Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00008
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
[177Lu]Lu-P17-081
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
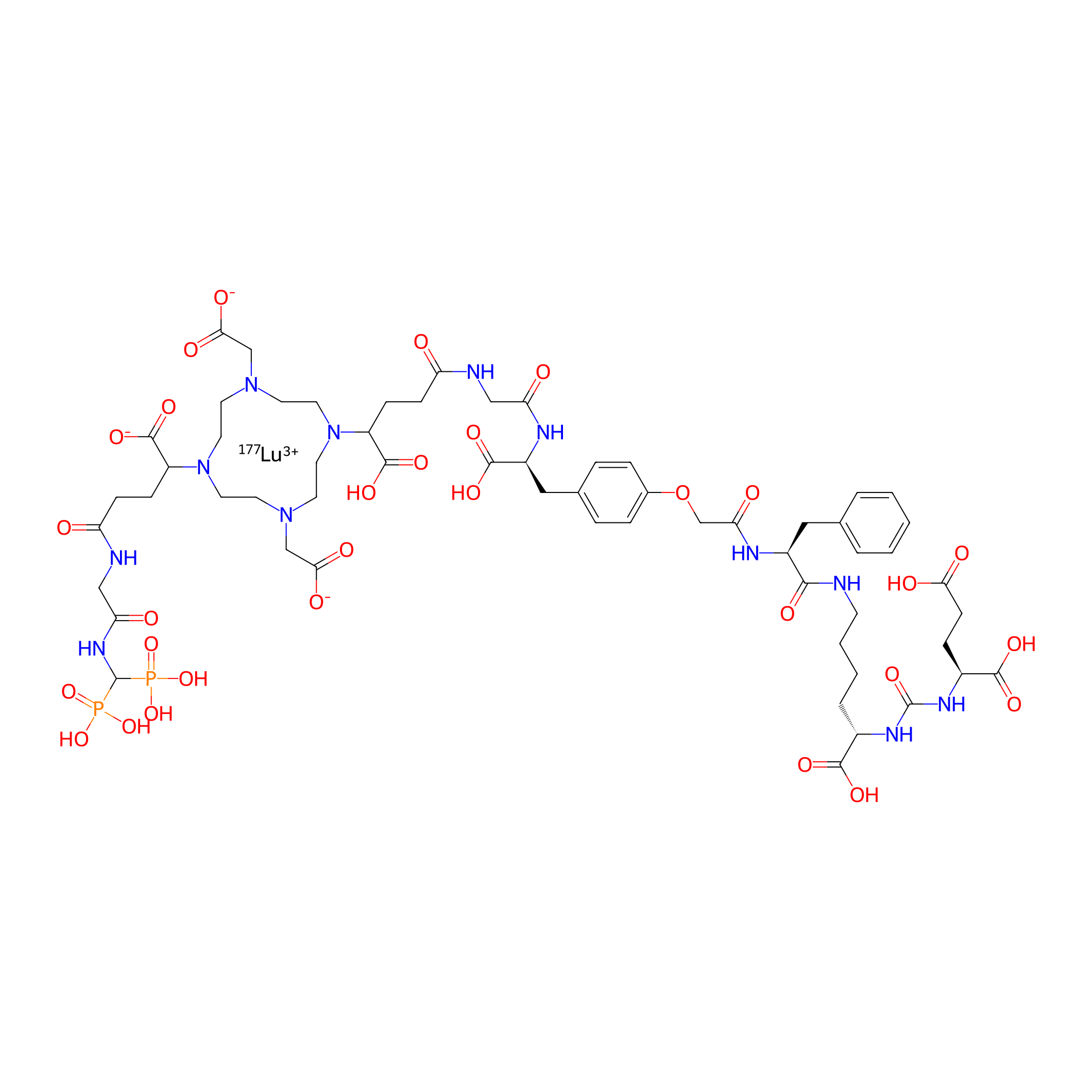
|
|||||
| Peptide Name |
PSMA-617
|
Peptide Info | ||||
| Receptor Name |
Glutamate carboxypeptidase 2 (FOLH1)
|
Receptor Info | ||||
| Drug Name |
Lutetium-177
|
Drug Info | ||||
| Therapeutic Target |
Human Deoxyribonucleic acid (hDNA)
|
Target Info | ||||
| Linker Name |
rac-(R)-2-(2-aminoacetamido)-3-(4-(2-(((R)-1-carboxy-2-phenylethyl)amino)-2-oxoethoxy)phenyl)propanoic acid
|
Linker Info | ||||
| Peptide Modified Type |
The modification of binding with chemical molecules
|
|||||
| Modified Segment |
Urea
|
|||||
| Formula |
C59H83LuN12O30P2
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 1679.258758 | ||||
| Lipid-water partition coefficient (xlogp) | -8.8972 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 17 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 25 | |||||
| Rotatable Bond Count (rotbonds) | 44 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell uptake rate |
3.50%
|
|||
| Administration Time | 5 min | ||||
| Administration Dosage | 37 kBq | ||||
| Evaluation Method | Gamma counter assay | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is an excellent target for imaging and radionuclide therapy of prostate cancer. Recently, [177Lu]Lu-PSMA-617 (Pluvicto) was approved by the FDA for radionuclide therapy. To develop hetero-bivalent agents targeting both PSMA and bone metastasis, [177Lu]Lu-P17-079 ([177Lu]Lu-1) and [177Lu]Lu-P17-081 ([177Lu]Lu-2) were prepared. In vivo biodistribution studies of [177Lu]Lu-PSMA-617, [177Lu]Lu-1, and [177Lu]Lu-2 in mice bearing PC3-PIP (PSMA positive) tumor showed high uptake in PSMA-positive tumor (14.5, 14.7, and 11.3% ID/g at 1 h, respectively) and distinctively different bone uptakes (0.52, 6.52, and 5.82% ID/g at 1 h, respectively). PET imaging using [68Ga]Ga-P17-079 ([68Ga]Ga-1) in the same mouse model displayed excellent images confirming the expected dual-targeting to PSMA-positive tumor and bone. Results suggest that [177Lu]Lu-P17-079 ([177Lu]Lu-1) is a promising candidate for further development as a hetero-bivalent radionuclide therapy agent targeting both PSMA expression and bone metastases for the treatment of prostate cancer.
Click to Show/Hide
|
||||
| Description |
As expected, the PSMA-targeting hetero-bivalent agents, [177Lu]Lu-1 and [177Lu]Lu-2, displayed excellent cell uptake in PC3-PIP PSMA-positive cells. Furthermore, [177Lu]Lu-1 displayed a higher uptake (about 2 times higher, p < 0.05) as that of [177Lu]Lu-2. The higher uptake in PSMA-positive cells suggested that [177Lu]Lu-1 may more readily penetrate the cell membrane and may be more localized in the PSMA-positive tumor cells. The uptake was specifically inhibited by blocking study (in the presence of 1 uM cold PSMA-617). Both agents did not show any significant cell uptakes in PSMA-negative cells (PC3 cells). Cell uptake kinetics of [177Lu]Lu-1 and [177Lu]Lu-2 showed very rapid uptakes in PC3-PIP PSMA-positive cells (5, 15, 30, 60, and 120 min, respectively). As a positive control, [177Lu]Lu-PSMA-617 also showed excellent cell uptake under a similar condition. It is noted that [177Lu]Lu-2 showed a very similar uptake kinetics as that of [177Lu]Lu-PSMA-617, while [177Lu]Lu-1 displayed a significantly higher and faster uptake. Apparently, a small change in the chemical structure of the location of the bisphosphonate group between [177Lu]Lu-1 and [177Lu]Lu-2 may have contributed to this observation. No obvious reason could be contributed to the superior cell uptake of [177Lu]Lu-1. This distinctive difference in in vitro cell uptake may also be responsible for variations of PSMA-positive tumor uptakes in vivo.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC3-PIP PSMA-positive cell | CVCL_0035 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell uptake rate |
5.50%
|
|||
| Administration Time | 15 min | ||||
| Administration Dosage | 37 kBq | ||||
| Evaluation Method | Gamma counter assay | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is an excellent target for imaging and radionuclide therapy of prostate cancer. Recently, [177Lu]Lu-PSMA-617 (Pluvicto) was approved by the FDA for radionuclide therapy. To develop hetero-bivalent agents targeting both PSMA and bone metastasis, [177Lu]Lu-P17-079 ([177Lu]Lu-1) and [177Lu]Lu-P17-081 ([177Lu]Lu-2) were prepared. In vivo biodistribution studies of [177Lu]Lu-PSMA-617, [177Lu]Lu-1, and [177Lu]Lu-2 in mice bearing PC3-PIP (PSMA positive) tumor showed high uptake in PSMA-positive tumor (14.5, 14.7, and 11.3% ID/g at 1 h, respectively) and distinctively different bone uptakes (0.52, 6.52, and 5.82% ID/g at 1 h, respectively). PET imaging using [68Ga]Ga-P17-079 ([68Ga]Ga-1) in the same mouse model displayed excellent images confirming the expected dual-targeting to PSMA-positive tumor and bone. Results suggest that [177Lu]Lu-P17-079 ([177Lu]Lu-1) is a promising candidate for further development as a hetero-bivalent radionuclide therapy agent targeting both PSMA expression and bone metastases for the treatment of prostate cancer.
Click to Show/Hide
|
||||
| Description |
As expected, the PSMA-targeting hetero-bivalent agents, [177Lu]Lu-1 and [177Lu]Lu-2, displayed excellent cell uptake in PC3-PIP PSMA-positive cells. Furthermore, [177Lu]Lu-1 displayed a higher uptake (about 2 times higher, p < 0.05) as that of [177Lu]Lu-2. The higher uptake in PSMA-positive cells suggested that [177Lu]Lu-1 may more readily penetrate the cell membrane and may be more localized in the PSMA-positive tumor cells. The uptake was specifically inhibited by blocking study (in the presence of 1 uM cold PSMA-617). Both agents did not show any significant cell uptakes in PSMA-negative cells (PC3 cells). Cell uptake kinetics of [177Lu]Lu-1 and [177Lu]Lu-2 showed very rapid uptakes in PC3-PIP PSMA-positive cells (5, 15, 30, 60, and 120 min, respectively). As a positive control, [177Lu]Lu-PSMA-617 also showed excellent cell uptake under a similar condition. It is noted that [177Lu]Lu-2 showed a very similar uptake kinetics as that of [177Lu]Lu-PSMA-617, while [177Lu]Lu-1 displayed a significantly higher and faster uptake. Apparently, a small change in the chemical structure of the location of the bisphosphonate group between [177Lu]Lu-1 and [177Lu]Lu-2 may have contributed to this observation. No obvious reason could be contributed to the superior cell uptake of [177Lu]Lu-1. This distinctive difference in in vitro cell uptake may also be responsible for variations of PSMA-positive tumor uptakes in vivo.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC3-PIP PSMA-positive cell | CVCL_0035 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell uptake rate |
7.50%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 37 kBq | ||||
| Evaluation Method | Gamma counter assay | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is an excellent target for imaging and radionuclide therapy of prostate cancer. Recently, [177Lu]Lu-PSMA-617 (Pluvicto) was approved by the FDA for radionuclide therapy. To develop hetero-bivalent agents targeting both PSMA and bone metastasis, [177Lu]Lu-P17-079 ([177Lu]Lu-1) and [177Lu]Lu-P17-081 ([177Lu]Lu-2) were prepared. In vivo biodistribution studies of [177Lu]Lu-PSMA-617, [177Lu]Lu-1, and [177Lu]Lu-2 in mice bearing PC3-PIP (PSMA positive) tumor showed high uptake in PSMA-positive tumor (14.5, 14.7, and 11.3% ID/g at 1 h, respectively) and distinctively different bone uptakes (0.52, 6.52, and 5.82% ID/g at 1 h, respectively). PET imaging using [68Ga]Ga-P17-079 ([68Ga]Ga-1) in the same mouse model displayed excellent images confirming the expected dual-targeting to PSMA-positive tumor and bone. Results suggest that [177Lu]Lu-P17-079 ([177Lu]Lu-1) is a promising candidate for further development as a hetero-bivalent radionuclide therapy agent targeting both PSMA expression and bone metastases for the treatment of prostate cancer.
Click to Show/Hide
|
||||
| Description |
As expected, the PSMA-targeting hetero-bivalent agents, [177Lu]Lu-1 and [177Lu]Lu-2, displayed excellent cell uptake in PC3-PIP PSMA-positive cells. Furthermore, [177Lu]Lu-1 displayed a higher uptake (about 2 times higher, p < 0.05) as that of [177Lu]Lu-2. The higher uptake in PSMA-positive cells suggested that [177Lu]Lu-1 may more readily penetrate the cell membrane and may be more localized in the PSMA-positive tumor cells. The uptake was specifically inhibited by blocking study (in the presence of 1 uM cold PSMA-617). Both agents did not show any significant cell uptakes in PSMA-negative cells (PC3 cells). Cell uptake kinetics of [177Lu]Lu-1 and [177Lu]Lu-2 showed very rapid uptakes in PC3-PIP PSMA-positive cells (5, 15, 30, 60, and 120 min, respectively). As a positive control, [177Lu]Lu-PSMA-617 also showed excellent cell uptake under a similar condition. It is noted that [177Lu]Lu-2 showed a very similar uptake kinetics as that of [177Lu]Lu-PSMA-617, while [177Lu]Lu-1 displayed a significantly higher and faster uptake. Apparently, a small change in the chemical structure of the location of the bisphosphonate group between [177Lu]Lu-1 and [177Lu]Lu-2 may have contributed to this observation. No obvious reason could be contributed to the superior cell uptake of [177Lu]Lu-1. This distinctive difference in in vitro cell uptake may also be responsible for variations of PSMA-positive tumor uptakes in vivo.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC3-PIP PSMA-positive cell | CVCL_0035 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell uptake rate |
8%
|
|||
| Administration Time | 60 min | ||||
| Administration Dosage | 37 kBq | ||||
| Evaluation Method | Gamma counter assay | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is an excellent target for imaging and radionuclide therapy of prostate cancer. Recently, [177Lu]Lu-PSMA-617 (Pluvicto) was approved by the FDA for radionuclide therapy. To develop hetero-bivalent agents targeting both PSMA and bone metastasis, [177Lu]Lu-P17-079 ([177Lu]Lu-1) and [177Lu]Lu-P17-081 ([177Lu]Lu-2) were prepared. In vivo biodistribution studies of [177Lu]Lu-PSMA-617, [177Lu]Lu-1, and [177Lu]Lu-2 in mice bearing PC3-PIP (PSMA positive) tumor showed high uptake in PSMA-positive tumor (14.5, 14.7, and 11.3% ID/g at 1 h, respectively) and distinctively different bone uptakes (0.52, 6.52, and 5.82% ID/g at 1 h, respectively). PET imaging using [68Ga]Ga-P17-079 ([68Ga]Ga-1) in the same mouse model displayed excellent images confirming the expected dual-targeting to PSMA-positive tumor and bone. Results suggest that [177Lu]Lu-P17-079 ([177Lu]Lu-1) is a promising candidate for further development as a hetero-bivalent radionuclide therapy agent targeting both PSMA expression and bone metastases for the treatment of prostate cancer.
Click to Show/Hide
|
||||
| Description |
As expected, the PSMA-targeting hetero-bivalent agents, [177Lu]Lu-1 and [177Lu]Lu-2, displayed excellent cell uptake in PC3-PIP PSMA-positive cells. Furthermore, [177Lu]Lu-1 displayed a higher uptake (about 2 times higher, p < 0.05) as that of [177Lu]Lu-2. The higher uptake in PSMA-positive cells suggested that [177Lu]Lu-1 may more readily penetrate the cell membrane and may be more localized in the PSMA-positive tumor cells. The uptake was specifically inhibited by blocking study (in the presence of 1 uM cold PSMA-617). Both agents did not show any significant cell uptakes in PSMA-negative cells (PC3 cells). Cell uptake kinetics of [177Lu]Lu-1 and [177Lu]Lu-2 showed very rapid uptakes in PC3-PIP PSMA-positive cells (5, 15, 30, 60, and 120 min, respectively). As a positive control, [177Lu]Lu-PSMA-617 also showed excellent cell uptake under a similar condition. It is noted that [177Lu]Lu-2 showed a very similar uptake kinetics as that of [177Lu]Lu-PSMA-617, while [177Lu]Lu-1 displayed a significantly higher and faster uptake. Apparently, a small change in the chemical structure of the location of the bisphosphonate group between [177Lu]Lu-1 and [177Lu]Lu-2 may have contributed to this observation. No obvious reason could be contributed to the superior cell uptake of [177Lu]Lu-1. This distinctive difference in in vitro cell uptake may also be responsible for variations of PSMA-positive tumor uptakes in vivo.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC3-PIP PSMA-positive cell | CVCL_0035 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell uptake rate |
10%
|
|||
| Administration Time | 120 min | ||||
| Administration Dosage | 37 kBq | ||||
| Evaluation Method | Gamma counter assay | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is an excellent target for imaging and radionuclide therapy of prostate cancer. Recently, [177Lu]Lu-PSMA-617 (Pluvicto) was approved by the FDA for radionuclide therapy. To develop hetero-bivalent agents targeting both PSMA and bone metastasis, [177Lu]Lu-P17-079 ([177Lu]Lu-1) and [177Lu]Lu-P17-081 ([177Lu]Lu-2) were prepared. In vivo biodistribution studies of [177Lu]Lu-PSMA-617, [177Lu]Lu-1, and [177Lu]Lu-2 in mice bearing PC3-PIP (PSMA positive) tumor showed high uptake in PSMA-positive tumor (14.5, 14.7, and 11.3% ID/g at 1 h, respectively) and distinctively different bone uptakes (0.52, 6.52, and 5.82% ID/g at 1 h, respectively). PET imaging using [68Ga]Ga-P17-079 ([68Ga]Ga-1) in the same mouse model displayed excellent images confirming the expected dual-targeting to PSMA-positive tumor and bone. Results suggest that [177Lu]Lu-P17-079 ([177Lu]Lu-1) is a promising candidate for further development as a hetero-bivalent radionuclide therapy agent targeting both PSMA expression and bone metastases for the treatment of prostate cancer.
Click to Show/Hide
|
||||
| Description |
As expected, the PSMA-targeting hetero-bivalent agents, [177Lu]Lu-1 and [177Lu]Lu-2, displayed excellent cell uptake in PC3-PIP PSMA-positive cells. Furthermore, [177Lu]Lu-1 displayed a higher uptake (about 2 times higher, p < 0.05) as that of [177Lu]Lu-2. The higher uptake in PSMA-positive cells suggested that [177Lu]Lu-1 may more readily penetrate the cell membrane and may be more localized in the PSMA-positive tumor cells. The uptake was specifically inhibited by blocking study (in the presence of 1 uM cold PSMA-617). Both agents did not show any significant cell uptakes in PSMA-negative cells (PC3 cells). Cell uptake kinetics of [177Lu]Lu-1 and [177Lu]Lu-2 showed very rapid uptakes in PC3-PIP PSMA-positive cells (5, 15, 30, 60, and 120 min, respectively). As a positive control, [177Lu]Lu-PSMA-617 also showed excellent cell uptake under a similar condition. It is noted that [177Lu]Lu-2 showed a very similar uptake kinetics as that of [177Lu]Lu-PSMA-617, while [177Lu]Lu-1 displayed a significantly higher and faster uptake. Apparently, a small change in the chemical structure of the location of the bisphosphonate group between [177Lu]Lu-1 and [177Lu]Lu-2 may have contributed to this observation. No obvious reason could be contributed to the superior cell uptake of [177Lu]Lu-1. This distinctive difference in in vitro cell uptake may also be responsible for variations of PSMA-positive tumor uptakes in vivo.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC3-PIP PSMA-positive cell | CVCL_0035 | ||
References
