACE for HCC therapy in the future.
Click to Show/Hide
|
||||
| Description |
The effect of the peptide conjugate QR-KLU on the proliferation of Huh7 and HUVEC cells was assessed by CCK8 assays. First, we investigated anti-proliferation ability of three peptides QR, KLU and QR-KLU on Huh7 and HUVEC cells with different concentrations. As shown in Fig. Fig.3A,3A, B, cells were treated with QR peptide, KLU peptide, QR-KLU peptide, DOX with different concentration respectively. The inhibition rate increased in a dose-dependent manner in KLU and QR-KLU groups in both cell lines. In HUVEC cells, peptide QR-KLU (IC50 10.7 ± 0.292 uM) showed more potent inhibition effect than KLU (IC50 33.8 ± 0.98 uM), and in Huh7 cells, QR-KLU (IC50 7.3 ± 0.74 uM) also showed more potent anti-tumor effect than KLU (IC50 36.27 ± 2.7 uM). Meanwhile, peptide QR showed negligible toxicity even under 80 uM.
Click to Show/Hide
|
||||
| In Vitro Model | Hepatocellular carcinoma | Huh-7 cell | CVCL_0336 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
10.7 ± 0.292 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | CCK8 assay | ||||
| MOA of PDC |
In this paper, we designed a novel PDC which was a conjugation of VEGFR targeting peptide VEGF125-136 and a lytic peptide. This novel peptide conjugate may not only target VEGFR expressed on endothelial cells and inhibit angiogenesis, but also potently inhibit cancer cell proliferation through destroying cell membrane. As its different mechanism from chemotherapeutics, this PDC has a good potential for chemo-resistance cancer therapy. We recognized this peptide could be a potential drug candidate delivered through TAE for HCC therapy. As we know, there has not been a peptide inhibitor used in combination with TACE for HCC therapy. So we developed a VX2 rabbit tumor model and applied this peptide conjugate to TAE for liver cancer therapy, in which this peptide demonstrated better in vivo anti-tumor and anti-angiogenesis effect than conventional TACE. This work may provide an alternative option in combination with TACE for HCC therapy in the future.
Click to Show/Hide
|
||||
| Description |
The effect of the peptide conjugate QR-KLU on the proliferation of Huh7 and HUVEC cells was assessed by CCK8 assays. First, we investigated anti-proliferation ability of three peptides QR, KLU and QR-KLU on Huh7 and HUVEC cells with different concentrations. As shown in Fig. Fig.3A,3A, B, cells were treated with QR peptide, KLU peptide, QR-KLU peptide, DOX with different concentration respectively. The inhibition rate increased in a dose-dependent manner in KLU and QR-KLU groups in both cell lines. In HUVEC cells, peptide QR-KLU (IC50 10.7 ± 0.292 uM) showed more potent inhibition effect than KLU (IC50 33.8 ± 0.98 uM), and in Huh7 cells, QR-KLU (IC50 7.3 ± 0.74 uM) also showed more potent anti-tumor effect than KLU (IC50 36.27 ± 2.7 uM). Meanwhile, peptide QR showed negligible toxicity even under 80 uM.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Apoptosis rate |
40%
|
|||
| Administration Dosage | 10 µM | ||||
| MOA of PDC |
In this paper, we designed a novel PDC which was a conjugation of VEGFR targeting peptide VEGF125-136 and a lytic peptide. This novel peptide conjugate may not only target VEGFR expressed on endothelial cells and inhibit angiogenesis, but also potently inhibit cancer cell proliferation through destroying cell membrane. As its different mechanism from chemotherapeutics, this PDC has a good potential for chemo-resistance cancer therapy. We recognized this peptide could be a potential drug candidate delivered through TAE for HCC therapy. As we know, there has not been a peptide inhibitor used in combination with TACE for HCC therapy. So we developed a VX2 rabbit tumor model and applied this peptide conjugate to TAE for liver cancer therapy, in which this peptide demonstrated better in vivo anti-tumor and anti-angiogenesis effect than conventional TACE. This work may provide an alternative option in combination with TACE for HCC therapy in the future.
Click to Show/Hide
|
||||
| Description |
The improved proapoptotic activity of QR-KLU was further confirmed by Annexin V-PI staining through FACS. Viable cells, early apoptotic cells, necrotic cells, late apoptotic cells were represented by Q4, Q3, Q2 and Q1 respectively. Peptide QR-KLU displayed a significant proapoptotic effect under 10 uM with apoptosis rate over 60% in Huh 7 cells and over 40% in HUVEC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 4 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Apoptosis rate |
60%
|
|||
| Administration Dosage | 10 µM | ||||
| MOA of PDC |
In this paper, we designed a novel PDC which was a conjugation of VEGFR targeting peptide VEGF125-136 and a lytic peptide. This novel peptide conjugate may not only target VEGFR expressed on endothelial cells and inhibit angiogenesis, but also potently inhibit cancer cell proliferation through destroying cell membrane. As its different mechanism from chemotherapeutics, this PDC has a good potential for chemo-resistance cancer therapy. We recognized this peptide could be a potential drug candidate delivered through TAE for HCC therapy. As we know, there has not been a peptide inhibitor used in combination with TACE for HCC therapy. So we developed a VX2 rabbit tumor model and applied this peptide conjugate to TAE for liver cancer therapy, in which this peptide demonstrated better in vivo anti-tumor and anti-angiogenesis effect than conventional TACE. This work may provide an alternative option in combination with TACE for HCC therapy in the future.
Click to Show/Hide
|
||||
| Description |
The improved proapoptotic activity of QR-KLU was further confirmed by Annexin V-PI staining through FACS. Viable cells, early apoptotic cells, necrotic cells, late apoptotic cells were represented by Q4, Q3, Q2 and Q1 respectively. Peptide QR-KLU displayed a significant proapoptotic effect under 10 uM with apoptosis rate over 60% in Huh 7 cells and over 40% in HUVEC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Hepatocellular carcinoma | Huh-7 cell | CVCL_0336 | ||
Linear arginineglycineaspartic acid (RGD) - Naproxen conjugate 4 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate |
0%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [6] | ||||
| Efficacy Data | Cell inhibition rate |
0%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Ovarian serous adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate |
12.00%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate |
57 ± 0.4%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
Linear asparagineglycinearginine (NGR) 1 - Naproxen conjugate 2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate |
0%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [6] | ||||
| Efficacy Data | Cell inhibition rate |
0%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate |
31 ± 2.1%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Cell inhibition rate |
66 ± 0.5%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Ovarian serous cystadenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate |
68 ± 0.9%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
Linear arginineglycineaspartic acid (RGD) - Ibuprofen conjugate 2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate |
0%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [6] | ||||
| Efficacy Data | Cell inhibition rate |
0%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Ovarian serous adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate |
28 ± 3.01%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate |
35 ± 2.1%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
XNRG - Ibuprofen conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate |
0%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [6] | ||||
| Efficacy Data | Cell inhibition rate |
0%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate |
40 ± 3.6%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Ovarian serous cystadenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate |
59 ± 1.1%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Cell inhibition rate |
61 ± 0.8%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
References
