Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00319
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
PTX-SM-TAR
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
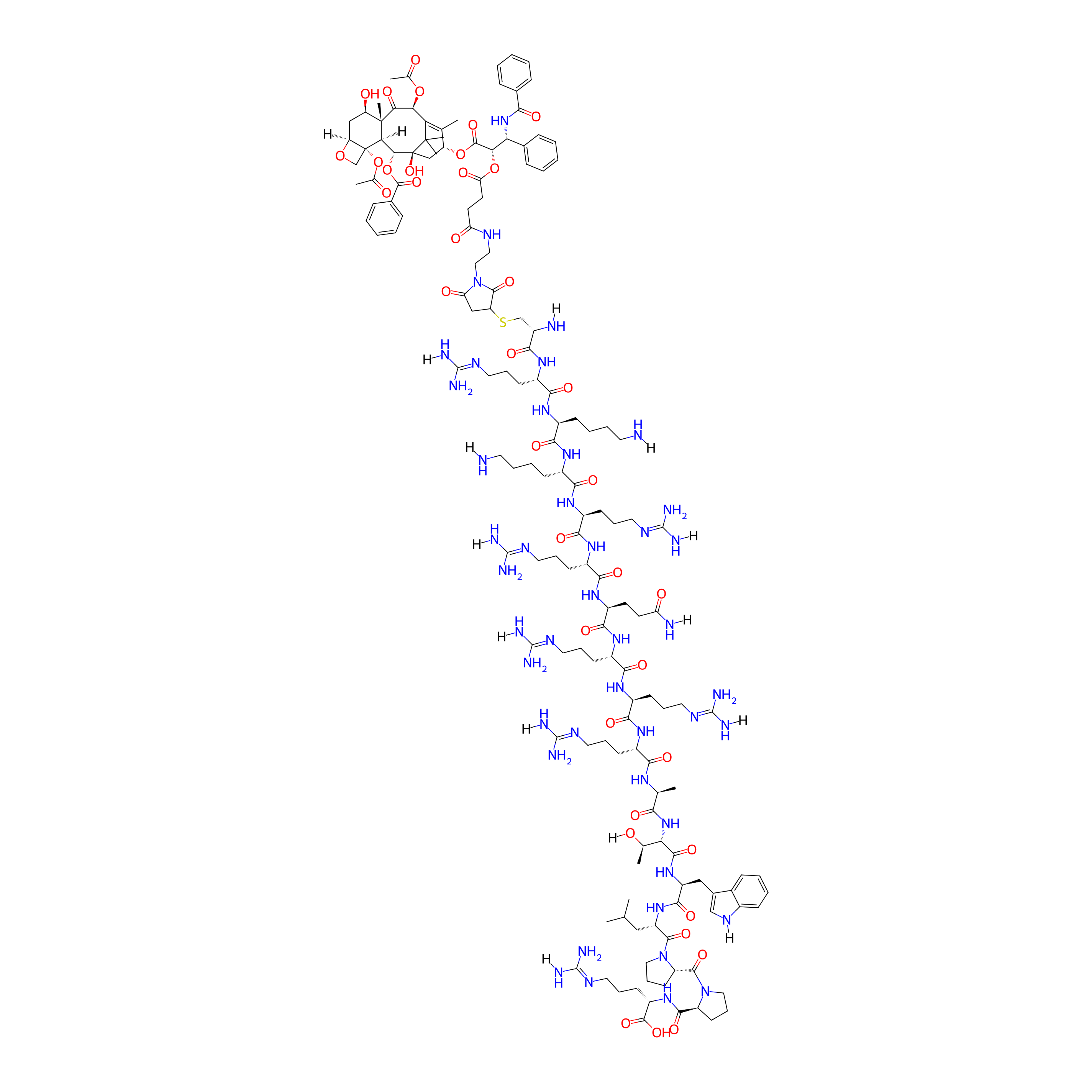
|
|||||
| Peptide Name |
TAR
|
Peptide Info | ||||
| Receptor Name |
Neuropilin-1 (NRP1)
|
Receptor Info | ||||
| Drug Name |
Paclitaxel
|
Drug Info | ||||
| Therapeutic Target |
Microtubule (MT)
|
Target Info | ||||
| Linker Name |
SA-MAL
|
Linker Info | ||||
| Formula |
C153H231N45O38S
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 3340.875 | ||||
| Lipid-water partition coefficient (xlogp) | -9.7057 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 39 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 48 | |||||
| Rotatable Bond Count (rotbonds) | 99 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
43.24%
|
|||
| Administration Time | 16 d | ||||
| Administration Dosage | 10.6 mg/kg | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is an extremely aggressive subtype associated with a poor prognosis. At present, the treatment for TNBC mainly relies on surgery and traditional chemotherapy. As a key component in the standard treatment of TNBC, paclitaxel (PTX) effectively inhibits the growth and proliferation of tumor cells. However, the application of PTX in clinical treatment is limited due to its inherent hydrophobicity, weak penetrability, nonspecific accumulation, and side effects. To counter these problems, we constructed a novel PTX conjugate based on the peptide-drug conjugates (PDCs) strategy. In this PTX conjugate, a novel fused peptide TAR consisting of a tumor-targeting peptide, A7R, and a cell-penetrating peptide, TAT, is used to modify PTX. After modification, this conjugate is named PTX-SM-TAR, which is expected to improve the specificity and penetrability of PTX at the tumor site. Depending on hydrophilic TAR peptide and hydrophobic PTX, PTX-SM-TAR can self-assemble into nanoparticles and improve the water solubility of PTX. In terms of linkage, the acid- and esterase-sensitive ester bond was used as the linking bond, with which PTX-SM-TAR NPs could remain stable in the physiological environment, whereas PTX-SM-TAR NPs could be broken and PTX be released at the tumor site. A cell uptake assay showed that PTX-SM-TAR NPs were receptor-targeting and could mediate endocytosis by binding to NRP-1. The vascular barrier, transcellular migration, and tumor spheroids experiments showed that PTX-SM-TAR NPs exhibit great transvascular transport and tumor penetration ability. In vivo experiments, PTX-SM-TAR NPs showed higher antitumor effects than PTX. As a result, PTX-SM-TAR NPs may overcome the shortcomings of PTX and present a new transcytosable and targeted delivery system for PTX in TNBC treatment.
Click to Show/Hide
|
||||
| Description |
The mice TNBC 4T1-mCherry-Luc model was constructed by subcutaneous inoculation to study the anti-tumor effect of PTX-SM-TAR NPs in vivo. 4T1-mCherry-Luc cells reacted with D-luciferin potassium salt to produce bioluminescence, which was determined by an IVIS spectrum imaging system. The intensity of the fluorescence signal is related to tumor size, hence tumor growth in mice can be monitored in real-time. As shown in Figure 9a, the tumor growth rate of the PTX-SM-TAR NPs group was lower than that of the NS, TAR, and PTX groups. At the end of the experiment, the tumor tissue was weighed, and the results were consistent with the trend of fluorescence intensity. The tumor inhibition rate was 43.24% in the PTX-SM-TAR NPs, 28.47% in the PTX, and 7.81% in the TAR. The tumor inhibitory effect of the PTX-SM-TAR NPs group was stronger than that of the PTX group, and the difference was significant.
Click to Show/Hide
|
||||
| In Vivo Model | Female BALB/c mice 4T1-mCherry-Luc cells xenograft model. | ||||
| In Vitro Model | Malignant neoplasms of the mouse mammary gland | 4T1-mCherry-Luc cell | CVCL_C8UZ | ||
References
