Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00139
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
DOXO-EMCH-(RNWELRLK-PEG4)2
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
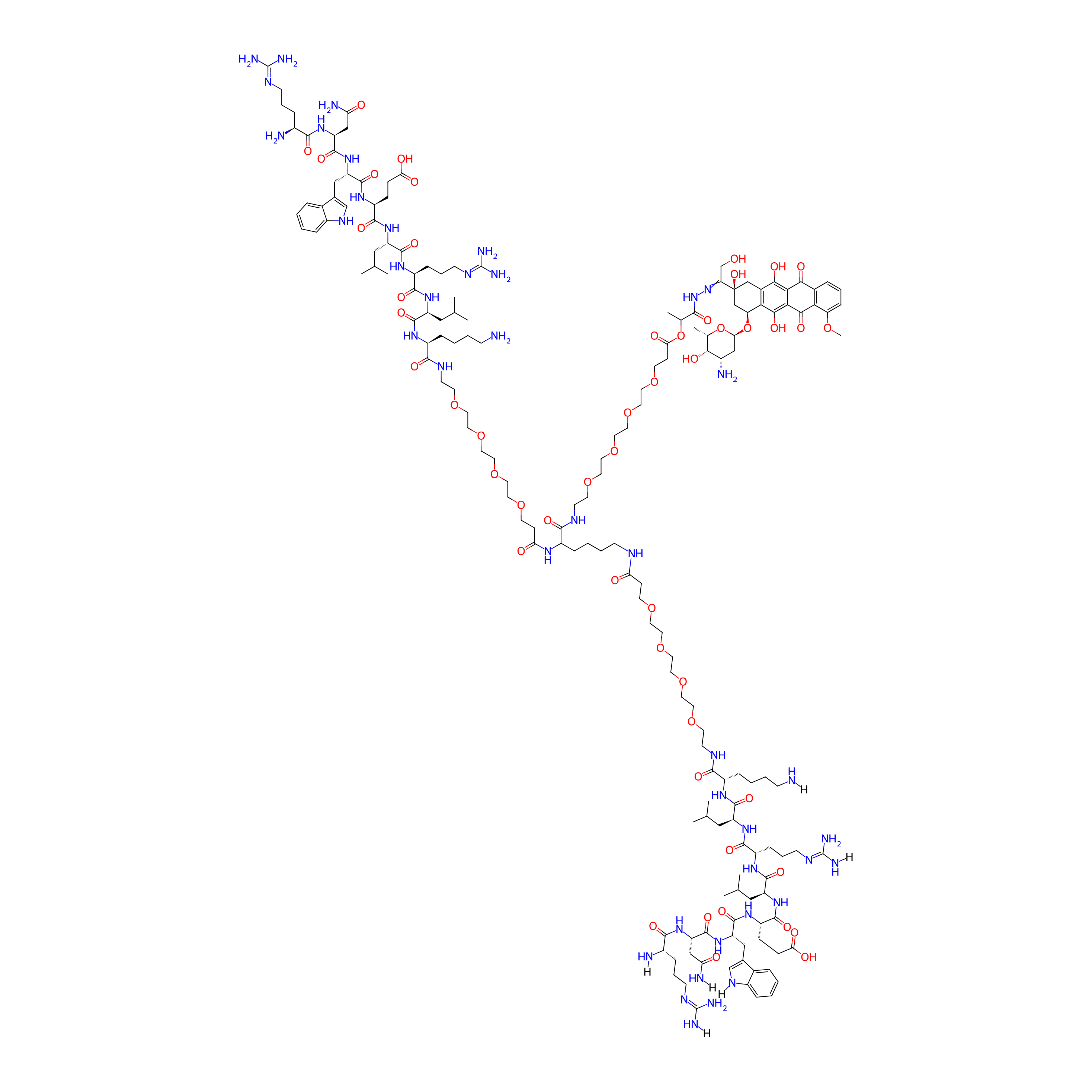
|
|||||
| Peptide Name |
RNWELRLK
|
Peptide Info | ||||
| Receptor Name |
Receptor tyrosine-protein kinase erbB-2 (ERBB2)
|
Receptor Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
EMCH
|
Linker Info | ||||
| Peptide Modified Type |
The modification of binding with chemical macromolecules
|
|||||
| Modified Segment |
PEG4
|
|||||
| Formula |
C169H272N42O50
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 3692.279 | ||||
| Lipid-water partition coefficient (xlogp) | -8.6475 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 44 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 58 | |||||
| Rotatable Bond Count (rotbonds) | 140 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
51.1 ± 3.1%
|
|||
| Administration Time | 14 days | ||||
| Administration Dosage | 5 mg/kg | ||||
| MOA of PDC |
Tumor-targeting peptide-drug conjugates (PDCs) have become a focus of research in recent years. However, due to the instability of peptides and their short in vivo effective half-life, they have limited clinical application. Herein, we propose a new DOX PDC based on a homodimer HER-2-targeting peptide and acid-sensitive hydrazone bond, which could enhance the anti-tumor effect of DOX and reduce systemic toxicities. The PDC could accurately deliver DOX into HER2-positive SKBR-3 cells, with it showing 2.9 times higher cellular uptake than free DOX and enhanced cytotoxicity with respect to IC50of 140 nM (vs. 410 nM for free DOX). In vitro assays showed that the PDC had high cellular internalization efficiency and cytotoxicity. In vivo anti-tumor experiments indicated that the PDC could significantly inhibit the growth of HER2-positive breast cancer xenografts in mice and reduce the side effects of DOX. In summary, we constructed a novel PDC molecule targeting HER2-positive tumors, which may overcome some deficiencies of DOX in breast cancer therapy.
Click to Show/Hide
|
||||
| Description |
In vivo anti-tumor studies were evaluated by using SKBR-3 xenografted (BALB/c nude) mice treated with the PDC, free DOX, or saline. Figure 5a demonstrates that the PDC had a much more powerful anti-tumor effect than free DOX, reducing tumor growth by 51.1 ± 3.1% on day 14 post treatment, while free DOX only achieved a 23.13 ± 2.4% reduction. Additionally, the PDC had a significantly higher tumor weight inhibition of 57.5 ± 3.4% compared to free DOX.
Click to Show/Hide
|
||||
| In Vivo Model | SKBR-3 cells female BALB/c mice xenograft model. | ||||
| In Vitro Model | Breast adenocarcinoma | SK-BR-3 cell | CVCL_0033 | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
140 nM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | CCK8 assay | ||||
| MOA of PDC |
Tumor-targeting peptide-drug conjugates (PDCs) have become a focus of research in recent years. However, due to the instability of peptides and their short in vivo effective half-life, they have limited clinical application. Herein, we propose a new DOX PDC based on a homodimer HER-2-targeting peptide and acid-sensitive hydrazone bond, which could enhance the anti-tumor effect of DOX and reduce systemic toxicities. The PDC could accurately deliver DOX into HER2-positive SKBR-3 cells, with it showing 2.9 times higher cellular uptake than free DOX and enhanced cytotoxicity with respect to IC50of 140 nM (vs. 410 nM for free DOX). In vitro assays showed that the PDC had high cellular internalization efficiency and cytotoxicity. In vivo anti-tumor experiments indicated that the PDC could significantly inhibit the growth of HER2-positive breast cancer xenografts in mice and reduce the side effects of DOX. In summary, we constructed a novel PDC molecule targeting HER2-positive tumors, which may overcome some deficiencies of DOX in breast cancer therapy.
Click to Show/Hide
|
||||
| Description |
Tumor-targeting peptide-drug conjugates (PDCs) have become a focus of research in recent years. However, due to the instability of peptides and their short in vivo effective half-life, they have limited clinical application. Herein, we propose a new DOX PDC based on a homodimer HER-2-targeting peptide and acid-sensitive hydrazone bond, which could enhance the anti-tumor effect of DOX and reduce systemic toxicities. The PDC could accurately deliver DOX into HER2-positive SKBR-3 cells, with it showing 2.9 times higher cellular uptake than free DOX and enhanced cytotoxicity with respect to IC50of 140 nM (vs. 410 nM for free DOX). In vitro assays showed that the PDC had high cellular internalization efficiency and cytotoxicity. In vivo anti-tumor experiments indicated that the PDC could significantly inhibit the growth of HER2-positive breast cancer xenografts in mice and reduce the side effects of DOX. In summary, we constructed a novel PDC molecule targeting HER2-positive tumors, which may overcome some deficiencies of DOX in breast cancer therapy.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | SK-BR-3 cell | CVCL_0033 | ||
References
