Linker Information
General Information of This Linker
| Linker ID |
LIN00040
|
|||||
|---|---|---|---|---|---|---|
| Linker Name |
Sulfo-SMCC
|
|||||
| Structure |
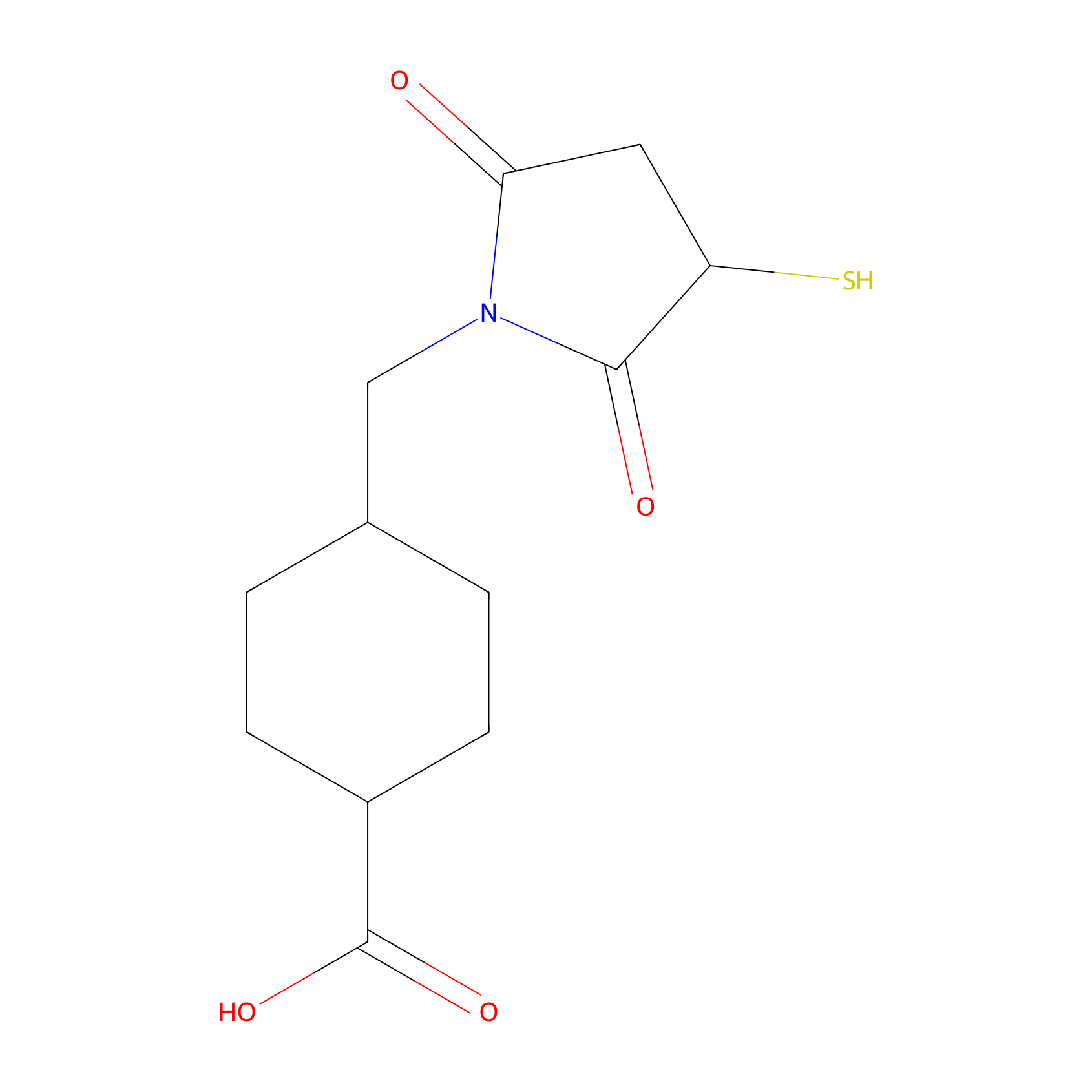
|
|||||
| Formula |
C12H17NO4S
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 271.33 | ||||
| Lipid-water partition coefficient (xlogp) | 0.6 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | |||||
| Rotatable Bond Count (rotbonds) | 3 | |||||
| PubChem CID | ||||||
| Canonical smiles |
C1CC(CCC1CN2C(=O)CC(C2=O)S)C(=O)O
|
|||||
| InChI |
InChI=1S/C12H17NO4S/c14-10-5-9(18)11(15)13(10)6-7-1-3-8(4-2-7)12(16)17/h7-9,18H,1-6H2,(H,16,17)
|
|||||
| InChIKey |
WHRFQGNUPCMDER-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
4-[(2,5-dioxo-3-sulfanylpyrrolidin-1-yl)methyl]cyclohexane-1-carboxylic acid
|
|||||
Each Peptide-drug Conjugate Related to This Linker
Full Information of The Activity Data of The PDC(s) Related to This Linker
cRPQ-SMCC-DM1 [Investigative]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumer volume |
2400 mm3
|
|||
| Administration Time | Given every other day for a total of five times | ||||
| Administration Dosage | 400 µg/kg (calculated by free DM1) | ||||
| Description |
It was demonstrated here, RCCD@NPs and RSSD@NPs exhibited significantly better tumor-growth inhibition compared with that of the free DM1, QCCD@NPs or QSSD@NPs (Figure (Figure4A).4A). RSSD@NPs showed the most suppression on B16 tumor up to 25 days, and resulted in a tumor volume 4 times smaller than the saline group at the end of the experiment. The final tumor volumes in various nano-DDS groups ranked from the greatest to the least: QCCD@NPs, QSSD@NPs>DM1>RCCD@NPs>RSSD@NPs. Almost the same trends were found in terms of tumor weight and tumor size (Figures (Figures4B,4B, C).
Click to Show/Hide
|
||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
67.61 ± 13.67 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
331.13 ± 85.56 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
389.05 ± 75.25 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Melanoma | B16 cell | CVCL_F936 | ||
cRGD-SMCC-DM1 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Uric acid levels |
185
|
|||
| Administration Time | 21d | ||||
| Description |
Remarkably, leucopenia and myelosuppression did not occur during the use of PDC. In addition, the values of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP), as the biomarkers of hepatic function, were all in the normal range in comparison to the saline group (Figure 5F-H). Meanwhile, normal renal function was also identified after treatment by monitoring CREA, UA, and BUN (Figure 5I-K).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumer volume |
1000 mm3
|
|||
| Administration Time | 21d | ||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumer volume |
2100 mm3
|
|||
| Administration Time | Given every other day for a total of five times | ||||
| Administration Dosage | 400 µg/kg (calculated by free DM1) | ||||
| Description |
It was demonstrated here, RCCD@NPs and RSSD@NPs exhibited significantly better tumor-growth inhibition compared with that of the free DM1, QCCD@NPs or QSSD@NPs (Figure (Figure4A).4A). RSSD@NPs showed the most suppression on B16 tumor up to 25 days, and resulted in a tumor volume 4 times smaller than the saline group at the end of the experiment. The final tumor volumes in various nano-DDS groups ranked from the greatest to the least: QCCD@NPs, QSSD@NPs>DM1>RCCD@NPs>RSSD@NPs. Almost the same trends were found in terms of tumor weight and tumor size (Figures (Figures4B,4B, C).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing C57BL/6 mice model. | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | BUN level |
7
|
|||
| Administration Time | 21d | ||||
| Description |
Remarkably, leucopenia and myelosuppression did not occur during the use of PDC. In addition, the values of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP), as the biomarkers of hepatic function, were all in the normal range in comparison to the saline group (Figure 5F-H). Meanwhile, normal renal function was also identified after treatment by monitoring CREA, UA, and BUN (Figure 5I-K).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | AST level |
175
|
|||
| Administration Time | 21d | ||||
| Description |
Remarkably, leucopenia and myelosuppression did not occur during the use of PDC. In addition, the values of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP), as the biomarkers of hepatic function, were all in the normal range in comparison to the saline group (Figure 5F-H). Meanwhile, normal renal function was also identified after treatment by monitoring CREA, UA, and BUN (Figure 5I-K).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | ALT level |
60
|
|||
| Administration Time | 21d | ||||
| Description |
Remarkably, leucopenia and myelosuppression did not occur during the use of PDC. In addition, the values of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP), as the biomarkers of hepatic function, were all in the normal range in comparison to the saline group (Figure 5F-H). Meanwhile, normal renal function was also identified after treatment by monitoring CREA, UA, and BUN (Figure 5I-K).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | ALP level |
80
|
|||
| Administration Time | 21d | ||||
| Description |
Remarkably, leucopenia and myelosuppression did not occur during the use of PDC. In addition, the values of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP), as the biomarkers of hepatic function, were all in the normal range in comparison to the saline group (Figure 5F-H). Meanwhile, normal renal function was also identified after treatment by monitoring CREA, UA, and BUN (Figure 5I-K).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
33.84 ± 6.30 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
68.5 nM
|
|||
| Description |
After the MDA-MB-231 cell line was confirmed to be integrin v3-positive by immunofluorescence assay, it was demonstrated that cRGD-SMCC-DM1 had an IC50 value of 68.5 nM to MDA-MB-231 cells.
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
102.33 ± 38.92 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Melanoma | B16 cell | CVCL_F936 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
407.38 ± 54.05 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
Peptide 18-4-doxorubicin [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.3 µM
|
|||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that the cytotoxicity of both conjugates 1 and 2 (IC50 = 1.3 and 2.2 uM, respectively), as well as the free Dox (IC50 = 1.5 uM) on MDA-MB-231 breast cancer cell line, were in the low micromolar range (Figure 4). For the breast cancer cell line MDA-MB-468, the free Dox (IC50 = 0.35 uM) was slightly more toxic compared to conjugates 1 (4.7 uM) and 2 (1.2 uM). For the non-cancerous cell line MCF 10A, the free Dox was highly toxic (IC50 = 0.24 uM) whereas conjugates 1 and 2 displayed much-reduced toxicity (IC50 = 38.6 and 15.1 uM, respectively).
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.3 ± 0.2µM
|
|||
| MOA of PDC |
We engineered peptides that bind to cell-surface K1 and are internalized by breast cancer cells via cell-surface K1 receptor-mediated endocytosis. Peptide 18-4 (WxEAAYQrFL), with two D-amino acids, is a second-generation breast cancer cell-targeting peptide that is proteolytically stable (100% intact up to 24 h when incubated with human serum or liver homogenate from mice) and has shown high specific uptake by breast cancer cells and minimal/no binding to non-cancerous cells. Affinity purification of breast cancer cell lysates using the immobilized peptide, followed by liquid chromatography-tandem mass spectrometry and proteomics were used to identify K1 as the novel target for peptide 18-4 in cancer cells. Further, we showed that the uptake of the peptide by the cancer cells is dependent on K1 expression.
Click to Show/Hide
|
||||
| Description |
Several PDCs have been prepared with peptide 18-4 and doxorubicin (Dox) using different linker chemistries such as ester, amide, succinimidyl thioether, or acyl hydrazone. In vitro results showed that these PDCs were highly specific toward breast cancer cells. PDCs displayed similar toxicity as free Dox toward the breast cancer cells and several-fold (7-40 times) less toxicity toward the non-cancerous cells such as MCF-10A and human umbilical vein endothelial cells (HUVECs). A peptide 18-4-Dox conjugate with amide/succinimidyl thioether linkage showed high selective toxicity toward triple negative breast cancer (TNBC) cell lines, i.e., MDA-MB-231 cells (IC50 1.3 ± 0.2 uM) and MDA-MB-468 cells (IC50 4.7 ± 0.3 uM) compared to the normal breast MCF10A cells (IC50 38.6 ± 1.1 uM). The linkage between the drug and the peptide was stable as the degradation half-life of peptide 18-4-Dox conjugate in the presence of human serum was found to be ˜18 h. Herein, we describe the first in vivo evidence for improved efficacy of this PDC targeting K1 receptor in an orthotopic TNBC mouse model. We also show a higher accumulation of PDC in TNBC tumors in mice, in accord with K1 overexpression in tumor over non-tumor tissues in MDA-MB-231 xenografted mice.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
4.7 µM
|
|||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that the cytotoxicity of both conjugates 1 and 2 (IC50 = 1.3 and 2.2 uM, respectively), as well as the free Dox (IC50 = 1.5 uM) on MDA-MB-231 breast cancer cell line, were in the low micromolar range (Figure 4). For the breast cancer cell line MDA-MB-468, the free Dox (IC50 = 0.35 uM) was slightly more toxic compared to conjugates 1 (4.7 uM) and 2 (1.2 uM). For the non-cancerous cell line MCF 10A, the free Dox was highly toxic (IC50 = 0.24 uM) whereas conjugates 1 and 2 displayed much-reduced toxicity (IC50 = 38.6 and 15.1 uM, respectively).
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-468 cell | CVCL_0419 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
4.7 ± 0.3µM
|
|||
| MOA of PDC |
We engineered peptides that bind to cell-surface K1 and are internalized by breast cancer cells via cell-surface K1 receptor-mediated endocytosis. Peptide 18-4 (WxEAAYQrFL), with two D-amino acids, is a second-generation breast cancer cell-targeting peptide that is proteolytically stable (100% intact up to 24 h when incubated with human serum or liver homogenate from mice) and has shown high specific uptake by breast cancer cells and minimal/no binding to non-cancerous cells. Affinity purification of breast cancer cell lysates using the immobilized peptide, followed by liquid chromatography-tandem mass spectrometry and proteomics were used to identify K1 as the novel target for peptide 18-4 in cancer cells. Further, we showed that the uptake of the peptide by the cancer cells is dependent on K1 expression.
Click to Show/Hide
|
||||
| Description |
Several PDCs have been prepared with peptide 18-4 and doxorubicin (Dox) using different linker chemistries such as ester, amide, succinimidyl thioether, or acyl hydrazone. In vitro results showed that these PDCs were highly specific toward breast cancer cells. PDCs displayed similar toxicity as free Dox toward the breast cancer cells and several-fold (7-40 times) less toxicity toward the non-cancerous cells such as MCF-10A and human umbilical vein endothelial cells (HUVECs). A peptide 18-4-Dox conjugate with amide/succinimidyl thioether linkage showed high selective toxicity toward triple negative breast cancer (TNBC) cell lines, i.e., MDA-MB-231 cells (IC50 1.3 ± 0.2 uM) and MDA-MB-468 cells (IC50 4.7 ± 0.3 uM) compared to the normal breast MCF10A cells (IC50 38.6 ± 1.1 uM). The linkage between the drug and the peptide was stable as the degradation half-life of peptide 18-4-Dox conjugate in the presence of human serum was found to be ˜18 h. Herein, we describe the first in vivo evidence for improved efficacy of this PDC targeting K1 receptor in an orthotopic TNBC mouse model. We also show a higher accumulation of PDC in TNBC tumors in mice, in accord with K1 overexpression in tumor over non-tumor tissues in MDA-MB-231 xenografted mice.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-468 cell | CVCL_0419 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
38.6 µM
|
|||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that the cytotoxicity of both conjugates 1 and 2 (IC50 = 1.3 and 2.2 uM, respectively), as well as the free Dox (IC50 = 1.5 uM) on MDA-MB-231 breast cancer cell line, were in the low micromolar range (Figure 4). For the breast cancer cell line MDA-MB-468, the free Dox (IC50 = 0.35 uM) was slightly more toxic compared to conjugates 1 (4.7 uM) and 2 (1.2 uM). For the non-cancerous cell line MCF 10A, the free Dox was highly toxic (IC50 = 0.24 uM) whereas conjugates 1 and 2 displayed much-reduced toxicity (IC50 = 38.6 and 15.1 uM, respectively).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MCF-10A cell | CVCL_0598 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
38.6 ± 1.1µM
|
|||
| MOA of PDC |
We engineered peptides that bind to cell-surface K1 and are internalized by breast cancer cells via cell-surface K1 receptor-mediated endocytosis. Peptide 18-4 (WxEAAYQrFL), with two D-amino acids, is a second-generation breast cancer cell-targeting peptide that is proteolytically stable (100% intact up to 24 h when incubated with human serum or liver homogenate from mice) and has shown high specific uptake by breast cancer cells and minimal/no binding to non-cancerous cells. Affinity purification of breast cancer cell lysates using the immobilized peptide, followed by liquid chromatography-tandem mass spectrometry and proteomics were used to identify K1 as the novel target for peptide 18-4 in cancer cells. Further, we showed that the uptake of the peptide by the cancer cells is dependent on K1 expression.
Click to Show/Hide
|
||||
| Description |
Several PDCs have been prepared with peptide 18-4 and doxorubicin (Dox) using different linker chemistries such as ester, amide, succinimidyl thioether, or acyl hydrazone. In vitro results showed that these PDCs were highly specific toward breast cancer cells. PDCs displayed similar toxicity as free Dox toward the breast cancer cells and several-fold (7-40 times) less toxicity toward the non-cancerous cells such as MCF-10A and human umbilical vein endothelial cells (HUVECs). A peptide 18-4-Dox conjugate with amide/succinimidyl thioether linkage showed high selective toxicity toward triple negative breast cancer (TNBC) cell lines, i.e., MDA-MB-231 cells (IC50 1.3 ± 0.2 uM) and MDA-MB-468 cells (IC50 4.7 ± 0.3 uM) compared to the normal breast MCF10A cells (IC50 38.6 ± 1.1 uM). The linkage between the drug and the peptide was stable as the degradation half-life of peptide 18-4-Dox conjugate in the presence of human serum was found to be ˜18 h. Herein, we describe the first in vivo evidence for improved efficacy of this PDC targeting K1 receptor in an orthotopic TNBC mouse model. We also show a higher accumulation of PDC in TNBC tumors in mice, in accord with K1 overexpression in tumor over non-tumor tissues in MDA-MB-231 xenografted mice.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MCF-10A cell | CVCL_0598 | ||
rL-A9-DOX [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
7%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 15 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
13%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 15 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
26%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 7.5 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
33%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 7.5 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
49%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 3.75 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
67%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 1.87 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
87%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 3.75 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
91%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.94 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
94%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 1.87 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
95%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.47 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
96%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.94 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
97%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.47 µM | ||||
| Evaluation Method | Flow cytometry assay | ||||
| MOA of PDC |
The peptide A9 has been reported in the literature to exhibit high affinity and specificity towards the HER2 receptor. In our previous report, we observed the retro variant of A9 peptide, rL-A9, to be a promising molecule for targeting HER2-expressing breast cancer cells. The present study, thus aimed at designing and synthesis of a peptide-drug conjugate by linking the rL-A9 peptide with DOX. A linker, N-succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) was utilized for conjugation of DOX at one end and the peptide at the other end. The N-hydroxysuccinimidyl ester group of SMCC was conjugated with the amine (-NH2) group of DOX resulting in the formation of amide bond. The thiol (-SH) functionality was introduced by coupling cysteine amino acid at the N-terminus of the rL-A9 peptide for covalent linkage with the maleimide group of SMCC. Successful synthesis of the conjugate was confirmed by MALDI-TOF mass spectrometry. Cytotoxicity, cellular uptake and internalization of the peptide, drug and peptide-drug conjugate were assessed in SKOV3 cells using flow cytometry and confocal fluorescence microscopy.
Click to Show/Hide
|
||||
| Description |
Analysis of dot-plots suggests that the rL-A9 peptide alone does not exert any significant cytotoxic effects on either HER2-positive, SKOV3 cells or HER2-negative, MDA-MB-231 cells at any of the investigated concentrations. In contrast, incubation of drug DOX with either of the cells resulted in enhanced cell death with a negligible viable population even at lower concentrations. However, it was observed that the peptide-drug conjugate, rL-A9-DOX had a concentration-dependent impact on cell death (SKOV3 cells). Notably, nearly half of the cell population died at 3.75 uM. At a concentration of 15 uM, there were only 5% viable SKOV3 cells. The higher percent of the viable cell population was observed at corresponding concentrations in the case of incubation of rL-A9-DOX with HER2-negative, MDA-MB-231 cells. Comparative data of viability in two different cell lines is presented in Figure 6 for the peptide-drug conjugate, rL-A9-DOX.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
pHLIP-M-DOX [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
97%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTS assay | ||||
| Description |
pHLIP-M-DOX had a negligible effect on HeLa cell proliferation when incubated on cells at either pH at pHLIP concentrations as high as 10 μM. This is consistent with our imaging data that showed no release of DOX into the cell interior at either pH.
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
97%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 10 µM | ||||
| Evaluation Method | MTS assay | ||||
| Description |
pHLIP-M-DOX had a negligible effect on HeLa cell proliferation when incubated on cells at either pH at pHLIP concentrations as high as 10 μM. This is consistent with our imaging data that showed no release of DOX into the cell interior at either pH.
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
98%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 2.5 µM | ||||
| Evaluation Method | MTS assay | ||||
| Description |
pHLIP-M-DOX had a negligible effect on HeLa cell proliferation when incubated on cells at either pH at pHLIP concentrations as high as 10 μM. This is consistent with our imaging data that showed no release of DOX into the cell interior at either pH.
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
99%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTS assay | ||||
| Description |
pHLIP-M-DOX had a negligible effect on HeLa cell proliferation when incubated on cells at either pH at pHLIP concentrations as high as 10 μM. This is consistent with our imaging data that showed no release of DOX into the cell interior at either pH.
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [6] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
100%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 2.5 µM | ||||
| Evaluation Method | MTS assay | ||||
| Description |
pHLIP-M-DOX had a negligible effect on HeLa cell proliferation when incubated on cells at either pH at pHLIP concentrations as high as 10 μM. This is consistent with our imaging data that showed no release of DOX into the cell interior at either pH.
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
References
