Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00303
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
Peptide 18-4-doxorubicin
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 2 Indication(s)
|
|||||
| Structure |
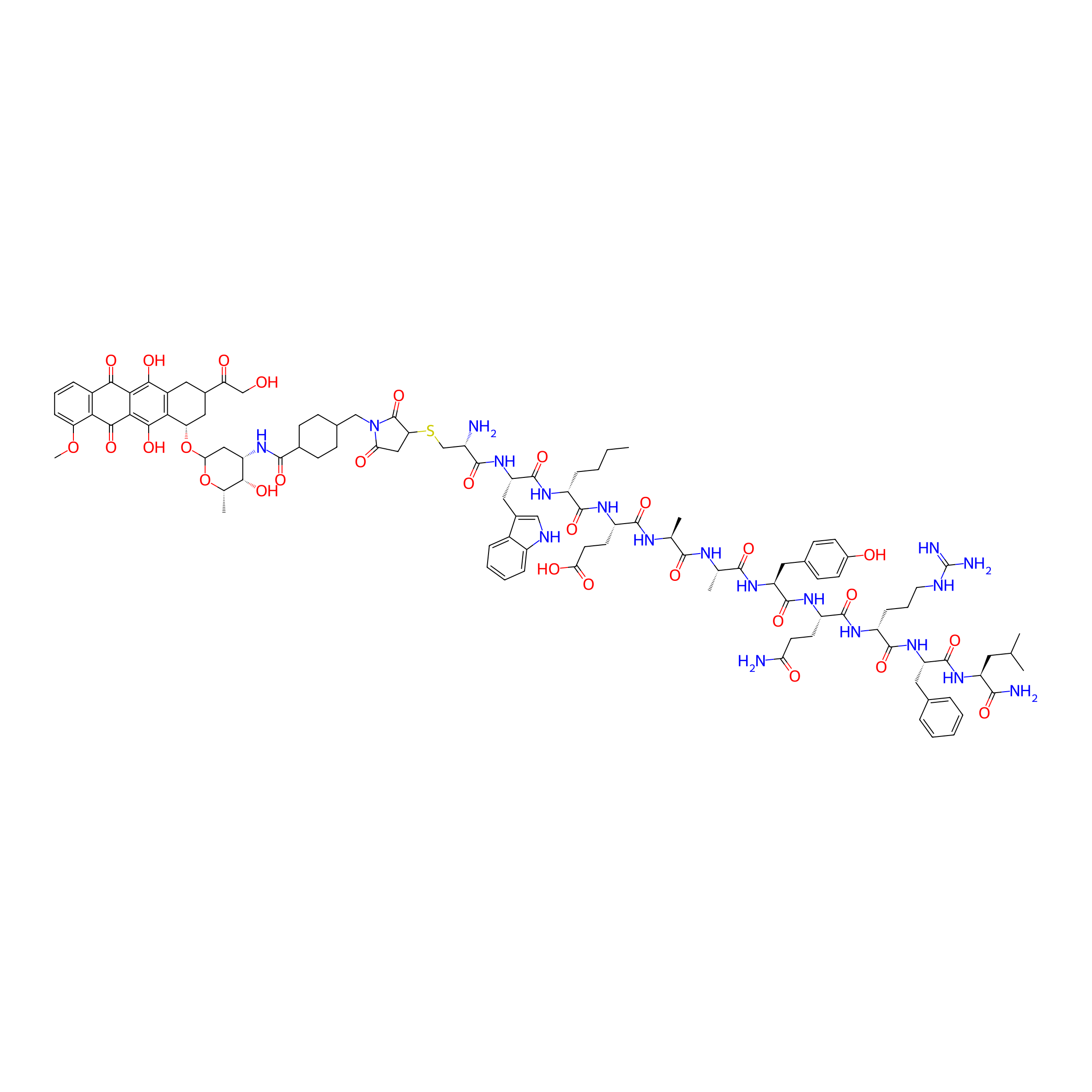
|
|||||
| Peptide Name |
Peptide 18-4
|
Peptide Info | ||||
| Receptor Name |
Keratin, type II cytoskeletal 1 (KRT1)
|
Receptor Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
Sulfo-SMCC
|
Linker Info | ||||
| Peptide Modified Type |
Amino acid modifications
|
|||||
| Modified Segment |
Use D-amino acids instead of L-amino acids
|
|||||
| Formula |
C105H137N19O28S
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 2145.423 | ||||
| Lipid-water partition coefficient (xlogp) | -0.04373 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 24 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 30 | |||||
| Rotatable Bond Count (rotbonds) | 54 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.3 µM
|
|||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that the cytotoxicity of both conjugates 1 and 2 (IC50 = 1.3 and 2.2 uM, respectively), as well as the free Dox (IC50 = 1.5 uM) on MDA-MB-231 breast cancer cell line, were in the low micromolar range (Figure 4). For the breast cancer cell line MDA-MB-468, the free Dox (IC50 = 0.35 uM) was slightly more toxic compared to conjugates 1 (4.7 uM) and 2 (1.2 uM). For the non-cancerous cell line MCF 10A, the free Dox was highly toxic (IC50 = 0.24 uM) whereas conjugates 1 and 2 displayed much-reduced toxicity (IC50 = 38.6 and 15.1 uM, respectively).
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.3 ± 0.2µM
|
|||
| MOA of PDC |
We engineered peptides that bind to cell-surface K1 and are internalized by breast cancer cells via cell-surface K1 receptor-mediated endocytosis. Peptide 18-4 (WxEAAYQrFL), with two D-amino acids, is a second-generation breast cancer cell-targeting peptide that is proteolytically stable (100% intact up to 24 h when incubated with human serum or liver homogenate from mice) and has shown high specific uptake by breast cancer cells and minimal/no binding to non-cancerous cells. Affinity purification of breast cancer cell lysates using the immobilized peptide, followed by liquid chromatography-tandem mass spectrometry and proteomics were used to identify K1 as the novel target for peptide 18-4 in cancer cells. Further, we showed that the uptake of the peptide by the cancer cells is dependent on K1 expression.
Click to Show/Hide
|
||||
| Description |
Several PDCs have been prepared with peptide 18-4 and doxorubicin (Dox) using different linker chemistries such as ester, amide, succinimidyl thioether, or acyl hydrazone. In vitro results showed that these PDCs were highly specific toward breast cancer cells. PDCs displayed similar toxicity as free Dox toward the breast cancer cells and several-fold (7-40 times) less toxicity toward the non-cancerous cells such as MCF-10A and human umbilical vein endothelial cells (HUVECs). A peptide 18-4-Dox conjugate with amide/succinimidyl thioether linkage showed high selective toxicity toward triple negative breast cancer (TNBC) cell lines, i.e., MDA-MB-231 cells (IC50 1.3 ± 0.2 uM) and MDA-MB-468 cells (IC50 4.7 ± 0.3 uM) compared to the normal breast MCF10A cells (IC50 38.6 ± 1.1 uM). The linkage between the drug and the peptide was stable as the degradation half-life of peptide 18-4-Dox conjugate in the presence of human serum was found to be ˜18 h. Herein, we describe the first in vivo evidence for improved efficacy of this PDC targeting K1 receptor in an orthotopic TNBC mouse model. We also show a higher accumulation of PDC in TNBC tumors in mice, in accord with K1 overexpression in tumor over non-tumor tissues in MDA-MB-231 xenografted mice.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
4.7 µM
|
|||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that the cytotoxicity of both conjugates 1 and 2 (IC50 = 1.3 and 2.2 uM, respectively), as well as the free Dox (IC50 = 1.5 uM) on MDA-MB-231 breast cancer cell line, were in the low micromolar range (Figure 4). For the breast cancer cell line MDA-MB-468, the free Dox (IC50 = 0.35 uM) was slightly more toxic compared to conjugates 1 (4.7 uM) and 2 (1.2 uM). For the non-cancerous cell line MCF 10A, the free Dox was highly toxic (IC50 = 0.24 uM) whereas conjugates 1 and 2 displayed much-reduced toxicity (IC50 = 38.6 and 15.1 uM, respectively).
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-468 cell | CVCL_0419 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
4.7 ± 0.3µM
|
|||
| MOA of PDC |
We engineered peptides that bind to cell-surface K1 and are internalized by breast cancer cells via cell-surface K1 receptor-mediated endocytosis. Peptide 18-4 (WxEAAYQrFL), with two D-amino acids, is a second-generation breast cancer cell-targeting peptide that is proteolytically stable (100% intact up to 24 h when incubated with human serum or liver homogenate from mice) and has shown high specific uptake by breast cancer cells and minimal/no binding to non-cancerous cells. Affinity purification of breast cancer cell lysates using the immobilized peptide, followed by liquid chromatography-tandem mass spectrometry and proteomics were used to identify K1 as the novel target for peptide 18-4 in cancer cells. Further, we showed that the uptake of the peptide by the cancer cells is dependent on K1 expression.
Click to Show/Hide
|
||||
| Description |
Several PDCs have been prepared with peptide 18-4 and doxorubicin (Dox) using different linker chemistries such as ester, amide, succinimidyl thioether, or acyl hydrazone. In vitro results showed that these PDCs were highly specific toward breast cancer cells. PDCs displayed similar toxicity as free Dox toward the breast cancer cells and several-fold (7-40 times) less toxicity toward the non-cancerous cells such as MCF-10A and human umbilical vein endothelial cells (HUVECs). A peptide 18-4-Dox conjugate with amide/succinimidyl thioether linkage showed high selective toxicity toward triple negative breast cancer (TNBC) cell lines, i.e., MDA-MB-231 cells (IC50 1.3 ± 0.2 uM) and MDA-MB-468 cells (IC50 4.7 ± 0.3 uM) compared to the normal breast MCF10A cells (IC50 38.6 ± 1.1 uM). The linkage between the drug and the peptide was stable as the degradation half-life of peptide 18-4-Dox conjugate in the presence of human serum was found to be ˜18 h. Herein, we describe the first in vivo evidence for improved efficacy of this PDC targeting K1 receptor in an orthotopic TNBC mouse model. We also show a higher accumulation of PDC in TNBC tumors in mice, in accord with K1 overexpression in tumor over non-tumor tissues in MDA-MB-231 xenografted mice.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-468 cell | CVCL_0419 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
38.6 µM
|
|||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that the cytotoxicity of both conjugates 1 and 2 (IC50 = 1.3 and 2.2 uM, respectively), as well as the free Dox (IC50 = 1.5 uM) on MDA-MB-231 breast cancer cell line, were in the low micromolar range (Figure 4). For the breast cancer cell line MDA-MB-468, the free Dox (IC50 = 0.35 uM) was slightly more toxic compared to conjugates 1 (4.7 uM) and 2 (1.2 uM). For the non-cancerous cell line MCF 10A, the free Dox was highly toxic (IC50 = 0.24 uM) whereas conjugates 1 and 2 displayed much-reduced toxicity (IC50 = 38.6 and 15.1 uM, respectively).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MCF-10A cell | CVCL_0598 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
38.6 ± 1.1µM
|
|||
| MOA of PDC |
We engineered peptides that bind to cell-surface K1 and are internalized by breast cancer cells via cell-surface K1 receptor-mediated endocytosis. Peptide 18-4 (WxEAAYQrFL), with two D-amino acids, is a second-generation breast cancer cell-targeting peptide that is proteolytically stable (100% intact up to 24 h when incubated with human serum or liver homogenate from mice) and has shown high specific uptake by breast cancer cells and minimal/no binding to non-cancerous cells. Affinity purification of breast cancer cell lysates using the immobilized peptide, followed by liquid chromatography-tandem mass spectrometry and proteomics were used to identify K1 as the novel target for peptide 18-4 in cancer cells. Further, we showed that the uptake of the peptide by the cancer cells is dependent on K1 expression.
Click to Show/Hide
|
||||
| Description |
Several PDCs have been prepared with peptide 18-4 and doxorubicin (Dox) using different linker chemistries such as ester, amide, succinimidyl thioether, or acyl hydrazone. In vitro results showed that these PDCs were highly specific toward breast cancer cells. PDCs displayed similar toxicity as free Dox toward the breast cancer cells and several-fold (7-40 times) less toxicity toward the non-cancerous cells such as MCF-10A and human umbilical vein endothelial cells (HUVECs). A peptide 18-4-Dox conjugate with amide/succinimidyl thioether linkage showed high selective toxicity toward triple negative breast cancer (TNBC) cell lines, i.e., MDA-MB-231 cells (IC50 1.3 ± 0.2 uM) and MDA-MB-468 cells (IC50 4.7 ± 0.3 uM) compared to the normal breast MCF10A cells (IC50 38.6 ± 1.1 uM). The linkage between the drug and the peptide was stable as the degradation half-life of peptide 18-4-Dox conjugate in the presence of human serum was found to be ˜18 h. Herein, we describe the first in vivo evidence for improved efficacy of this PDC targeting K1 receptor in an orthotopic TNBC mouse model. We also show a higher accumulation of PDC in TNBC tumors in mice, in accord with K1 overexpression in tumor over non-tumor tissues in MDA-MB-231 xenografted mice.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MCF-10A cell | CVCL_0598 | ||
References
