Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00026
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
175Lu-PSMA-617
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
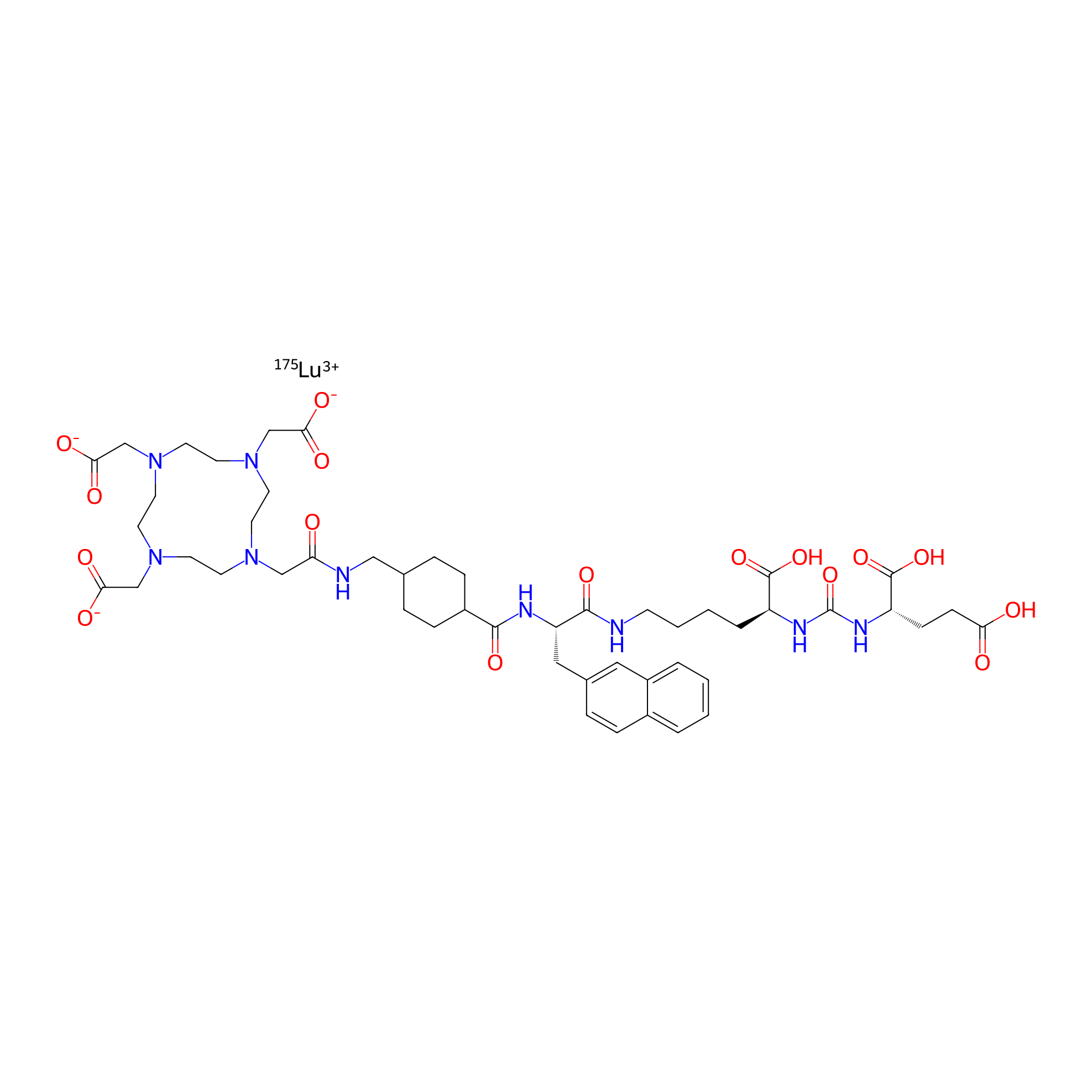
|
|||||
| Peptide Name |
PSMA-617
|
Peptide Info | ||||
| Receptor Name |
Glutamate carboxypeptidase 2 (FOLH1)
|
Receptor Info | ||||
| Drug Name |
Lutetium-175
|
Drug Info | ||||
| Therapeutic Target |
Human Deoxyribonucleic acid (hDNA)
|
Target Info | ||||
| Linker Name |
(S)-2-(4-(aminomethyl)cyclohexane-1-carboxamido)-3-(naphthalen-2-yl)propanoic acid
|
Linker Info | ||||
| Peptide Modified Type |
The modification of binding with chemical molecules
|
|||||
| Modified Segment |
Urea
|
|||||
| Formula |
C49H68LuN9O16
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 1214.070772 | ||||
| Lipid-water partition coefficient (xlogp) | -4.3821 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 8 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 17 | |||||
| Rotatable Bond Count (rotbonds) | 27 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell blocking rate | < 0.50% | |||
| Administration Time | 2 h | ||||
| Administration Dosage | 3.75 nM | ||||
| Evaluation Method | ICP-MS or γ-counting assay | ||||
| MOA of PDC |
This study addresses the question whether inductively coupled plasma mass spectrometry (ICP-MS) can be used as a method for the in vitro and in vivo characterization of non-radioactive metal conjugates to predict the properties of analogous radiopharmaceuticals. In a proof-of-concept study, the prostate-specific membrane antigen (PSMA)-targeting [175Lu]Lu-PSMA-617 and [159Tb]Tb-PSMA-617 were compared with their respective radiolabeled analogues, [177Lu]Lu-PSMA-617 (PLUVICTO, Novartis) and [161Tb]Tb-PSMA-617. ICP-MS and conventional γ-counting of the cell samples revealed almost identical results (<6% absolute difference between the two technologies) for the in vitro uptake and internalization of the (radio)metal conjugates, irrespective of the employed methodology. In vivo, an equal uptake in PSMA-positive PC-3 PIP tumor xenografts was determined 1 h after the injection of [175Lu]Lu-/[177Lu]Lu-PSMA-617 (41 ± 6% ID/g and 44 ± 12% IA/g, respectively) and [159Tb]Tb-/[161Tb]Tb-PSMA-617 (44 ± 5% ID/g and 44 ± 5% IA/g, respectively). It was further revealed that it is crucial to use the same ratios of the (radio)metal-labeled and unlabeled ligands for both methodologies to obtain equal data in organs in which receptor saturation was reached such as the kidneys (12 ± 2% ID/g vs 10 ± 1% IA/g, 1 h after injection). The data of this study demonstrate that the use of high-sensitivity ICP-MS allows reliable and predictive quantification of compounds labeled with stable metal isotopes in cell and tissue samples obtained in preclinical studies. It can, hence, be employed as a valid alternative to the state-of-the-art γ-counting methodology to detect radioactive ligands.
Click to Show/Hide
|
||||
| Description |
In LNCaP cells, the uptake of [175Lu]Lu-PSMA-617 determined by ICP-MS was 13 ± 2% (after 2 h) and 18 ± 2% (after 4 h). γ-Counting revealed the uptake of [177Lu]Lu-PSMA-617 in LNCaP cells of 11 ± 1% (after 2 h) and 14 ± 1% (after 4 h). For [159Tb]Tb-PSMA-617, analysis by ICP-MS revealed the uptake in LNCaP cells of 12 ± 1% (after 2 h) and 13 ± 1% (after 4 h). For the radioligand [161Tb]Tb-PSMA-617, the uptake in LNCaP cells determined by γ-counting was 7.8 ± 0.8% (after 2 h) and 9.7 ± 0.2% (after 4 h).
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP cell | CVCL_0395 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell blocking rate | < 0.50% | |||
| Administration Time | 4 h | ||||
| Administration Dosage | 3.75 nM | ||||
| Evaluation Method | ICP-MS or γ-counting assay | ||||
| MOA of PDC |
This study addresses the question whether inductively coupled plasma mass spectrometry (ICP-MS) can be used as a method for the in vitro and in vivo characterization of non-radioactive metal conjugates to predict the properties of analogous radiopharmaceuticals. In a proof-of-concept study, the prostate-specific membrane antigen (PSMA)-targeting [175Lu]Lu-PSMA-617 and [159Tb]Tb-PSMA-617 were compared with their respective radiolabeled analogues, [177Lu]Lu-PSMA-617 (PLUVICTO, Novartis) and [161Tb]Tb-PSMA-617. ICP-MS and conventional γ-counting of the cell samples revealed almost identical results (<6% absolute difference between the two technologies) for the in vitro uptake and internalization of the (radio)metal conjugates, irrespective of the employed methodology. In vivo, an equal uptake in PSMA-positive PC-3 PIP tumor xenografts was determined 1 h after the injection of [175Lu]Lu-/[177Lu]Lu-PSMA-617 (41 ± 6% ID/g and 44 ± 12% IA/g, respectively) and [159Tb]Tb-/[161Tb]Tb-PSMA-617 (44 ± 5% ID/g and 44 ± 5% IA/g, respectively). It was further revealed that it is crucial to use the same ratios of the (radio)metal-labeled and unlabeled ligands for both methodologies to obtain equal data in organs in which receptor saturation was reached such as the kidneys (12 ± 2% ID/g vs 10 ± 1% IA/g, 1 h after injection). The data of this study demonstrate that the use of high-sensitivity ICP-MS allows reliable and predictive quantification of compounds labeled with stable metal isotopes in cell and tissue samples obtained in preclinical studies. It can, hence, be employed as a valid alternative to the state-of-the-art γ-counting methodology to detect radioactive ligands.
Click to Show/Hide
|
||||
| Description |
In LNCaP cells, the uptake of [175Lu]Lu-PSMA-617 determined by ICP-MS was 13 ± 2% (after 2 h) and 18 ± 2% (after 4 h). γ-Counting revealed the uptake of [177Lu]Lu-PSMA-617 in LNCaP cells of 11 ± 1% (after 2 h) and 14 ± 1% (after 4 h). For [159Tb]Tb-PSMA-617, analysis by ICP-MS revealed the uptake in LNCaP cells of 12 ± 1% (after 2 h) and 13 ± 1% (after 4 h). For the radioligand [161Tb]Tb-PSMA-617, the uptake in LNCaP cells determined by γ-counting was 7.8 ± 0.8% (after 2 h) and 9.7 ± 0.2% (after 4 h).
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP cell | CVCL_0395 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell internalization rate |
8.3 ± 1.4 %
|
|||
| Administration Time | 2 h | ||||
| Administration Dosage | 3.75 nM | ||||
| Evaluation Method | ICP-MS or γ-counting assay | ||||
| MOA of PDC |
This study addresses the question whether inductively coupled plasma mass spectrometry (ICP-MS) can be used as a method for the in vitro and in vivo characterization of non-radioactive metal conjugates to predict the properties of analogous radiopharmaceuticals. In a proof-of-concept study, the prostate-specific membrane antigen (PSMA)-targeting [175Lu]Lu-PSMA-617 and [159Tb]Tb-PSMA-617 were compared with their respective radiolabeled analogues, [177Lu]Lu-PSMA-617 (PLUVICTO, Novartis) and [161Tb]Tb-PSMA-617. ICP-MS and conventional γ-counting of the cell samples revealed almost identical results (<6% absolute difference between the two technologies) for the in vitro uptake and internalization of the (radio)metal conjugates, irrespective of the employed methodology. In vivo, an equal uptake in PSMA-positive PC-3 PIP tumor xenografts was determined 1 h after the injection of [175Lu]Lu-/[177Lu]Lu-PSMA-617 (41 ± 6% ID/g and 44 ± 12% IA/g, respectively) and [159Tb]Tb-/[161Tb]Tb-PSMA-617 (44 ± 5% ID/g and 44 ± 5% IA/g, respectively). It was further revealed that it is crucial to use the same ratios of the (radio)metal-labeled and unlabeled ligands for both methodologies to obtain equal data in organs in which receptor saturation was reached such as the kidneys (12 ± 2% ID/g vs 10 ± 1% IA/g, 1 h after injection). The data of this study demonstrate that the use of high-sensitivity ICP-MS allows reliable and predictive quantification of compounds labeled with stable metal isotopes in cell and tissue samples obtained in preclinical studies. It can, hence, be employed as a valid alternative to the state-of-the-art γ-counting methodology to detect radioactive ligands.
Click to Show/Hide
|
||||
| Description |
In LNCaP cells, the uptake of [175Lu]Lu-PSMA-617 determined by ICP-MS was 13 ± 2% (after 2 h) and 18 ± 2% (after 4 h). γ-Counting revealed the uptake of [177Lu]Lu-PSMA-617 in LNCaP cells of 11 ± 1% (after 2 h) and 14 ± 1% (after 4 h). For [159Tb]Tb-PSMA-617, analysis by ICP-MS revealed the uptake in LNCaP cells of 12 ± 1% (after 2 h) and 13 ± 1% (after 4 h). For the radioligand [161Tb]Tb-PSMA-617, the uptake in LNCaP cells determined by γ-counting was 7.8 ± 0.8% (after 2 h) and 9.7 ± 0.2% (after 4 h).
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP cell | CVCL_0395 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell internalization rate |
13 ± 2 %
|
|||
| Administration Time | 4 h | ||||
| Administration Dosage | 3.75 nM | ||||
| Evaluation Method | ICP-MS or γ-counting assay | ||||
| MOA of PDC |
This study addresses the question whether inductively coupled plasma mass spectrometry (ICP-MS) can be used as a method for the in vitro and in vivo characterization of non-radioactive metal conjugates to predict the properties of analogous radiopharmaceuticals. In a proof-of-concept study, the prostate-specific membrane antigen (PSMA)-targeting [175Lu]Lu-PSMA-617 and [159Tb]Tb-PSMA-617 were compared with their respective radiolabeled analogues, [177Lu]Lu-PSMA-617 (PLUVICTO, Novartis) and [161Tb]Tb-PSMA-617. ICP-MS and conventional γ-counting of the cell samples revealed almost identical results (<6% absolute difference between the two technologies) for the in vitro uptake and internalization of the (radio)metal conjugates, irrespective of the employed methodology. In vivo, an equal uptake in PSMA-positive PC-3 PIP tumor xenografts was determined 1 h after the injection of [175Lu]Lu-/[177Lu]Lu-PSMA-617 (41 ± 6% ID/g and 44 ± 12% IA/g, respectively) and [159Tb]Tb-/[161Tb]Tb-PSMA-617 (44 ± 5% ID/g and 44 ± 5% IA/g, respectively). It was further revealed that it is crucial to use the same ratios of the (radio)metal-labeled and unlabeled ligands for both methodologies to obtain equal data in organs in which receptor saturation was reached such as the kidneys (12 ± 2% ID/g vs 10 ± 1% IA/g, 1 h after injection). The data of this study demonstrate that the use of high-sensitivity ICP-MS allows reliable and predictive quantification of compounds labeled with stable metal isotopes in cell and tissue samples obtained in preclinical studies. It can, hence, be employed as a valid alternative to the state-of-the-art γ-counting methodology to detect radioactive ligands.
Click to Show/Hide
|
||||
| Description |
In LNCaP cells, the uptake of [175Lu]Lu-PSMA-617 determined by ICP-MS was 13 ± 2% (after 2 h) and 18 ± 2% (after 4 h). γ-Counting revealed the uptake of [177Lu]Lu-PSMA-617 in LNCaP cells of 11 ± 1% (after 2 h) and 14 ± 1% (after 4 h). For [159Tb]Tb-PSMA-617, analysis by ICP-MS revealed the uptake in LNCaP cells of 12 ± 1% (after 2 h) and 13 ± 1% (after 4 h). For the radioligand [161Tb]Tb-PSMA-617, the uptake in LNCaP cells determined by γ-counting was 7.8 ± 0.8% (after 2 h) and 9.7 ± 0.2% (after 4 h).
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP cell | CVCL_0395 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell internalization rate |
15 ± 1 %
|
|||
| Administration Time | 2 h | ||||
| Administration Dosage | 3.75 nM | ||||
| Evaluation Method | ICP-MS or γ-counting assay | ||||
| MOA of PDC |
This study addresses the question whether inductively coupled plasma mass spectrometry (ICP-MS) can be used as a method for the in vitro and in vivo characterization of non-radioactive metal conjugates to predict the properties of analogous radiopharmaceuticals. In a proof-of-concept study, the prostate-specific membrane antigen (PSMA)-targeting [175Lu]Lu-PSMA-617 and [159Tb]Tb-PSMA-617 were compared with their respective radiolabeled analogues, [177Lu]Lu-PSMA-617 (PLUVICTO, Novartis) and [161Tb]Tb-PSMA-617. ICP-MS and conventional γ-counting of the cell samples revealed almost identical results (<6% absolute difference between the two technologies) for the in vitro uptake and internalization of the (radio)metal conjugates, irrespective of the employed methodology. In vivo, an equal uptake in PSMA-positive PC-3 PIP tumor xenografts was determined 1 h after the injection of [175Lu]Lu-/[177Lu]Lu-PSMA-617 (41 ± 6% ID/g and 44 ± 12% IA/g, respectively) and [159Tb]Tb-/[161Tb]Tb-PSMA-617 (44 ± 5% ID/g and 44 ± 5% IA/g, respectively). It was further revealed that it is crucial to use the same ratios of the (radio)metal-labeled and unlabeled ligands for both methodologies to obtain equal data in organs in which receptor saturation was reached such as the kidneys (12 ± 2% ID/g vs 10 ± 1% IA/g, 1 h after injection). The data of this study demonstrate that the use of high-sensitivity ICP-MS allows reliable and predictive quantification of compounds labeled with stable metal isotopes in cell and tissue samples obtained in preclinical studies. It can, hence, be employed as a valid alternative to the state-of-the-art γ-counting methodology to detect radioactive ligands.
Click to Show/Hide
|
||||
| Description |
The uptake of [175Lu]Lu-PSMA-617 in PC-3 PIP tumor cells determined by ICP-MS was 57 ± 5% (after 2 h) and 69 ± 6% (after 4 h), whereas it was 55 ± 2% (after 2 h) and 68 ± 5% (after 4 h) as determined by γ-counting for [177Lu]Lu-PSMA-617. For [159Tb]Tb-PSMA-617, the uptake in PC-3 PIP tumor cells determined by ICP-MS was 51 ± 4% (after 2 h) and 60 ± 5% (after 4 h), whereas for [161Tb]Tb-PSMA-617, it was 47 ± 3% (after 2 h) and 54 ± 6% (after 4 h) as determined by γ-counting.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PSMA-positive PC-3 PIP cell | CVCL_0035 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell internalization rate |
22 ± 3 %
|
|||
| Administration Time | 4 h | ||||
| Administration Dosage | 3.75 nM | ||||
| Evaluation Method | ICP-MS or γ-counting assay | ||||
| MOA of PDC |
This study addresses the question whether inductively coupled plasma mass spectrometry (ICP-MS) can be used as a method for the in vitro and in vivo characterization of non-radioactive metal conjugates to predict the properties of analogous radiopharmaceuticals. In a proof-of-concept study, the prostate-specific membrane antigen (PSMA)-targeting [175Lu]Lu-PSMA-617 and [159Tb]Tb-PSMA-617 were compared with their respective radiolabeled analogues, [177Lu]Lu-PSMA-617 (PLUVICTO, Novartis) and [161Tb]Tb-PSMA-617. ICP-MS and conventional γ-counting of the cell samples revealed almost identical results (<6% absolute difference between the two technologies) for the in vitro uptake and internalization of the (radio)metal conjugates, irrespective of the employed methodology. In vivo, an equal uptake in PSMA-positive PC-3 PIP tumor xenografts was determined 1 h after the injection of [175Lu]Lu-/[177Lu]Lu-PSMA-617 (41 ± 6% ID/g and 44 ± 12% IA/g, respectively) and [159Tb]Tb-/[161Tb]Tb-PSMA-617 (44 ± 5% ID/g and 44 ± 5% IA/g, respectively). It was further revealed that it is crucial to use the same ratios of the (radio)metal-labeled and unlabeled ligands for both methodologies to obtain equal data in organs in which receptor saturation was reached such as the kidneys (12 ± 2% ID/g vs 10 ± 1% IA/g, 1 h after injection). The data of this study demonstrate that the use of high-sensitivity ICP-MS allows reliable and predictive quantification of compounds labeled with stable metal isotopes in cell and tissue samples obtained in preclinical studies. It can, hence, be employed as a valid alternative to the state-of-the-art γ-counting methodology to detect radioactive ligands.
Click to Show/Hide
|
||||
| Description |
The uptake of [175Lu]Lu-PSMA-617 in PC-3 PIP tumor cells determined by ICP-MS was 57 ± 5% (after 2 h) and 69 ± 6% (after 4 h), whereas it was 55 ± 2% (after 2 h) and 68 ± 5% (after 4 h) as determined by γ-counting for [177Lu]Lu-PSMA-617. For [159Tb]Tb-PSMA-617, the uptake in PC-3 PIP tumor cells determined by ICP-MS was 51 ± 4% (after 2 h) and 60 ± 5% (after 4 h), whereas for [161Tb]Tb-PSMA-617, it was 47 ± 3% (after 2 h) and 54 ± 6% (after 4 h) as determined by γ-counting.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PSMA-positive PC-3 PIP cell | CVCL_0035 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell uptake rate | < 0.10% | |||
| Administration Time | 4 h | ||||
| Administration Dosage | 3.75 nM | ||||
| Evaluation Method | ICP-MS or γ-counting assay | ||||
| MOA of PDC |
This study addresses the question whether inductively coupled plasma mass spectrometry (ICP-MS) can be used as a method for the in vitro and in vivo characterization of non-radioactive metal conjugates to predict the properties of analogous radiopharmaceuticals. In a proof-of-concept study, the prostate-specific membrane antigen (PSMA)-targeting [175Lu]Lu-PSMA-617 and [159Tb]Tb-PSMA-617 were compared with their respective radiolabeled analogues, [177Lu]Lu-PSMA-617 (PLUVICTO, Novartis) and [161Tb]Tb-PSMA-617. ICP-MS and conventional γ-counting of the cell samples revealed almost identical results (<6% absolute difference between the two technologies) for the in vitro uptake and internalization of the (radio)metal conjugates, irrespective of the employed methodology. In vivo, an equal uptake in PSMA-positive PC-3 PIP tumor xenografts was determined 1 h after the injection of [175Lu]Lu-/[177Lu]Lu-PSMA-617 (41 ± 6% ID/g and 44 ± 12% IA/g, respectively) and [159Tb]Tb-/[161Tb]Tb-PSMA-617 (44 ± 5% ID/g and 44 ± 5% IA/g, respectively). It was further revealed that it is crucial to use the same ratios of the (radio)metal-labeled and unlabeled ligands for both methodologies to obtain equal data in organs in which receptor saturation was reached such as the kidneys (12 ± 2% ID/g vs 10 ± 1% IA/g, 1 h after injection). The data of this study demonstrate that the use of high-sensitivity ICP-MS allows reliable and predictive quantification of compounds labeled with stable metal isotopes in cell and tissue samples obtained in preclinical studies. It can, hence, be employed as a valid alternative to the state-of-the-art γ-counting methodology to detect radioactive ligands.
Click to Show/Hide
|
||||
| Description |
The uptake of [175Lu]Lu-PSMA-617 in PC-3 PIP tumor cells determined by ICP-MS was 57 ± 5% (after 2 h) and 69 ± 6% (after 4 h), whereas it was 55 ± 2% (after 2 h) and 68 ± 5% (after 4 h) as determined by γ-counting for [177Lu]Lu-PSMA-617. For [159Tb]Tb-PSMA-617, the uptake in PC-3 PIP tumor cells determined by ICP-MS was 51 ± 4% (after 2 h) and 60 ± 5% (after 4 h), whereas for [161Tb]Tb-PSMA-617, it was 47 ± 3% (after 2 h) and 54 ± 6% (after 4 h) as determined by γ-counting.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PSMA-negative PC-3 flu cell | CVCL_0035 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell uptake rate |
13 ± 2 %
|
|||
| Administration Time | 2 h | ||||
| Administration Dosage | 3.75 nM | ||||
| Evaluation Method | ICP-MS or γ-counting assay | ||||
| MOA of PDC |
This study addresses the question whether inductively coupled plasma mass spectrometry (ICP-MS) can be used as a method for the in vitro and in vivo characterization of non-radioactive metal conjugates to predict the properties of analogous radiopharmaceuticals. In a proof-of-concept study, the prostate-specific membrane antigen (PSMA)-targeting [175Lu]Lu-PSMA-617 and [159Tb]Tb-PSMA-617 were compared with their respective radiolabeled analogues, [177Lu]Lu-PSMA-617 (PLUVICTO, Novartis) and [161Tb]Tb-PSMA-617. ICP-MS and conventional γ-counting of the cell samples revealed almost identical results (<6% absolute difference between the two technologies) for the in vitro uptake and internalization of the (radio)metal conjugates, irrespective of the employed methodology. In vivo, an equal uptake in PSMA-positive PC-3 PIP tumor xenografts was determined 1 h after the injection of [175Lu]Lu-/[177Lu]Lu-PSMA-617 (41 ± 6% ID/g and 44 ± 12% IA/g, respectively) and [159Tb]Tb-/[161Tb]Tb-PSMA-617 (44 ± 5% ID/g and 44 ± 5% IA/g, respectively). It was further revealed that it is crucial to use the same ratios of the (radio)metal-labeled and unlabeled ligands for both methodologies to obtain equal data in organs in which receptor saturation was reached such as the kidneys (12 ± 2% ID/g vs 10 ± 1% IA/g, 1 h after injection). The data of this study demonstrate that the use of high-sensitivity ICP-MS allows reliable and predictive quantification of compounds labeled with stable metal isotopes in cell and tissue samples obtained in preclinical studies. It can, hence, be employed as a valid alternative to the state-of-the-art γ-counting methodology to detect radioactive ligands.
Click to Show/Hide
|
||||
| Description |
In LNCaP cells, the uptake of [175Lu]Lu-PSMA-617 determined by ICP-MS was 13 ± 2% (after 2 h) and 18 ± 2% (after 4 h). γ-Counting revealed the uptake of [177Lu]Lu-PSMA-617 in LNCaP cells of 11 ± 1% (after 2 h) and 14 ± 1% (after 4 h). For [159Tb]Tb-PSMA-617, analysis by ICP-MS revealed the uptake in LNCaP cells of 12 ± 1% (after 2 h) and 13 ± 1% (after 4 h). For the radioligand [161Tb]Tb-PSMA-617, the uptake in LNCaP cells determined by γ-counting was 7.8 ± 0.8% (after 2 h) and 9.7 ± 0.2% (after 4 h).
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP cell | CVCL_0395 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell uptake rate |
18 ± 2 %
|
|||
| Administration Time | 4 h | ||||
| Administration Dosage | 3.75 nM | ||||
| Evaluation Method | ICP-MS or γ-counting assay | ||||
| MOA of PDC |
This study addresses the question whether inductively coupled plasma mass spectrometry (ICP-MS) can be used as a method for the in vitro and in vivo characterization of non-radioactive metal conjugates to predict the properties of analogous radiopharmaceuticals. In a proof-of-concept study, the prostate-specific membrane antigen (PSMA)-targeting [175Lu]Lu-PSMA-617 and [159Tb]Tb-PSMA-617 were compared with their respective radiolabeled analogues, [177Lu]Lu-PSMA-617 (PLUVICTO, Novartis) and [161Tb]Tb-PSMA-617. ICP-MS and conventional γ-counting of the cell samples revealed almost identical results (<6% absolute difference between the two technologies) for the in vitro uptake and internalization of the (radio)metal conjugates, irrespective of the employed methodology. In vivo, an equal uptake in PSMA-positive PC-3 PIP tumor xenografts was determined 1 h after the injection of [175Lu]Lu-/[177Lu]Lu-PSMA-617 (41 ± 6% ID/g and 44 ± 12% IA/g, respectively) and [159Tb]Tb-/[161Tb]Tb-PSMA-617 (44 ± 5% ID/g and 44 ± 5% IA/g, respectively). It was further revealed that it is crucial to use the same ratios of the (radio)metal-labeled and unlabeled ligands for both methodologies to obtain equal data in organs in which receptor saturation was reached such as the kidneys (12 ± 2% ID/g vs 10 ± 1% IA/g, 1 h after injection). The data of this study demonstrate that the use of high-sensitivity ICP-MS allows reliable and predictive quantification of compounds labeled with stable metal isotopes in cell and tissue samples obtained in preclinical studies. It can, hence, be employed as a valid alternative to the state-of-the-art γ-counting methodology to detect radioactive ligands.
Click to Show/Hide
|
||||
| Description |
In LNCaP cells, the uptake of [175Lu]Lu-PSMA-617 determined by ICP-MS was 13 ± 2% (after 2 h) and 18 ± 2% (after 4 h). γ-Counting revealed the uptake of [177Lu]Lu-PSMA-617 in LNCaP cells of 11 ± 1% (after 2 h) and 14 ± 1% (after 4 h). For [159Tb]Tb-PSMA-617, analysis by ICP-MS revealed the uptake in LNCaP cells of 12 ± 1% (after 2 h) and 13 ± 1% (after 4 h). For the radioligand [161Tb]Tb-PSMA-617, the uptake in LNCaP cells determined by γ-counting was 7.8 ± 0.8% (after 2 h) and 9.7 ± 0.2% (after 4 h).
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP cell | CVCL_0395 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell uptake rate |
57 ± 5 %
|
|||
| Administration Time | 2 h | ||||
| Administration Dosage | 3.75 nM | ||||
| Evaluation Method | ICP-MS or γ-counting assay | ||||
| MOA of PDC |
This study addresses the question whether inductively coupled plasma mass spectrometry (ICP-MS) can be used as a method for the in vitro and in vivo characterization of non-radioactive metal conjugates to predict the properties of analogous radiopharmaceuticals. In a proof-of-concept study, the prostate-specific membrane antigen (PSMA)-targeting [175Lu]Lu-PSMA-617 and [159Tb]Tb-PSMA-617 were compared with their respective radiolabeled analogues, [177Lu]Lu-PSMA-617 (PLUVICTO, Novartis) and [161Tb]Tb-PSMA-617. ICP-MS and conventional γ-counting of the cell samples revealed almost identical results (<6% absolute difference between the two technologies) for the in vitro uptake and internalization of the (radio)metal conjugates, irrespective of the employed methodology. In vivo, an equal uptake in PSMA-positive PC-3 PIP tumor xenografts was determined 1 h after the injection of [175Lu]Lu-/[177Lu]Lu-PSMA-617 (41 ± 6% ID/g and 44 ± 12% IA/g, respectively) and [159Tb]Tb-/[161Tb]Tb-PSMA-617 (44 ± 5% ID/g and 44 ± 5% IA/g, respectively). It was further revealed that it is crucial to use the same ratios of the (radio)metal-labeled and unlabeled ligands for both methodologies to obtain equal data in organs in which receptor saturation was reached such as the kidneys (12 ± 2% ID/g vs 10 ± 1% IA/g, 1 h after injection). The data of this study demonstrate that the use of high-sensitivity ICP-MS allows reliable and predictive quantification of compounds labeled with stable metal isotopes in cell and tissue samples obtained in preclinical studies. It can, hence, be employed as a valid alternative to the state-of-the-art γ-counting methodology to detect radioactive ligands.
Click to Show/Hide
|
||||
| Description |
The uptake of [175Lu]Lu-PSMA-617 in PC-3 PIP tumor cells determined by ICP-MS was 57 ± 5% (after 2 h) and 69 ± 6% (after 4 h), whereas it was 55 ± 2% (after 2 h) and 68 ± 5% (after 4 h) as determined by γ-counting for [177Lu]Lu-PSMA-617. For [159Tb]Tb-PSMA-617, the uptake in PC-3 PIP tumor cells determined by ICP-MS was 51 ± 4% (after 2 h) and 60 ± 5% (after 4 h), whereas for [161Tb]Tb-PSMA-617, it was 47 ± 3% (after 2 h) and 54 ± 6% (after 4 h) as determined by γ-counting.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PSMA-positive PC-3 PIP cell | CVCL_0035 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Cell uptake rate |
69 ± 6 %
|
|||
| Administration Time | 4 h | ||||
| Administration Dosage | 3.75 nM | ||||
| Evaluation Method | ICP-MS or γ-counting assay | ||||
| MOA of PDC |
This study addresses the question whether inductively coupled plasma mass spectrometry (ICP-MS) can be used as a method for the in vitro and in vivo characterization of non-radioactive metal conjugates to predict the properties of analogous radiopharmaceuticals. In a proof-of-concept study, the prostate-specific membrane antigen (PSMA)-targeting [175Lu]Lu-PSMA-617 and [159Tb]Tb-PSMA-617 were compared with their respective radiolabeled analogues, [177Lu]Lu-PSMA-617 (PLUVICTO, Novartis) and [161Tb]Tb-PSMA-617. ICP-MS and conventional γ-counting of the cell samples revealed almost identical results (<6% absolute difference between the two technologies) for the in vitro uptake and internalization of the (radio)metal conjugates, irrespective of the employed methodology. In vivo, an equal uptake in PSMA-positive PC-3 PIP tumor xenografts was determined 1 h after the injection of [175Lu]Lu-/[177Lu]Lu-PSMA-617 (41 ± 6% ID/g and 44 ± 12% IA/g, respectively) and [159Tb]Tb-/[161Tb]Tb-PSMA-617 (44 ± 5% ID/g and 44 ± 5% IA/g, respectively). It was further revealed that it is crucial to use the same ratios of the (radio)metal-labeled and unlabeled ligands for both methodologies to obtain equal data in organs in which receptor saturation was reached such as the kidneys (12 ± 2% ID/g vs 10 ± 1% IA/g, 1 h after injection). The data of this study demonstrate that the use of high-sensitivity ICP-MS allows reliable and predictive quantification of compounds labeled with stable metal isotopes in cell and tissue samples obtained in preclinical studies. It can, hence, be employed as a valid alternative to the state-of-the-art γ-counting methodology to detect radioactive ligands.
Click to Show/Hide
|
||||
| Description |
The uptake of [175Lu]Lu-PSMA-617 in PC-3 PIP tumor cells determined by ICP-MS was 57 ± 5% (after 2 h) and 69 ± 6% (after 4 h), whereas it was 55 ± 2% (after 2 h) and 68 ± 5% (after 4 h) as determined by γ-counting for [177Lu]Lu-PSMA-617. For [159Tb]Tb-PSMA-617, the uptake in PC-3 PIP tumor cells determined by ICP-MS was 51 ± 4% (after 2 h) and 60 ± 5% (after 4 h), whereas for [161Tb]Tb-PSMA-617, it was 47 ± 3% (after 2 h) and 54 ± 6% (after 4 h) as determined by γ-counting.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PSMA-positive PC-3 PIP cell | CVCL_0035 | ||
References
