Drug Information
General Information of This Drug
| Drug ID | DRG00028 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Gallium-68 | |||||
| Synonyms |
Gallium-68; Gallium Ga-68; 15757-14-9; Gallium, isotope of mass 68; 98B30EPP5S; 68Ga; UNII-98B30EPP5S; GA 68; Q2516934
Click to Show/Hide
|
|||||
| Target(s) | Human Deoxyribonucleic acid (hDNA) | Target Info | ||||
| Structure |
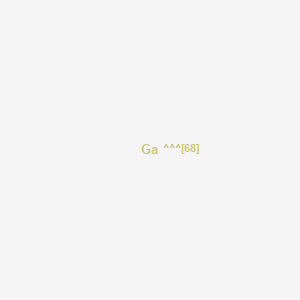
|
|||||
| Formula |
Ga
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 67.927 | ||||
| Lipid-water partition coefficient (xlogp) | Not Available | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 0 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 0 | |||||
| Rotatable Bond Count (rotbonds) | 0 | |||||
| PubChem CID | ||||||
| Canonical smiles |
[Ga]
|
|||||
| InChI |
InChI=1S/Ga/i1-2
|
|||||
| InChIKey |
GYHNNYVSQQEPJS-YPZZEJLDSA-N
|
|||||
| IUPAC Name |
gallium-68
|
|||||
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
68Ga-DOTATATE [Approved]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Neuroendocrine tumour | ||||
| Efficacy Data | Median detection ratio | 75.30% | |||
| Patients Enrolled |
75 patients with histologically confirmed neuroendocrine tumours and routine clinical.
|
||||
| MOA of PDC |
18F-AlF-OC is noninferior and even superior to 68Ga-DOTATATE/NOC PET in NET patients. This validates 18F-AlF-OC as an option for clinical practice somatostatin receptor PET.
|
||||
| Description |
In total, 4,709 different tumor lesions were detected: 3,454 with 68Ga-DOTATATE/NOC and 4,278 with 18F-AlF-OC. The mean DR with 18F-AlF-OC was significantly higher than with 68Ga-DOTATATE/NOC (91.1% vs. 75.3%; P < 10-5).
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Neuroendocrine tumour | ||||
| Efficacy Data | Difference in detection ratio | 11.80% | |||
| Patients Enrolled |
75 patients with histologically confirmed neuroendocrine tumours and routine clinical.
|
||||
| MOA of PDC |
18F-AlF-OC is noninferior and even superior to 68Ga-DOTATATE/NOC PET in NET patients. This validates 18F-AlF-OC as an option for clinical practice somatostatin receptor PET.
|
||||
| Description |
The resulting mean DDR was 15.8%, with a lower margin of the 95% CI (95% CI, 9.6%-22.0%) higher than -15%, which is the prespecified boundary for noninferiority. The mean DDRs for the 68Ga-DOTATATE and 68Ga-DOTANOC subgroups were 11.8% (95% CI, 4.3-19.3) and 27.5% (95% CI, 17.8-37.1), respectively.
|
||||
68Ga-NODAGA-E[c(RGDyK)]2 [Phase 3]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Radiochemical purity | 89%-99% | |||
| In Vivo Model | Nude mice bearing either human glioblastoma (U87MG)/human neuroendocrine (H727) xenograft tumors. | ||||
Debio-0328 [Phase 1]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Clear cell renal cell carcinoma | ||||
| Efficacy Data | SUV mean values | 39 | |||
| Patients Enrolled |
48 years old patient with metastatic ccRCC.
|
||||
| Administration Dosage | 185 MBq (±20%) | ||||
| MOA of PDC |
Carbonic anhydrase IX (CAIX) is a cell-surface glycoprotein involved in acidbase regulation. Aberrant tumor expression contributes to extracellular acidification, promoting tumor progression. The CAIX-encoding gene is overexpressed in more than 90% of ccRCC cases, often because of alterations in the von HippelLindau gene. With physiologic CAIX expression restricted to gastrointestinal epithelia, high tumoral expression presents diagnostic and therapeutic opportunities. Indeed, PET/CT-based tumor visualization with 89Zr-labeled anti-CAIX antibodies ([89Zr]Zr-girentuximab) can aid diagnosis of localized and metastatic ccRCC and enable differentiation of indolent versus benign tumors, which is challenging with conventional imaging. The CAIX-binding cyclic peptide DPI-4452, labeled with diagnostic (68Ga) or therapeutic (177Lu) radioisotopes, provides a novel theranostic pair to target CAIX-expressing tumors. Here, we report the characteristics of diagnostic [68Ga]Ga-DPI-4452 in patients with ccRCC.
Click to Show/Hide
|
||||
| Description |
After [68Ga]Ga-DPI-4452 administration, high tumor-specific uptake was observed as early as 15min and was sustained for all time points assessed. One hour was chosen as the optimal time point on the basis of central reader visual assessment of image quality, visualization of all lesions, and heterogeneity in tumor uptake. Among all lesions, 17 metastases in bone, lymph nodes, lungs, pancreas, and parotid glands were not readily identifiable by conventional imaging. Low renal parenchymal uptake enabled identification of renal tumors. SUVmax 1h after administration across 36 lesions ranged from 6.8 to 211.6 (mean, 64.6 [SD, 54.8]). In patients 1, 2, and 3, the highest SUVmax was 109, 106, and 212, respectively, whereas the highest SUVmean was 39, 62, and 89, respectively.
Click to Show/Hide
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Clear cell renal cell carcinoma | ||||
| Efficacy Data | SUV mean values | 62 | |||
| Patients Enrolled |
51 years old patient with metastatic ccRCC.
|
||||
| Administration Dosage | 185 MBq (±20%) | ||||
| MOA of PDC |
Carbonic anhydrase IX (CAIX) is a cell-surface glycoprotein involved in acidbase regulation. Aberrant tumor expression contributes to extracellular acidification, promoting tumor progression. The CAIX-encoding gene is overexpressed in more than 90% of ccRCC cases, often because of alterations in the von HippelLindau gene. With physiologic CAIX expression restricted to gastrointestinal epithelia, high tumoral expression presents diagnostic and therapeutic opportunities. Indeed, PET/CT-based tumor visualization with 89Zr-labeled anti-CAIX antibodies ([89Zr]Zr-girentuximab) can aid diagnosis of localized and metastatic ccRCC and enable differentiation of indolent versus benign tumors, which is challenging with conventional imaging. The CAIX-binding cyclic peptide DPI-4452, labeled with diagnostic (68Ga) or therapeutic (177Lu) radioisotopes, provides a novel theranostic pair to target CAIX-expressing tumors. Here, we report the characteristics of diagnostic [68Ga]Ga-DPI-4452 in patients with ccRCC.
Click to Show/Hide
|
||||
| Description |
After [68Ga]Ga-DPI-4452 administration, high tumor-specific uptake was observed as early as 15min and was sustained for all time points assessed. One hour was chosen as the optimal time point on the basis of central reader visual assessment of image quality, visualization of all lesions, and heterogeneity in tumor uptake. Among all lesions, 17 metastases in bone, lymph nodes, lungs, pancreas, and parotid glands were not readily identifiable by conventional imaging. Low renal parenchymal uptake enabled identification of renal tumors. SUVmax 1h after administration across 36 lesions ranged from 6.8 to 211.6 (mean, 64.6 [SD, 54.8]). In patients 1, 2, and 3, the highest SUVmax was 109, 106, and 212, respectively, whereas the highest SUVmean was 39, 62, and 89, respectively.
Click to Show/Hide
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Clear cell renal cell carcinoma | ||||
| Efficacy Data | SUV mean values | 89 | |||
| Patients Enrolled |
54 years old patient with metastatic ccRCC.
|
||||
| Administration Dosage | 185 MBq (±20%) | ||||
| MOA of PDC |
Carbonic anhydrase IX (CAIX) is a cell-surface glycoprotein involved in acidbase regulation. Aberrant tumor expression contributes to extracellular acidification, promoting tumor progression. The CAIX-encoding gene is overexpressed in more than 90% of ccRCC cases, often because of alterations in the von HippelLindau gene. With physiologic CAIX expression restricted to gastrointestinal epithelia, high tumoral expression presents diagnostic and therapeutic opportunities. Indeed, PET/CT-based tumor visualization with 89Zr-labeled anti-CAIX antibodies ([89Zr]Zr-girentuximab) can aid diagnosis of localized and metastatic ccRCC and enable differentiation of indolent versus benign tumors, which is challenging with conventional imaging. The CAIX-binding cyclic peptide DPI-4452, labeled with diagnostic (68Ga) or therapeutic (177Lu) radioisotopes, provides a novel theranostic pair to target CAIX-expressing tumors. Here, we report the characteristics of diagnostic [68Ga]Ga-DPI-4452 in patients with ccRCC.
Click to Show/Hide
|
||||
| Description |
After [68Ga]Ga-DPI-4452 administration, high tumor-specific uptake was observed as early as 15min and was sustained for all time points assessed. One hour was chosen as the optimal time point on the basis of central reader visual assessment of image quality, visualization of all lesions, and heterogeneity in tumor uptake. Among all lesions, 17 metastases in bone, lymph nodes, lungs, pancreas, and parotid glands were not readily identifiable by conventional imaging. Low renal parenchymal uptake enabled identification of renal tumors. SUVmax 1h after administration across 36 lesions ranged from 6.8 to 211.6 (mean, 64.6 [SD, 54.8]). In patients 1, 2, and 3, the highest SUVmax was 109, 106, and 212, respectively, whereas the highest SUVmean was 39, 62, and 89, respectively.
Click to Show/Hide
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Clear cell renal cell carcinoma | ||||
| Efficacy Data | SUV max values | 106 | |||
| Patients Enrolled |
51 years old patient with metastatic ccRCC.
|
||||
| Administration Dosage | 185 MBq (±20%) | ||||
| MOA of PDC |
Carbonic anhydrase IX (CAIX) is a cell-surface glycoprotein involved in acidbase regulation. Aberrant tumor expression contributes to extracellular acidification, promoting tumor progression. The CAIX-encoding gene is overexpressed in more than 90% of ccRCC cases, often because of alterations in the von HippelLindau gene. With physiologic CAIX expression restricted to gastrointestinal epithelia, high tumoral expression presents diagnostic and therapeutic opportunities. Indeed, PET/CT-based tumor visualization with 89Zr-labeled anti-CAIX antibodies ([89Zr]Zr-girentuximab) can aid diagnosis of localized and metastatic ccRCC and enable differentiation of indolent versus benign tumors, which is challenging with conventional imaging. The CAIX-binding cyclic peptide DPI-4452, labeled with diagnostic (68Ga) or therapeutic (177Lu) radioisotopes, provides a novel theranostic pair to target CAIX-expressing tumors. Here, we report the characteristics of diagnostic [68Ga]Ga-DPI-4452 in patients with ccRCC.
Click to Show/Hide
|
||||
| Description |
After [68Ga]Ga-DPI-4452 administration, high tumor-specific uptake was observed as early as 15min and was sustained for all time points assessed. One hour was chosen as the optimal time point on the basis of central reader visual assessment of image quality, visualization of all lesions, and heterogeneity in tumor uptake. Among all lesions, 17 metastases in bone, lymph nodes, lungs, pancreas, and parotid glands were not readily identifiable by conventional imaging. Low renal parenchymal uptake enabled identification of renal tumors. SUVmax 1h after administration across 36 lesions ranged from 6.8 to 211.6 (mean, 64.6 [SD, 54.8]). In patients 1, 2, and 3, the highest SUVmax was 109, 106, and 212, respectively, whereas the highest SUVmean was 39, 62, and 89, respectively.
Click to Show/Hide
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Clear cell renal cell carcinoma | ||||
| Efficacy Data | SUV max values | 109 | |||
| Patients Enrolled |
48 years old patient with metastatic ccRCC.
|
||||
| Administration Dosage | 185 MBq (±20%) | ||||
| MOA of PDC |
Carbonic anhydrase IX (CAIX) is a cell-surface glycoprotein involved in acidbase regulation. Aberrant tumor expression contributes to extracellular acidification, promoting tumor progression. The CAIX-encoding gene is overexpressed in more than 90% of ccRCC cases, often because of alterations in the von HippelLindau gene. With physiologic CAIX expression restricted to gastrointestinal epithelia, high tumoral expression presents diagnostic and therapeutic opportunities. Indeed, PET/CT-based tumor visualization with 89Zr-labeled anti-CAIX antibodies ([89Zr]Zr-girentuximab) can aid diagnosis of localized and metastatic ccRCC and enable differentiation of indolent versus benign tumors, which is challenging with conventional imaging. The CAIX-binding cyclic peptide DPI-4452, labeled with diagnostic (68Ga) or therapeutic (177Lu) radioisotopes, provides a novel theranostic pair to target CAIX-expressing tumors. Here, we report the characteristics of diagnostic [68Ga]Ga-DPI-4452 in patients with ccRCC.
Click to Show/Hide
|
||||
| Description |
After [68Ga]Ga-DPI-4452 administration, high tumor-specific uptake was observed as early as 15min and was sustained for all time points assessed. One hour was chosen as the optimal time point on the basis of central reader visual assessment of image quality, visualization of all lesions, and heterogeneity in tumor uptake. Among all lesions, 17 metastases in bone, lymph nodes, lungs, pancreas, and parotid glands were not readily identifiable by conventional imaging. Low renal parenchymal uptake enabled identification of renal tumors. SUVmax 1h after administration across 36 lesions ranged from 6.8 to 211.6 (mean, 64.6 [SD, 54.8]). In patients 1, 2, and 3, the highest SUVmax was 109, 106, and 212, respectively, whereas the highest SUVmean was 39, 62, and 89, respectively.
Click to Show/Hide
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Clear cell renal cell carcinoma | ||||
| Efficacy Data | SUV max values | 212 | |||
| Patients Enrolled |
54 years old patient with metastatic ccRCC.
|
||||
| Administration Dosage | 185 MBq (±20%) | ||||
| MOA of PDC |
Carbonic anhydrase IX (CAIX) is a cell-surface glycoprotein involved in acidbase regulation. Aberrant tumor expression contributes to extracellular acidification, promoting tumor progression. The CAIX-encoding gene is overexpressed in more than 90% of ccRCC cases, often because of alterations in the von HippelLindau gene. With physiologic CAIX expression restricted to gastrointestinal epithelia, high tumoral expression presents diagnostic and therapeutic opportunities. Indeed, PET/CT-based tumor visualization with 89Zr-labeled anti-CAIX antibodies ([89Zr]Zr-girentuximab) can aid diagnosis of localized and metastatic ccRCC and enable differentiation of indolent versus benign tumors, which is challenging with conventional imaging. The CAIX-binding cyclic peptide DPI-4452, labeled with diagnostic (68Ga) or therapeutic (177Lu) radioisotopes, provides a novel theranostic pair to target CAIX-expressing tumors. Here, we report the characteristics of diagnostic [68Ga]Ga-DPI-4452 in patients with ccRCC.
Click to Show/Hide
|
||||
| Description |
After [68Ga]Ga-DPI-4452 administration, high tumor-specific uptake was observed as early as 15min and was sustained for all time points assessed. One hour was chosen as the optimal time point on the basis of central reader visual assessment of image quality, visualization of all lesions, and heterogeneity in tumor uptake. Among all lesions, 17 metastases in bone, lymph nodes, lungs, pancreas, and parotid glands were not readily identifiable by conventional imaging. Low renal parenchymal uptake enabled identification of renal tumors. SUVmax 1h after administration across 36 lesions ranged from 6.8 to 211.6 (mean, 64.6 [SD, 54.8]). In patients 1, 2, and 3, the highest SUVmax was 109, 106, and 212, respectively, whereas the highest SUVmean was 39, 62, and 89, respectively.
Click to Show/Hide
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Clear cell renal cell carcinoma | ||||
| Efficacy Data | Headache | 33.30% | |||
| Patients Enrolled |
3 patients with metastatic ccRCC.
|
||||
| Administration Dosage | 185 MBq (±20%) | ||||
| MOA of PDC |
Carbonic anhydrase IX (CAIX) is a cell-surface glycoprotein involved in acidbase regulation. Aberrant tumor expression contributes to extracellular acidification, promoting tumor progression. The CAIX-encoding gene is overexpressed in more than 90% of ccRCC cases, often because of alterations in the von HippelLindau gene. With physiologic CAIX expression restricted to gastrointestinal epithelia, high tumoral expression presents diagnostic and therapeutic opportunities. Indeed, PET/CT-based tumor visualization with 89Zr-labeled anti-CAIX antibodies ([89Zr]Zr-girentuximab) can aid diagnosis of localized and metastatic ccRCC and enable differentiation of indolent versus benign tumors, which is challenging with conventional imaging. The CAIX-binding cyclic peptide DPI-4452, labeled with diagnostic (68Ga) or therapeutic (177Lu) radioisotopes, provides a novel theranostic pair to target CAIX-expressing tumors. Here, we report the characteristics of diagnostic [68Ga]Ga-DPI-4452 in patients with ccRCC.
Click to Show/Hide
|
||||
| Description |
No clinically significant toxicity was observed; treatment-emergent adverse events (headache and increased blood creatine kinase [1 each; 33.3%]) were not causally related to [68Ga]Ga-DPI-4452. No significant changes in vital signs, laboratory assessments, or electrocardiograms were observed.
|
||||
| Experiment 8 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Clear cell renal cell carcinoma | ||||
| Efficacy Data | Blood creatine kinase increase | 33.30% | |||
| Patients Enrolled |
3 patients with metastatic ccRCC.
|
||||
| Administration Dosage | 185 MBq (±20%) | ||||
| MOA of PDC |
Carbonic anhydrase IX (CAIX) is a cell-surface glycoprotein involved in acidbase regulation. Aberrant tumor expression contributes to extracellular acidification, promoting tumor progression. The CAIX-encoding gene is overexpressed in more than 90% of ccRCC cases, often because of alterations in the von HippelLindau gene. With physiologic CAIX expression restricted to gastrointestinal epithelia, high tumoral expression presents diagnostic and therapeutic opportunities. Indeed, PET/CT-based tumor visualization with 89Zr-labeled anti-CAIX antibodies ([89Zr]Zr-girentuximab) can aid diagnosis of localized and metastatic ccRCC and enable differentiation of indolent versus benign tumors, which is challenging with conventional imaging. The CAIX-binding cyclic peptide DPI-4452, labeled with diagnostic (68Ga) or therapeutic (177Lu) radioisotopes, provides a novel theranostic pair to target CAIX-expressing tumors. Here, we report the characteristics of diagnostic [68Ga]Ga-DPI-4452 in patients with ccRCC.
Click to Show/Hide
|
||||
| Description |
No clinically significant toxicity was observed; treatment-emergent adverse events (headache and increased blood creatine kinase [1 each; 33.3%]) were not causally related to [68Ga]Ga-DPI-4452. No significant changes in vital signs, laboratory assessments, or electrocardiograms were observed.
|
||||
68Ga-DOTANOC [Phase 1]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Neuroendocrine tumour | ||||
| Efficacy Data | Difference in detection ratio | 27.50% | |||
| Patients Enrolled |
75 patients with histologically confirmed neuroendocrine tumours and routine clinical.
|
||||
| MOA of PDC |
18F-AlF-OC is noninferior and even superior to 68Ga-DOTATATE/NOC PET in NET patients. This validates 18F-AlF-OC as an option for clinical practice somatostatin receptor PET.
|
||||
| Description |
The resulting mean DDR was 15.8%, with a lower margin of the 95% CI (95% CI, 9.6%-22.0%) higher than -15%, which is the prespecified boundary for noninferiority. The mean DDRs for the 68Ga-DOTATATE and 68Ga-DOTANOC subgroups were 11.8% (95% CI, 4.3-19.3) and 27.5% (95% CI, 17.8-37.1), respectively.
|
||||
68Ga-PSMA HBED-CC [Investigative]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Stable disease (SD) | 31.60% | |||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
A PSA response was seen in 18 patients (47.4%), stable disease in 12 (31.6%) and progressive disease in 8 (21.1%).
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Response Evaluation Criteria in Solid Tumours (RECIST) | 11.30% | |||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | PSA response | 47.40% | |||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
A PSA response was seen in 18 patients (47.4%), stable disease in 12 (31.6%) and progressive disease in 8 (21.1%).
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Progressive Disease (PD) | 21.10% | |||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
A PSA response was seen in 18 patients (47.4%), stable disease in 12 (31.6%) and progressive disease in 8 (21.1%).
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Mean standardized uptake values (SUVmean) | 26% | |||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Decrease of total tumor volumes (TTV) | 45% | |||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Decrease of median PSA level | 44% | |||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
After RLT the median PSA level decreased by 44%, TTV by 45.1%, SUVmean by 25.8% and RECIST by 11.3%.
|
||||
References
