Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00042
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
68Ga-PSMA HBED-CC
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
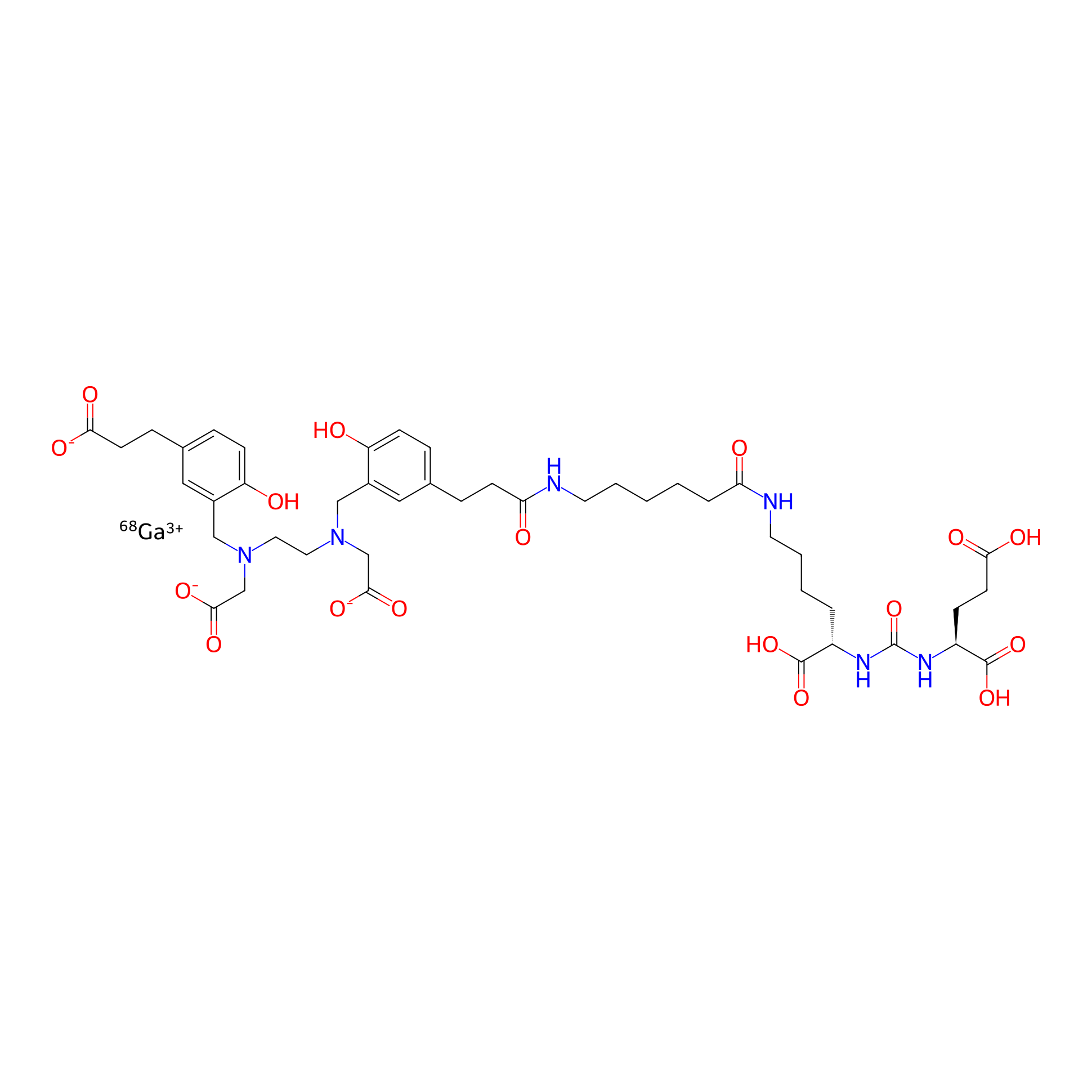
|
|||||
| Peptide Name |
PSMA-617
|
Peptide Info | ||||
| Receptor Name |
Glutamate carboxypeptidase 2 (FOLH1)
|
Receptor Info | ||||
| Drug Name |
Gallium-68
|
Drug Info | ||||
| Therapeutic Target |
Human Deoxyribonucleic acid (hDNA)
|
Target Info | ||||
| Linker Name |
Aminocaproic acid
|
Linker Info | ||||
| Distribution |
DTX-P7 can be detected in the plasma in the first 24 h post treatments with higher area under curve (AUC) and Cmax than DTX. In normal tissues, docetaxel was barely detectable in brain in 1, 2, 4 and 8 h after injection. In the liver, the docetaxel concentration of DTX group was 5 folds and 3 folds higher than that of DTX-P7 group after 1 and 2 h of injection. Similar distribution was also observed in heart and lungs.
|
|||||
| Formula |
C44H59GaN6O17
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 1011.90898 | ||||
| Lipid-water partition coefficient (xlogp) | -2.8304 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 9 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 16 | |||||
| Rotatable Bond Count (rotbonds) | 35 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Decrease of median PSA level |
44%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Decrease of total tumor volumes (TTV) |
45%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Mean standardized uptake values (SUVmean) |
26%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Progressive Disease (PD) |
21.10%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
A PSA response was seen in 18 patients (47.4%), stable disease in 12 (31.6%) and progressive disease in 8 (21.1%).
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | PSA response |
47.40%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
A PSA response was seen in 18 patients (47.4%), stable disease in 12 (31.6%) and progressive disease in 8 (21.1%).
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Response Evaluation Criteria in Solid Tumours (RECIST) |
11.30%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Stable disease (SD) |
31.60%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
A PSA response was seen in 18 patients (47.4%), stable disease in 12 (31.6%) and progressive disease in 8 (21.1%).
|
||||
References
