Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00050
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
LT7-SS-DOX
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 3 Indication(s)
|
|||||
| Structure |
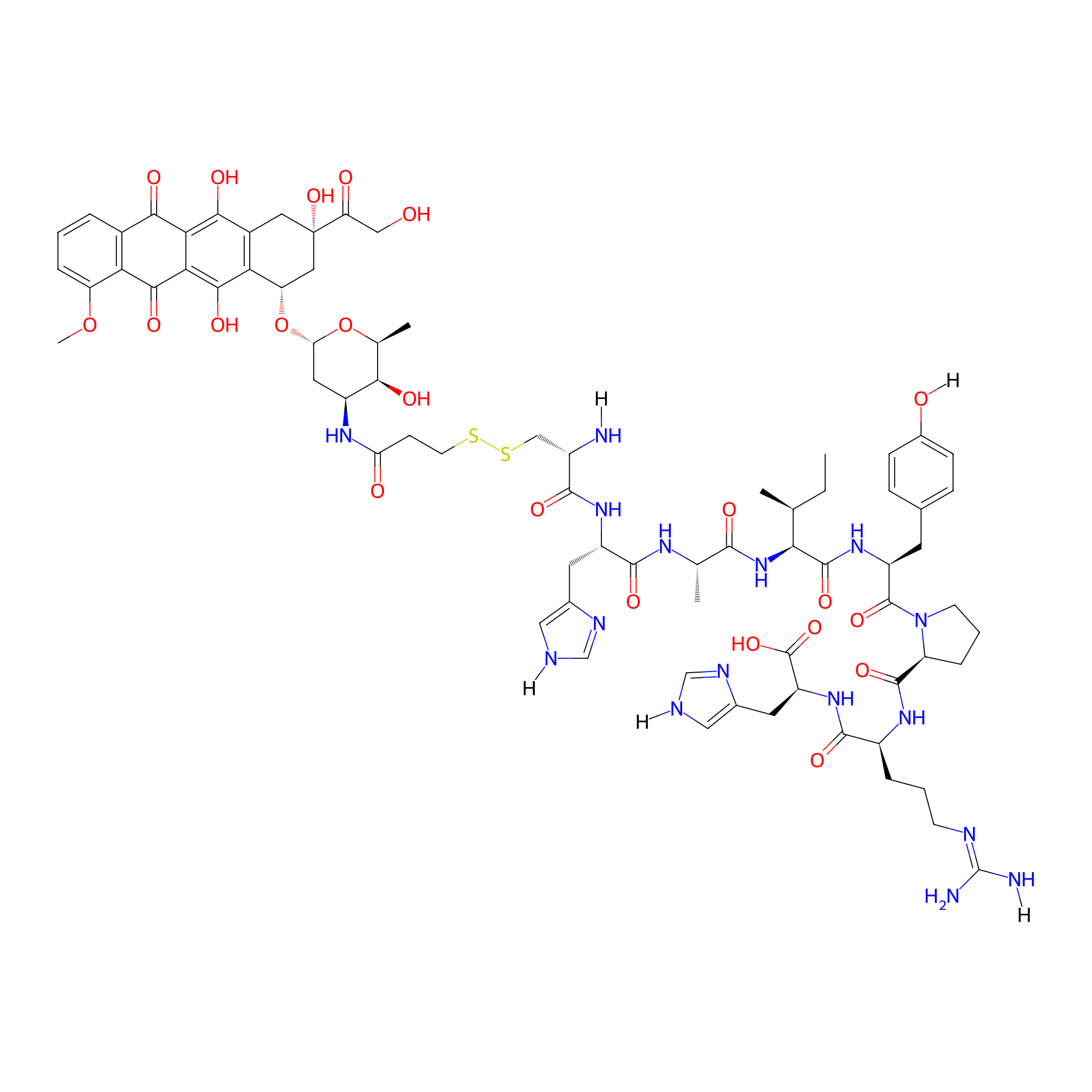
|
|||||
| Peptide Name |
DT7
|
Peptide Info | ||||
| Receptor Name |
Transferrin receptor protein 1 (TFRC)
|
Receptor Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
SPDP
|
Linker Info | ||||
| Formula |
C74H96N16O22S2
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 1625.806 | ||||
| Lipid-water partition coefficient (xlogp) | -1.7585 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 19 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 27 | |||||
| Rotatable Bond Count (rotbonds) | 40 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Proliferation inhibitory activity |
41.00%
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
Both conjugates exhibited targeted antiproliferative effects on TfR overexpressed tumor cells and little toxicity to TfR low-expressed normal cells compared with free DOX. Moreover, the DT7-SS-DOX conjugate possessed higher serum stability, more sustained reduction-triggered drug release characteristics, and stronger in vitro antiproliferative activity as compared to LT7-SS-DOX.
Click to Show/Hide
|
||||
| Description |
Even at an equal DOX concentration of 40 μM, the cell viability of the three types of tumor cells after exposure to this conjugate for 48 h were 41.0%, 61.5%, and 67.2%, respectively.
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Hepatoblastoma | ||||
| Efficacy Data | Proliferation inhibitory activity |
61.50%
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
Both conjugates exhibited targeted antiproliferative effects on TfR overexpressed tumor cells and little toxicity to TfR low-expressed normal cells compared with free DOX. Moreover, the DT7-SS-DOX conjugate possessed higher serum stability, more sustained reduction-triggered drug release characteristics, and stronger in vitro antiproliferative activity as compared to LT7-SS-DOX.
Click to Show/Hide
|
||||
| Description |
Even at an equal DOX concentration of 40 μM, the cell viability of the three types of tumor cells after exposure to this conjugate for 48 h were 41.0%, 61.5%, and 67.2%, respectively.
|
||||
| In Vitro Model | Hepatoblastoma | Hep-G2 cell | CVCL_0027 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Lung adenocarcinoma | ||||
| Efficacy Data | Proliferation inhibitory activity |
67.20%
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
Both conjugates exhibited targeted antiproliferative effects on TfR overexpressed tumor cells and little toxicity to TfR low-expressed normal cells compared with free DOX. Moreover, the DT7-SS-DOX conjugate possessed higher serum stability, more sustained reduction-triggered drug release characteristics, and stronger in vitro antiproliferative activity as compared to LT7-SS-DOX.
Click to Show/Hide
|
||||
| Description |
Even at an equal DOX concentration of 40 μM, the cell viability of the three types of tumor cells after exposure to this conjugate for 48 h were 41.0%, 61.5%, and 67.2%, respectively.
|
||||
| In Vitro Model | Lung adenocarcinoma | A-549 cell | CVCL_0023 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Hepatoblastoma | ||||
| Efficacy Data | Proliferation inhibitory activity |
73.10%
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
Both conjugates exhibited targeted antiproliferative effects on TfR overexpressed tumor cells and little toxicity to TfR low-expressed normal cells compared with free DOX. Moreover, the DT7-SS-DOX conjugate possessed higher serum stability, more sustained reduction-triggered drug release characteristics, and stronger in vitro antiproliferative activity as compared to LT7-SS-DOX.
Click to Show/Hide
|
||||
| Description |
The proliferation inhibitory activity of LT7-SS-DOX was the weakest among the three drugs because the cell viabilities of U87, HepG2, and A549 cells after incubation with LT7-SS-DOX (equal DOX concentration of 20 μM) for 48 h were 95.1%, 73.1%, and 83.2%, respectively.
|
||||
| In Vitro Model | Hepatoblastoma | Hep-G2 cell | CVCL_0027 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Lung adenocarcinoma | ||||
| Efficacy Data | Proliferation inhibitory activity |
83.20%
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
Both conjugates exhibited targeted antiproliferative effects on TfR overexpressed tumor cells and little toxicity to TfR low-expressed normal cells compared with free DOX. Moreover, the DT7-SS-DOX conjugate possessed higher serum stability, more sustained reduction-triggered drug release characteristics, and stronger in vitro antiproliferative activity as compared to LT7-SS-DOX.
Click to Show/Hide
|
||||
| Description |
The proliferation inhibitory activity of LT7-SS-DOX was the weakest among the three drugs because the cell viabilities of U87, HepG2, and A549 cells after incubation with LT7-SS-DOX (equal DOX concentration of 20 μM) for 48 h were 95.1%, 73.1%, and 83.2%, respectively.
|
||||
| In Vitro Model | Lung adenocarcinoma | A-549 cell | CVCL_0023 | ||
References
