Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00034
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
225Ac-PSMA-617
|
|||||
| Synonyms |
UH4J18XEL3; Actinium (225Ac) PSMA-617; UNII-UH4J18XEL3; Vipivotide tetraxetan actinium Ac-225; 2247839-16-1; ACTINIUM (225AC) PSMA-617 [WHO-DD]; ACTINATE(3-)-225AC, (N6-(N-((TRANS-4-(((2-(4,7,10-TRIS((CARBOXY-.KAPPA.O)METHYL)-1,4,7,10-TETRAAZACYCLODODEC-1-YL-.KAPPA.N1,.KAPPA.N4,.KAPPA.N7,KN10)ACETYL-.KAPPA.O)AMINO)METHYL)CYCLOHEXYL)CARBONYL)-3-(2-NAPHTHALENYL)-L-ALANYL)-N2-((((1S)-1,3-DICARBOXYPROPYL)AMINO)CARBONYL)-L-LYSINATO(6-))-
Click to Show/Hide
|
|||||
| PDC Status |
Phase 1
|
|||||
| Indication |
In total 2 Indication(s)
|
|||||
| Structure |
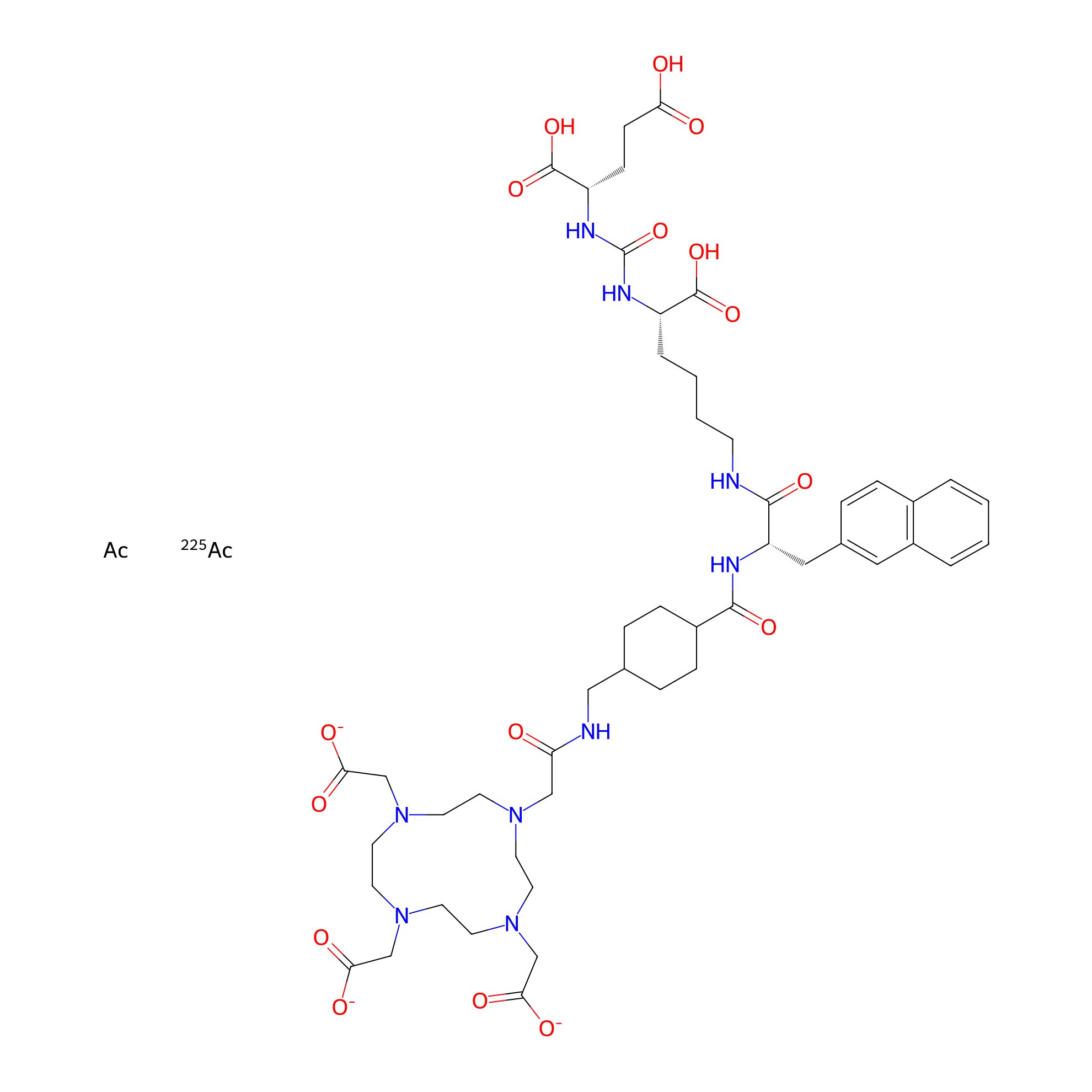
|
|||||
| Peptide Name |
PSMA-617
|
Peptide Info | ||||
| Receptor Name |
Glutamate carboxypeptidase 2 (FOLH1)
|
Receptor Info | ||||
| Drug Name |
Actinium-225
|
Drug Info | ||||
| Therapeutic Target |
Human Deoxyribonucleic acid (hDNA)
|
Target Info | ||||
| Linker Name |
(S)-2-(4-(aminomethyl)cyclohexane-1-carboxamido)-3-(naphthalen-2-yl)propanoic acid
|
Linker Info | ||||
| Peptide Modified Type |
The modification of binding with chemical molecules
|
|||||
| Modified Segment |
Urea
|
|||||
| Formula |
C49H68Ac2N9O16-3
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight (mw) | 1491.2 | ||||
| Lipid-water partition coefficient (xlogp) | Not Available | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 8 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 20 | |||||
| Rotatable Bond Count (rotbonds) | 24 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | >50% PSA decline |
65.00%
|
|||
| Patients Enrolled |
26 patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
A retrospective analysis of 26 men with progressive mCRPC that had undergone several previous therapies, including 177Lu PSMA-617, found that 225Ac PSMA-617 resulted in a ≥50% PSA drop in 65% of patients.
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | >50% PSA decline |
70.00%
|
|||
| Patients Enrolled |
Heavily pretreated, TRT-naive metastatic castration-resistant prostate cancer patients.
|
||||
| Evaluation Method | 68Ga-PSMA PET assay | ||||
| Description |
Therapy with 225Ac-PSMA-617 has shown remarkable efficacy (70% rate of PSA decline ≥ 50%, 29% complete response rate from 68Ga-PSMA PET) in heavily pretreated, TRT-nave patients.
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Complete response (CR) |
29%
|
|||
| Patients Enrolled |
Heavily pretreated, TRT-naive metastatic castration-resistant prostate cancer patients.
|
||||
| Evaluation Method | 68Ga-PSMA PET assay | ||||
| Description |
Therapy with 225Ac-PSMA-617 has shown remarkable efficacy (70% rate of PSA decline ≥ 50%, 29% complete response rate from 68Ga-PSMA PET) in heavily pretreated, TRT-nave patients.
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 1 xerostomia |
55.56%
|
|||
| Patients Enrolled |
18 patients with metastatic castration-resistant prostate cancer treated with tandem-cohort PRLT.
|
||||
| Administration Time | 1 week | ||||
| Administration Dosage | A median activity of 4.0 MBq (range 2.0-7.0 MBq) | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
One-third (6/18) of the patients reported grade 1 xerostomia at the baseline, while, after one cycle of Tandem-PRLT, xerostomia grade 1 in 10/18 patients and grade 2 in 2/18 patients was observed (p = 0.001), and 6/18 patients did not report any xerostomia at the follow-up. There was no patient-requested treatment discontinuation. The sXI-score increased significantly from 9.5 (95%CI: 7.0-14.2) before to 14.0 (95%CI: 11.5-19.6) after Tandem-PRLT (p = 0.005).
Click to Show/Hide
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 2 xerostomia |
11.11%
|
|||
| Patients Enrolled |
18 patients with metastatic castration-resistant prostate cancer treated with tandem-cohort PRLT.
|
||||
| Administration Time | 1 week | ||||
| Administration Dosage | A median activity of 4.0 MBq (range 2.0-7.0 MBq) | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
One-third (6/18) of the patients reported grade 1 xerostomia at the baseline, while, after one cycle of Tandem-PRLT, xerostomia grade 1 in 10/18 patients and grade 2 in 2/18 patients was observed (p = 0.001), and 6/18 patients did not report any xerostomia at the follow-up. There was no patient-requested treatment discontinuation. The sXI-score increased significantly from 9.5 (95%CI: 7.0-14.2) before to 14.0 (95%CI: 11.5-19.6) after Tandem-PRLT (p = 0.005).
Click to Show/Hide
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grading stage 0 visual salivary gland scintigraphy |
5.56%
|
|||
| Patients Enrolled |
18 patients with metastatic castration-resistant prostate cancer treated with tandem-cohort PRLT.
|
||||
| Administration Time | 1 week | ||||
| Administration Dosage | A median activity of 4.0 MBq (range 2.0-7.0 MBq) | ||||
| Evaluation Method | Salivary gland scintigraphy assay | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
The stage of xerostomia at the baseline according to SGS was 0 in 10/18 patients, 1 in 6/18 patients, and 2 in 2/18 patients, while, at the follow-up, stage 0 was noted in 1/18 patients, stage 1 in 8/18 patients, and stage 2 in 9/18 patients (p < 0.001).
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grading stage 1 visual salivary gland scintigraphy |
44.44%
|
|||
| Patients Enrolled |
18 patients with metastatic castration-resistant prostate cancer treated with tandem-cohort PRLT.
|
||||
| Administration Time | 1 week | ||||
| Administration Dosage | A median activity of 4.0 MBq (range 2.0-7.0 MBq) | ||||
| Evaluation Method | Salivary gland scintigraphy assay | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
The stage of xerostomia at the baseline according to SGS was 0 in 10/18 patients, 1 in 6/18 patients, and 2 in 2/18 patients, while, at the follow-up, stage 0 was noted in 1/18 patients, stage 1 in 8/18 patients, and stage 2 in 9/18 patients (p < 0.001).
|
||||
| Experiment 8 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grading stage 2 visual salivary gland scintigraphy |
50%
|
|||
| Patients Enrolled |
18 patients with metastatic castration-resistant prostate cancer treated with tandem-cohort PRLT.
|
||||
| Administration Time | 1 week | ||||
| Administration Dosage | A median activity of 4.0 MBq (range 2.0-7.0 MBq) | ||||
| Evaluation Method | Salivary gland scintigraphy assay | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
The stage of xerostomia at the baseline according to SGS was 0 in 10/18 patients, 1 in 6/18 patients, and 2 in 2/18 patients, while, at the follow-up, stage 0 was noted in 1/18 patients, stage 1 in 8/18 patients, and stage 2 in 9/18 patients (p < 0.001).
|
||||
| Experiment 9 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Adenocarcinoma prostate | ||||
| Efficacy Data | PSA decline |
41.6 ng/mL
|
|||
| Administration Time | 2 cycles | ||||
| Administration Dosage | 8.0 MBq/cycle | ||||
| MOA of PDC |
177Lu-PSMA-617 radioligand therapy (RLT) has evolved as a suitable alternative to existing therapeutic options in patients with metastatic castration-resistant prostate cancer. With the emergence of -emitters such as 225Ac, the efficacy of PSMA-RLT has further improved. Xerostomia and myelosuppression are common early treatment-emergent adverse events in patients receiving this therapy.
Click to Show/Hide
|
||||
| Description |
In view of refractory disease, the patient was administered 2 cycles of 225Ac-PSMA-617 (8.0 MBq/cycle) intravenously at 8 weeks intervals. Eight weeks following the second cycle, his serum PSA declined from 41.7 ng/mL to 0.1 ng/mL, and a repeat 68Ga-PSMA-11 PET/CT showed complete response.
|
||||
| In Vivo Model | A 76-year-old nondiabetic man, with adenocarcinoma prostate. | ||||
| Experiment 10 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Adenocarcinoma prostate | ||||
| Efficacy Data | PSA decline rate |
99.80%
|
|||
| Administration Time | 2 cycles | ||||
| Administration Dosage | 8.0 MBq/cycle | ||||
| MOA of PDC |
177Lu-PSMA-617 radioligand therapy (RLT) has evolved as a suitable alternative to existing therapeutic options in patients with metastatic castration-resistant prostate cancer. With the emergence of -emitters such as 225Ac, the efficacy of PSMA-RLT has further improved. Xerostomia and myelosuppression are common early treatment-emergent adverse events in patients receiving this therapy.
Click to Show/Hide
|
||||
| Description |
In view of refractory disease, the patient was administered 2 cycles of 225Ac-PSMA-617 (8.0 MBq/cycle) intravenously at 8 weeks intervals. Eight weeks following the second cycle, his serum PSA declined from 41.7 ng/mL to 0.1 ng/mL, and a repeat 68Ga-PSMA-11 PET/CT showed complete response.
|
||||
| In Vivo Model | A 76-year-old nondiabetic man, with adenocarcinoma prostate. | ||||
References
