Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00349
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
TH1902
|
|||||
| PDC Status |
Phase 3
|
|||||
| Indication |
In total 4 Indication(s)
|
|||||
| Structure |
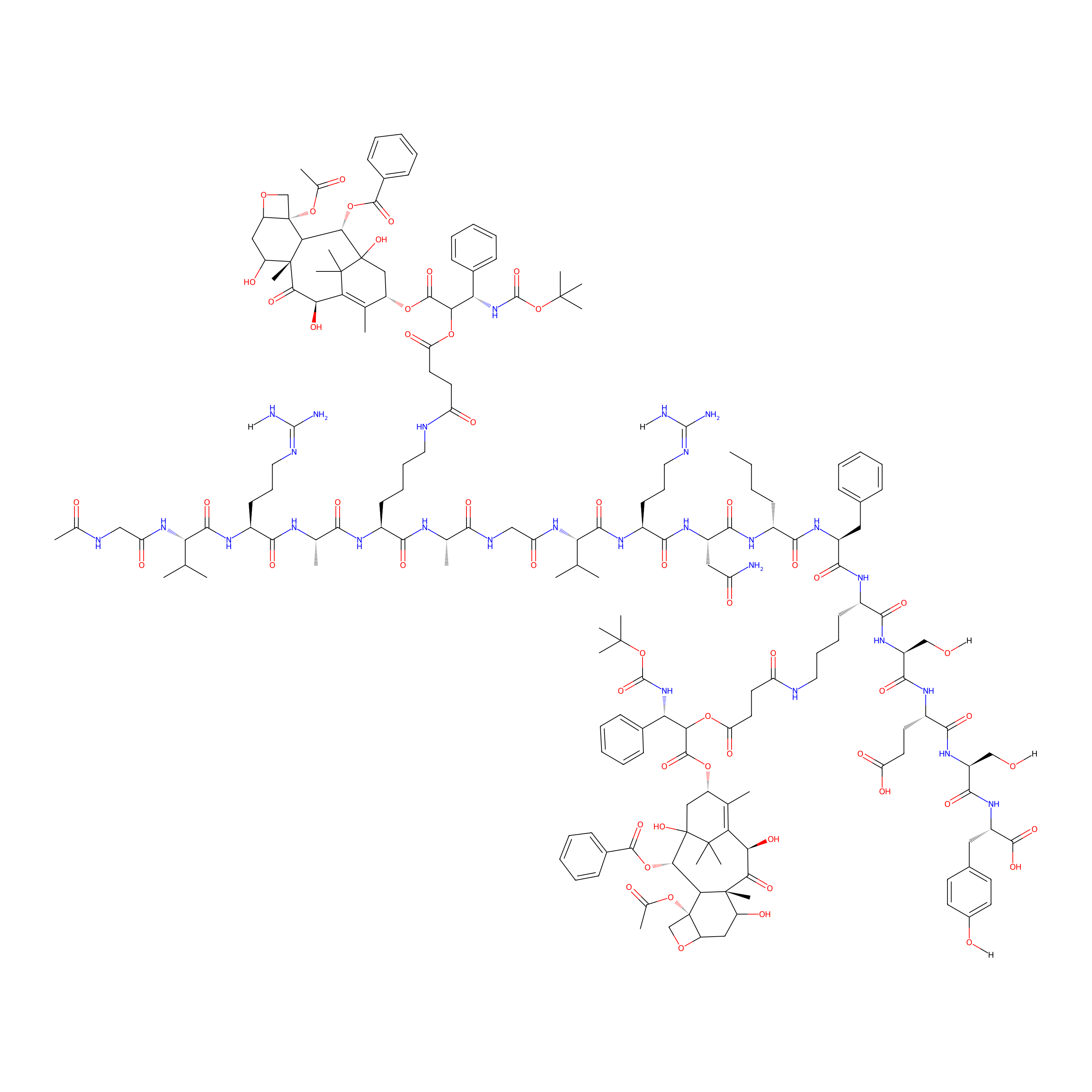
|
|||||
| Peptide Name |
TH19P01
|
Peptide Info | ||||
| Receptor Name |
Sortilin (SORT1)
|
Receptor Info | ||||
| Drug Name |
Docetaxel
|
Drug Info | ||||
| Therapeutic Target |
Microtubule (MT)
|
Target Info | ||||
| Linker Name |
Succinic Acid
|
Linker Info | ||||
| Formula |
C179H248N28O57
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 3704.092 | ||||
| Lipid-water partition coefficient (xlogp) | -2.0581 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 37 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 57 | |||||
| Rotatable Bond Count (rotbonds) | 95 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
30%
|
|||
| Administration Time | 20 days | ||||
| Administration Dosage | 8.75 mg/kg | ||||
| MOA of PDC |
Sortilin (SORT1) is a receptor protein which cycles from the cell surface through intracellular membrane bodies. It binds several circulating proteins and peptides, including progranulin and neurotensin, prior to their rapid intracellular internalization. It is also involved in their intracellular trafficking in endosomal vesicles from the plasma membrane to lysosomes through the endocytic pathway. Deregulation in SORT1 functions has been implicated in cardiovascular disease, Alzheimers disease, and diabetes, whereas its upregulation has been documented in several forms of cancer. Recently, TH1902, a peptide-drug conjugate (PDC) to which two docetaxel molecules were linked to TH19P01 peptide, was generated. TH1902 was demonstrated to recognize and exploit SORT1 functions, and to efficiently inhibit in vitro cell proliferation, and in vivo growth of xenografts from gynecological cancers including triple-negative breast cancer (TNBC)-derived MDA-MB-231 cells, ovarian cancer-derived ES2 and SKOV3 cells, as well as endometrial cancer-derived AN3-CA cells. Interestingly, TH1902 was found to reduce in vitro vasculogenic mimicry (VM) processes in breast and ovarian cancer cells. While some molecular aspects revealing the inter-relationship existence between CSC and VM have been addressed in breast cancer, gastrointestinal cancer, and melanoma, the specific targeting of the CSC subpopulation by TH1902 remains unexplored.
Click to Show/Hide
|
||||
| Description |
In vivo assessment in tumor-bearing animal models classically complements cell cultures data for comparing cancer treatment efficacies. For this reason, hTNBCSC and hOvCSC xenografts were implanted subcutaneously into immunodeficient mice as described in the Methods section with only 1000 cells given their highly tumorigenic nature. Three days later, the animals began receiving weekly IV bolus administration of either vehicle, docetaxel, or TH1902. Docetaxel was administered at a dose (15 mg/kg/week; for 3 cycles) in accordance with the estimated maximal tolerated dose (MTD) for mice, as well as at 1/4 of the MTD (3.75 mg/kg/week). TH1902 was administered at doses (35 and 8.75 mg/kg/week) which contained quantities of docetaxel equivalent to those in the two administrations of free docetaxel. From the size of the hTNBCSC and hOvCSC tumors, it is apparent that docetaxel had little impact on xenografts growth when administered neither at its MTD, for both hTNBCSC and hOvCSC, nor at 1/4 of this dosage as reflected by the growth curves. TH1902, when administered at a dosage equivalent to docetaxel at its MTD, provided greater tumor growth inhibition than did docetaxel for both xenograft models. Furthermore, higher dosage of administered TH1902 (up to 1.5 equivalent of docetaxel MTD) did not generate significant differences in terms of hOvCSC tumor inhibition without affecting mice body weights suggesting that TH1902, even at higher doses, is better tolerated compared to docetaxel. Then, in order to statistically compare the effects of docetaxel and TH1902 on tumor growth, the tumor sizes measured at the vehicle group endpoint were compared and statistically significant differences between the tumor sizes in vehicle-treated animals found in TH1902-treated animals for hTNBCSC and hOvCSC. Mouse body weight was used as an indicator of the morbidity associated with administration of docetaxel or TH1902. Administration of docetaxel at its MTD provoked a weight loss that approached 10% after the treatments, which is often observed in xenograft models with administration of docetaxel at this level. The body weights of animals treated with an equivalent quantity or a 1.5-fold equivalent of TH1902 were maintained at a roughly constant level throughout the experiment. The animals treated with vehicle recorded a slight weight gain (~5%) over this period while animals treated with the lower dosages of docetaxel or TH1902 were similar to the animals treated with vehicle. The body weight data indicates that TH1902 appears to be better tolerated than the equivalent quantity of free docetaxel, in addition to the fact that TH1902 is more efficacious than docetaxel when administered in vivo in the murine models of CSC tested.
Click to Show/Hide
|
||||
| In Vivo Model | Nude mice hTNBCSC cells xenograft tumor model. | ||||
| In Vitro Model | Triple-negative breast cancer | Human triple-negative breast cancer stem cell (hTNBCSC) | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
75%
|
|||
| Administration Time | 20 days | ||||
| Administration Dosage | 35 mg/kg | ||||
| MOA of PDC |
Sortilin (SORT1) is a receptor protein which cycles from the cell surface through intracellular membrane bodies. It binds several circulating proteins and peptides, including progranulin and neurotensin, prior to their rapid intracellular internalization. It is also involved in their intracellular trafficking in endosomal vesicles from the plasma membrane to lysosomes through the endocytic pathway. Deregulation in SORT1 functions has been implicated in cardiovascular disease, Alzheimers disease, and diabetes, whereas its upregulation has been documented in several forms of cancer. Recently, TH1902, a peptide-drug conjugate (PDC) to which two docetaxel molecules were linked to TH19P01 peptide, was generated. TH1902 was demonstrated to recognize and exploit SORT1 functions, and to efficiently inhibit in vitro cell proliferation, and in vivo growth of xenografts from gynecological cancers including triple-negative breast cancer (TNBC)-derived MDA-MB-231 cells, ovarian cancer-derived ES2 and SKOV3 cells, as well as endometrial cancer-derived AN3-CA cells. Interestingly, TH1902 was found to reduce in vitro vasculogenic mimicry (VM) processes in breast and ovarian cancer cells. While some molecular aspects revealing the inter-relationship existence between CSC and VM have been addressed in breast cancer, gastrointestinal cancer, and melanoma, the specific targeting of the CSC subpopulation by TH1902 remains unexplored.
Click to Show/Hide
|
||||
| Description |
In vivo assessment in tumor-bearing animal models classically complements cell cultures data for comparing cancer treatment efficacies. For this reason, hTNBCSC and hOvCSC xenografts were implanted subcutaneously into immunodeficient mice as described in the Methods section with only 1000 cells given their highly tumorigenic nature. Three days later, the animals began receiving weekly IV bolus administration of either vehicle, docetaxel, or TH1902. Docetaxel was administered at a dose (15 mg/kg/week; for 3 cycles) in accordance with the estimated maximal tolerated dose (MTD) for mice, as well as at 1/4 of the MTD (3.75 mg/kg/week). TH1902 was administered at doses (35 and 8.75 mg/kg/week) which contained quantities of docetaxel equivalent to those in the two administrations of free docetaxel. From the size of the hTNBCSC and hOvCSC tumors, it is apparent that docetaxel had little impact on xenografts growth when administered neither at its MTD, for both hTNBCSC and hOvCSC, nor at 1/4 of this dosage as reflected by the growth curves. TH1902, when administered at a dosage equivalent to docetaxel at its MTD, provided greater tumor growth inhibition than did docetaxel for both xenograft models. Furthermore, higher dosage of administered TH1902 (up to 1.5 equivalent of docetaxel MTD) did not generate significant differences in terms of hOvCSC tumor inhibition without affecting mice body weights suggesting that TH1902, even at higher doses, is better tolerated compared to docetaxel. Then, in order to statistically compare the effects of docetaxel and TH1902 on tumor growth, the tumor sizes measured at the vehicle group endpoint were compared and statistically significant differences between the tumor sizes in vehicle-treated animals found in TH1902-treated animals for hTNBCSC and hOvCSC. Mouse body weight was used as an indicator of the morbidity associated with administration of docetaxel or TH1902. Administration of docetaxel at its MTD provoked a weight loss that approached 10% after the treatments, which is often observed in xenograft models with administration of docetaxel at this level. The body weights of animals treated with an equivalent quantity or a 1.5-fold equivalent of TH1902 were maintained at a roughly constant level throughout the experiment. The animals treated with vehicle recorded a slight weight gain (~5%) over this period while animals treated with the lower dosages of docetaxel or TH1902 were similar to the animals treated with vehicle. The body weight data indicates that TH1902 appears to be better tolerated than the equivalent quantity of free docetaxel, in addition to the fact that TH1902 is more efficacious than docetaxel when administered in vivo in the murine models of CSC tested.
Click to Show/Hide
|
||||
| In Vivo Model | Nude mice hTNBCSC cells xenograft tumor model. | ||||
| In Vitro Model | Triple-negative breast cancer | Human triple-negative breast cancer stem cell (hTNBCSC) | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
90%
|
|||
| Administration Time | 12 days | ||||
| Administration Dosage | 35 mg/kg/wk | ||||
| MOA of PDC |
The efficacy of current chemotherapeutic treatments against most solid tumors is limited by their systemic toxicity, which is partly associated with the cytotoxic properties of agents such as docetaxel or doxorubicin. To avoid or minimize adverse effects from chemotherapeutic molecules, a promising targeted approach is through peptide-drug conjugates (PDCs) that link anticancer molecules to peptides designed to interact with receptors highly expressed on cancer cells, and which can mediate the molecules rapid internalization within those cells. One such receptor is sortilin (SORT1), also known as neurotensin receptor3, a membranebound receptor that belongs to the VPS10P family of receptors. TH19P01 peptide was recently designed to target and exploit SORT1s ligand internalization function. Studies have confirmed that both TH1902 (a docetaxel-TH19P01 conjugate) and TH1904 (a doxorubicin-TH19P01 conjugate) require a SORT1-dependent mechanism of action to exert anticancer activities. In recent preclinical studies performed in immunocompromised animal models, which are unable to produce mature T-cells, TH1902 was effective against several human SORT1-positive xenograft models including triple-negative breast cancer (TNBC), ovarian cancer, and endometrial cancer.
Click to Show/Hide
|
||||
| Description |
B16-F10 melanoma syngeneic tumors were generated, with tumor sizes monitored as described in the Methods section. Tumors in xenograft-bearing, vehicle-treated mice grew at an exponential rate. Partial inhibition of tumor growth was observed after IV administration of 15 mg/kg/wk docetaxel, whereas treatment with a docetaxel-equivalent quantity of TH1902 (35 mg/kg/wk) induced tumor regression after two treatments over the period measured. Due to rapid tumor growth, only two administrations of the test articles could be performed on a weekly schedule. B16-F10 melanoma tumors from the mice treated with either vehicle, docetaxel, or TH1902 were then excised, fixed in formalin, and immunohistochemically examined.
Click to Show/Hide
|
||||
| In Vivo Model | SORT1-positive B16-F10 cells female CD1 nude mice xenograft tumor models. | ||||
| In Vitro Model | Mouse melanoma | SORT1-positive B16-F10 cell | CVCL_0159 | ||
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
45.00%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 8.75 mg/kg | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
In the ES-2 study, mice treated with low and high doses of TH1902 significantly inhibited the growth of tumors by, respectively, 45% and 87%, whereas both docetaxel groups, at equivalent docetaxel content, produced little effect (Figure 6A and Table 1). Low doses of docetaxel and low and high doses of TH1902 were well-tolerated with slight weight gain, whereas three weekly cycles of treatments with high doses of docetaxel (the MTD for this agent in mice) produced a small weight loss when compared to initial mice weights
Click to Show/Hide
|
||||
| In Vivo Model | Female CD-1 nude mice xenograft model. | ||||
| In Vitro Model | Ewing sarcoma | ES2 cell | CVCL_AX39 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
69.00%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 8.75 mg/kg | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
In the SKOV3 study, mice treated with low and high doses of TH1902 showed significant inhibitions of tumor growth where only high dose of docetaxel produced this effect (Figure 6B). When compared to vehicle endpoint (Day 28), low dose of TH1902 produced similar tumor growth inhibitions when compared to high dose of docetaxel (69% vs. 84%, respectively) while high dose of TH1902 induced stronger inhibition with slight regression of tumors (Table 1). Moreover, mice treated with high dose of TH1902 showed prolonged inhibitory effect (up to Day 46) where docetaxel-treated mice could not sustain this effect, which is marked by tumor regrowth (Figure 6B).
Click to Show/Hide
|
||||
| In Vivo Model | Female CD-1 nude mice xenograft model. | ||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Endometrial cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
74.00%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 8.75 mg/kg | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
For mice bearing endometrial tumor xenografts, a similar dosage regimen to that for ovarian tumor xenografts was used as described previously for both docetaxel and TH1902. The growth of AN3-CA tumors was inhibited by both dosages of TH1902, whereas only a high dose of docetaxel could produce this effect. When compared to vehicle endpoint, low dose of TH1902 significantly inhibited the growth of AN3-CA tumors by 74%, whereas both high doses of docetaxel and TH1902 induced tumor regressions. Interestingly, a large portion of tumors (5 out of 6) within the high-dose TH1902 group were unmeasurable or remained in regression for a prolonged period (up to Day 54), whereas tumors in the equivalent docetaxel dose were unresponsive and regrew before the end of treatments. The two high doses were diminished by half for the final two treatments due to weight loss in the animals receiving high-dose docetaxel. Rapid weight loss was associated with administration of high dosage docetaxel, but this was reversed when the dosage was halved. In contrast, only mild weight loss was observed in animals administered with high-dosage TH1902.
Click to Show/Hide
|
||||
| In Vivo Model | Female CD-1 nude mice xenograft model. | ||||
| In Vitro Model | Endometrial adenocarcinoma | AN3-CA cell | CVCL_0028 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
84.00%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 35 mg/kg | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
In the SKOV3 study, mice treated with low and high doses of TH1902 showed significant inhibitions of tumor growth where only high dose of docetaxel produced this effect (Figure 6B). When compared to vehicle endpoint (Day 28), low dose of TH1902 produced similar tumor growth inhibitions when compared to high dose of docetaxel (69% vs. 84%, respectively) while high dose of TH1902 induced stronger inhibition with slight regression of tumors (Table 1). Moreover, mice treated with high dose of TH1902 showed prolonged inhibitory effect (up to Day 46) where docetaxel-treated mice could not sustain this effect, which is marked by tumor regrowth (Figure 6B).
Click to Show/Hide
|
||||
| In Vivo Model | Female CD-1 nude mice xenograft model. | ||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
87.00%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 35 mg/kg | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
In the ES-2 study, mice treated with low and high doses of TH1902 significantly inhibited the growth of tumors by, respectively, 45% and 87%, whereas both docetaxel groups, at equivalent docetaxel content, produced little effect (Figure 6A and Table 1). Low doses of docetaxel and low and high doses of TH1902 were well-tolerated with slight weight gain, whereas three weekly cycles of treatments with high doses of docetaxel (the MTD for this agent in mice) produced a small weight loss when compared to initial mice weights
Click to Show/Hide
|
||||
| In Vivo Model | Female CD-1 nude mice xenograft model. | ||||
| In Vitro Model | Ewing sarcoma | ES2 cell | CVCL_AX39 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Endometrial cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
100.00%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 35 mg/kg | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
For mice bearing endometrial tumor xenografts, a similar dosage regimen to that for ovarian tumor xenografts was used as described previously for both docetaxel and TH1902. The growth of AN3-CA tumors was inhibited by both dosages of TH1902, whereas only a high dose of docetaxel could produce this effect. When compared to vehicle endpoint, low dose of TH1902 significantly inhibited the growth of AN3-CA tumors by 74%, whereas both high doses of docetaxel and TH1902 induced tumor regressions. Interestingly, a large portion of tumors (5 out of 6) within the high-dose TH1902 group were unmeasurable or remained in regression for a prolonged period (up to Day 54), whereas tumors in the equivalent docetaxel dose were unresponsive and regrew before the end of treatments. The two high doses were diminished by half for the final two treatments due to weight loss in the animals receiving high-dose docetaxel. Rapid weight loss was associated with administration of high dosage docetaxel, but this was reversed when the dosage was halved. In contrast, only mild weight loss was observed in animals administered with high-dosage TH1902.
Click to Show/Hide
|
||||
| In Vivo Model | Female CD-1 nude mice xenograft model. | ||||
| In Vitro Model | Endometrial adenocarcinoma | AN3-CA cell | CVCL_0028 | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Apotosis rate |
9.00%
|
|||
| Administration Time | 5 h | ||||
| Administration Dosage | 5 μM | ||||
| Evaluation Method | Flow cytometry | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The impact of docetaxel and TH1902 on MDA-MB-231 cellular morphology was evaluated and visualized by light microscopy. A drastic change in cell shape was observed in both docetaxel- and TH1902-treated cells when compared to control cells. As already reported for docetaxel-treated human renal clear cell carcinoma, MDA-MB-231 cells treated with docetaxel appeared more contracted than control cells. Interestingly, TH1902-treated cell morphology appeared even smaller, less flattened, and presented greater inter-cell spacing in comparison to those treated with free docetaxel. This observation confirms the enhanced cytotoxic effect of TH1902 when compared to docetaxel. Docetaxel is further believed to exert its cytotoxic effects through both cell cycle regulation and apoptosis induction. MDA-MB-231 cells were therefore treated for 5 or 24 hours with various concentrations of either docetaxel or TH1902, followed by AnnexinV/PI staining. TH1902 increased MDA-MB-231 cell apoptosis in a dose-dependent manner when compared to docetaxel treatment. Less than 1% apoptotic cells were measured when cells were treated with 50 μM TH19P01 for 24 hours (data not shown). Overall, this suggests that, even within such a short time frame, receptor-mediated events account for the increased effects of TH1902. Finally, to confirm the role of SORT1 in TH1902 internalization and induced apoptosis, pharmacological inhibition of its apoptotic activity was measured after incubating MDA-MB-231 breast cancer cells with TH1902 in the presence or absence of the free unlabeled TH19P01 peptide or of one of the two different SORT1 ligands neurotensin or progranulin. The apoptosis assay showed that excess TH19P01 peptide, neurotensin or progranulin competitively reduced TH1902-induced cell death by 68%, 53% and 87%, respectively. This confirms the involvement of SORT1 in the internalization process of TH1902 leading to cell death in a TNBC cell model.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Apotosis rate |
19.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 5 μM | ||||
| Evaluation Method | Flow cytometry | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The impact of docetaxel and TH1902 on MDA-MB-231 cellular morphology was evaluated and visualized by light microscopy. A drastic change in cell shape was observed in both docetaxel- and TH1902-treated cells when compared to control cells. As already reported for docetaxel-treated human renal clear cell carcinoma, MDA-MB-231 cells treated with docetaxel appeared more contracted than control cells. Interestingly, TH1902-treated cell morphology appeared even smaller, less flattened, and presented greater inter-cell spacing in comparison to those treated with free docetaxel. This observation confirms the enhanced cytotoxic effect of TH1902 when compared to docetaxel. Docetaxel is further believed to exert its cytotoxic effects through both cell cycle regulation and apoptosis induction. MDA-MB-231 cells were therefore treated for 5 or 24 hours with various concentrations of either docetaxel or TH1902, followed by AnnexinV/PI staining. TH1902 increased MDA-MB-231 cell apoptosis in a dose-dependent manner when compared to docetaxel treatment. Less than 1% apoptotic cells were measured when cells were treated with 50 μM TH19P01 for 24 hours (data not shown). Overall, this suggests that, even within such a short time frame, receptor-mediated events account for the increased effects of TH1902. Finally, to confirm the role of SORT1 in TH1902 internalization and induced apoptosis, pharmacological inhibition of its apoptotic activity was measured after incubating MDA-MB-231 breast cancer cells with TH1902 in the presence or absence of the free unlabeled TH19P01 peptide or of one of the two different SORT1 ligands neurotensin or progranulin. The apoptosis assay showed that excess TH19P01 peptide, neurotensin or progranulin competitively reduced TH1902-induced cell death by 68%, 53% and 87%, respectively. This confirms the involvement of SORT1 in the internalization process of TH1902 leading to cell death in a TNBC cell model.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Apotosis rate |
22.00%
|
|||
| Administration Time | 5 h | ||||
| Administration Dosage | 10 μM | ||||
| Evaluation Method | Flow cytometry | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The impact of docetaxel and TH1902 on MDA-MB-231 cellular morphology was evaluated and visualized by light microscopy. A drastic change in cell shape was observed in both docetaxel- and TH1902-treated cells when compared to control cells. As already reported for docetaxel-treated human renal clear cell carcinoma, MDA-MB-231 cells treated with docetaxel appeared more contracted than control cells. Interestingly, TH1902-treated cell morphology appeared even smaller, less flattened, and presented greater inter-cell spacing in comparison to those treated with free docetaxel. This observation confirms the enhanced cytotoxic effect of TH1902 when compared to docetaxel. Docetaxel is further believed to exert its cytotoxic effects through both cell cycle regulation and apoptosis induction. MDA-MB-231 cells were therefore treated for 5 or 24 hours with various concentrations of either docetaxel or TH1902, followed by AnnexinV/PI staining. TH1902 increased MDA-MB-231 cell apoptosis in a dose-dependent manner when compared to docetaxel treatment. Less than 1% apoptotic cells were measured when cells were treated with 50 μM TH19P01 for 24 hours (data not shown). Overall, this suggests that, even within such a short time frame, receptor-mediated events account for the increased effects of TH1902. Finally, to confirm the role of SORT1 in TH1902 internalization and induced apoptosis, pharmacological inhibition of its apoptotic activity was measured after incubating MDA-MB-231 breast cancer cells with TH1902 in the presence or absence of the free unlabeled TH19P01 peptide or of one of the two different SORT1 ligands neurotensin or progranulin. The apoptosis assay showed that excess TH19P01 peptide, neurotensin or progranulin competitively reduced TH1902-induced cell death by 68%, 53% and 87%, respectively. This confirms the involvement of SORT1 in the internalization process of TH1902 leading to cell death in a TNBC cell model.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Apotosis rate |
35.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 10 μM | ||||
| Evaluation Method | Flow cytometry | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The impact of docetaxel and TH1902 on MDA-MB-231 cellular morphology was evaluated and visualized by light microscopy. A drastic change in cell shape was observed in both docetaxel- and TH1902-treated cells when compared to control cells. As already reported for docetaxel-treated human renal clear cell carcinoma, MDA-MB-231 cells treated with docetaxel appeared more contracted than control cells. Interestingly, TH1902-treated cell morphology appeared even smaller, less flattened, and presented greater inter-cell spacing in comparison to those treated with free docetaxel. This observation confirms the enhanced cytotoxic effect of TH1902 when compared to docetaxel. Docetaxel is further believed to exert its cytotoxic effects through both cell cycle regulation and apoptosis induction. MDA-MB-231 cells were therefore treated for 5 or 24 hours with various concentrations of either docetaxel or TH1902, followed by AnnexinV/PI staining. TH1902 increased MDA-MB-231 cell apoptosis in a dose-dependent manner when compared to docetaxel treatment. Less than 1% apoptotic cells were measured when cells were treated with 50 μM TH19P01 for 24 hours (data not shown). Overall, this suggests that, even within such a short time frame, receptor-mediated events account for the increased effects of TH1902. Finally, to confirm the role of SORT1 in TH1902 internalization and induced apoptosis, pharmacological inhibition of its apoptotic activity was measured after incubating MDA-MB-231 breast cancer cells with TH1902 in the presence or absence of the free unlabeled TH19P01 peptide or of one of the two different SORT1 ligands neurotensin or progranulin. The apoptosis assay showed that excess TH19P01 peptide, neurotensin or progranulin competitively reduced TH1902-induced cell death by 68%, 53% and 87%, respectively. This confirms the involvement of SORT1 in the internalization process of TH1902 leading to cell death in a TNBC cell model.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Apotosis rate |
45.00%
|
|||
| Administration Time | 5 h | ||||
| Administration Dosage | 2 μM | ||||
| Evaluation Method | Annexin X/PI staining combined with flow cytometry | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
The results show that TH1902 induces more efficiently apoptosis in both cell lines when compared to docetaxel especially in SKOV3 cells. Overall, this suggests that receptor-mediated events appear to account for the increased effects of TH1902 within such a short time frame. To confirm the implication of SORT1 in TH1902 internalization and apoptosis induction, SORT1 was transiently silenced in ES-2 and SKOV3 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Apotosis rate |
47.00%
|
|||
| Administration Time | 5 h | ||||
| Administration Dosage | 2 μM | ||||
| Evaluation Method | Annexin X/PI staining combined with flow cytometry | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
The results show that TH1902 induces more efficiently apoptosis in both cell lines when compared to docetaxel especially in SKOV3 cells. Overall, this suggests that receptor-mediated events appear to account for the increased effects of TH1902 within such a short time frame. To confirm the implication of SORT1 in TH1902 internalization and apoptosis induction, SORT1 was transiently silenced in ES-2 and SKOV3 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian clear cell adenocarcinoma | ES-2 cell | CVCL_3509 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
0.24 ± 0.28 nM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
To determine whether docetaxel or TH1902 inhibited TNBC cell proliferation, MTT assays were performed in MDA-MB-231 and HCC-70 cells exposed to various concentrations of TH19P01, docetaxel or TH1902. Inhibition of MDA-MB-231 cell proliferation was effectively triggered by both docetaxel and TH1902 compounds in a concentration-dependent manner whereas TH19P01 had no effect at the concentrations tested. The IC50 value of TH1902 was found to be comparable to that of docetaxel at low nM concentrations in both MDA-MB-231 and HCC-70 cells, which supports the rationale that conjugated docetaxel can indeed be released from TH1902 once internalized and exert its antiproliferative effect inside the targeted cancer cells. The effect of TH1902 on MBA-MB-231 cell-cycle arrest was also tested. The results show that more than 70% of treated cells were arrested in the G2/M phase of the cell cycle in comparison to vehicle-treated control cells where ~14% of the cells remained in the G2/M phase. While TH19P01 appears to be an inert peptide with no in vitro antiproliferative effects, these data confirm both the growth inhibitory effect of TH1902 and, more importantly, that the docetaxel anticancer potency was unaffected by its conjugation to the TH19P01 peptide.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 8 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
0.29 ± 0.24 nM
|
|||
| Administration Time | 72 h | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
Cells were thus exposed to various concentrations of TH1902 or docetaxel, and the effects on cell proliferation were measured using the MTT detection assay. It is apparent that both reagents exerted a cytotoxic effect with IC50 values of TH1902 comparable to that of docetaxel at low nM concentrations in the four cell lines tested. This supports the rationale that conjugated docetaxel can be released from TH1902 and exert its anti-proliferative effect inside the targeted cancer cells. Parallel experiment showed that cell proliferation was unaffected by the free peptide (TH19P01) at concentrations up to 1000 nM.
Click to Show/Hide
|
||||
| In Vitro Model | Endometrial adenocarcinoma | AN3-CA cell | CVCL_0028 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.38 nM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The cancer phenotype is commonly associated with aberrant glycosylation patterns. One glycan that is directly linked to cancer is the Thomsen-Friedenreich antigen (TF or CD176). It is a disaccharide composed of a galactose β1-3 N-acetylgalactosamine, O-linked to a glycoprotein through serine or threonine residues and commonly written as Galβ1-3GalNAc--O-Ser/Thr. The TF is therapeutically attractive due to its cryptic nature in normal cells and exposure in embryonic and cancer cells. The expression of the TF has been demonstrated in 90% of primary human carcinomas, including in the lung, the breast, and the pancreas. Additionally, cancer initiating cells or cancer stem cells in the lung, liver, and colon express the TF. The peptide sequence HGRFILPWWYAFSPS (TF-peptide) is known to bind tightly to the TF (Kd = 1.2 M) and has been demonstrated to inhibit processes directly involved in TF accessibility.
Click to Show/Hide
|
||||
| Description |
SORT1-positive SK-MEL-28 and B16-F10 melanoma cells were selected for testing the anti-proliferative effect of docetaxel and TH1902. When TH1902 biological effects were monitored, the half-maximal inhibitory concentration (IC50) of TH1902 was similar to that of docetaxel, averaging 0.38 vs. 0.39 nM, respectively, in human SK-MEL-28 cells, and 2.57 vs. 1.72 nM in murine B16-F10 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | SORT1-positive SKMEL28 cell | CVCL_0526 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
0.55 ± 0.21 nM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
Cells were thus exposed to various concentrations of TH1902 or docetaxel, and the effects on cell proliferation were measured using the MTT detection assay. It is apparent that both reagents exerted a cytotoxic effect with IC50 values of TH1902 comparable to that of docetaxel at low nM concentrations in the four cell lines tested. This supports the rationale that conjugated docetaxel can be released from TH1902 and exert its anti-proliferative effect inside the targeted cancer cells. Parallel experiment showed that cell proliferation was unaffected by the free peptide (TH19P01) at concentrations up to 1000 nM.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
1.00 ±0.55 nM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
To determine whether docetaxel or TH1902 inhibited TNBC cell proliferation, MTT assays were performed in MDA-MB-231 and HCC-70 cells exposed to various concentrations of TH19P01, docetaxel or TH1902. Inhibition of MDA-MB-231 cell proliferation was effectively triggered by both docetaxel and TH1902 compounds in a concentration-dependent manner whereas TH19P01 had no effect at the concentrations tested. The IC50 value of TH1902 was found to be comparable to that of docetaxel at low nM concentrations in both MDA-MB-231 and HCC-70 cells, which supports the rationale that conjugated docetaxel can indeed be released from TH1902 once internalized and exert its antiproliferative effect inside the targeted cancer cells. The effect of TH1902 on MBA-MB-231 cell-cycle arrest was also tested. The results show that more than 70% of treated cells were arrested in the G2/M phase of the cell cycle in comparison to vehicle-treated control cells where ~14% of the cells remained in the G2/M phase. While TH19P01 appears to be an inert peptide with no in vitro antiproliferative effects, these data confirm both the growth inhibitory effect of TH1902 and, more importantly, that the docetaxel anticancer potency was unaffected by its conjugation to the TH19P01 peptide.
Click to Show/Hide
|
||||
| In Vitro Model | Breast ductal carcinoma | HCC70 cell | CVCL_1270 | ||
| Half life period | 1.44 h | ||||
| Experiment 12 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
1.58±0.07 nM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
For mice bearing endometrial tumor xenografts, a similar dosage regimen to that for ovarian tumor xenografts was used as described previously for both docetaxel and TH1902. The growth of AN3-CA tumors was inhibited by both dosages of TH1902, whereas only
|
||||
| In Vitro Model | Ovarian clear cell adenocarcinoma | ES-2 cell | CVCL_3509 | ||
| Experiment 13 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
2.57 nM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
As a payload for PDCs (and ADCs), analogues of the duocarmycins are attractive. Duocarmycin SA and yatakemycin rank among the most potent natural cytotoxins discovered. The cyclopropyl and prodrug seco forms are both naturally occurring and equipotent in most circumstances. Studies of the binding-driven bonding model of their interaction with DNA suggest that their utility will be enhanced when targeted to tumor cells. In fact, SYD985, an ADC that utilizes a peptide linker for a duocarmycin analogue to trastuzumab, has recently been progressed to phase III clinical trial.
Click to Show/Hide
|
||||
| Description |
SORT1-positive SK-MEL-28 and B16-F10 melanoma cells were selected for testing the anti-proliferative effect of docetaxel and TH1902. When TH1902 biological effects were monitored, the half-maximal inhibitory concentration (IC50) of TH1902 was similar to that of docetaxel, averaging 0.38 vs. 0.39 nM, respectively, in human SK-MEL-28 cells, and 2.57 vs. 1.72 nM in murine B16-F10 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Mouse melanoma | SORT1-positive B16-F10 cell | CVCL_0159 | ||
| Experiment 14 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Endometrial cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
3.99 ± 0.96 nM
|
|||
| Administration Time | 72 h | ||||
| MOA of PDC |
TH1902 is a peptide-drug conjugate with a payload of two docetaxel molecules ester-linked to a peptide (TH19P01) designed to recognize SORT1. Studies in breast and ovarian cancer cell lines have shown that TH1902 exploited SORT1s ligand internalization functions and exerted potent antiproliferative and anti-migratory effects. Within the cell, the docetaxel molecules are released from the conjugate and can then affect polymerization of microtubules leading to aberrant mitosis and apoptosis. Intravenous administration of TH1902 to mice bearing xenografts of MDA-MB-231 breast cancer cells demonstrated a marked superiority of TH1902 over free docetaxel in preventing the growth and relapse of subcutaneous xenografts. In a previous study, SORT1 was reported to have a role in vasculogenic mimicry (VM), and TH1902 was shown to inhibit in vitro VM in these cells and in the ES-2 ovarian cancer cell line.
Click to Show/Hide
|
||||
| Description |
Cells were thus exposed to various concentrations of TH1902 or docetaxel, and the effects on cell proliferation were measured using the MTT detection assay. It is apparent that both reagents exerted a cytotoxic effect with IC50 values of TH1902 comparable to that of docetaxel at low nM concentrations in the four cell lines tested. This supports the rationale that conjugated docetaxel can be released from TH1902 and exert its anti-proliferative effect inside the targeted cancer cells. Parallel experiment showed that cell proliferation was unaffected by the free peptide (TH19P01) at concentrations up to 1000 nM.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 15 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Residual tumor burden |
2x106 RLU
|
|||
| Administration Time | 27 days | ||||
| Administration Dosage | 35 mg/kg | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The use of intravenous administration of TH1902 was next investigated in the TNBC-derived MDA-MB-231 xenograft model. Mice subjected to docetaxel treatment were administered with three intravenous injections at 15 mg/kg/wk (MTD), whereas those treated with TH1902 received five injections of an equivalent dose of docetaxel at 35 mg/kg/wk. Similar to what was seen with the intraperitoneal administration protocol, a sustained decrease in tumor size was observed following intravenous administration of TH1902 until day 70, whereas a restart of tumor growth was observed at day 50 for the docetaxel-treated mice. When lower doses, equivalent to the quarter of the MTD, were used of docetaxel (3.75 mg/kg) and TH1902 (8.75 mg/kg), tumor growth as assessed by luminescence intensity was unaffected by intravenous administration of docetaxel, whereas it was significantly inhibited in the TH1902-treated group. This was further confirmed through the measurement of the tumor burden luminescence where TH1902 reduced it significantly in comparison to vehicle- or docetaxel-treated groups. Interestingly, body weight changes remained within endpoint limits in mice on intravenous administration of either docetaxel or TH1902 (data not shown). Similar conclusions were reached on testing another TNBC-derived HCC-70 xenograft model. In fact, administration of TH1902 at 8.75 mg/kg/wk led to a 93% inhibition of HCC-70 tumor growth as compared to 24% for docetaxel alone at an equivalent dose.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 16 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Residual tumor burden |
6x106 RLU
|
|||
| Administration Time | 14 days | ||||
| Administration Dosage | 50mg /kg/wk | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The use of intravenous administration of TH1902 was next investigated in the TNBC-derived MDA-MB-231 xenograft model. Mice subjected to docetaxel treatment were administered with three intravenous injections at 15 mg/kg/wk (MTD), whereas those treated with TH1902 received five injections of an equivalent dose of docetaxel at 35 mg/kg/wk. Similar to what was seen with the intraperitoneal administration protocol, a sustained decrease in tumor size was observed following intravenous administration of TH1902 until day 70, whereas a restart of tumor growth was observed at day 50 for the docetaxel-treated mice. When lower doses, equivalent to the quarter of the MTD, were used of docetaxel (3.75 mg/kg) and TH1902 (8.75 mg/kg), tumor growth as assessed by luminescence intensity was unaffected by intravenous administration of docetaxel, whereas it was significantly inhibited in the TH1902-treated group. This was further confirmed through the measurement of the tumor burden luminescence where TH1902 reduced it significantly in comparison to vehicle- or docetaxel-treated groups. Interestingly, body weight changes remained within endpoint limits in mice on intravenous administration of either docetaxel or TH1902 (data not shown). Similar conclusions were reached on testing another TNBC-derived HCC-70 xenograft model. In fact, administration of TH1902 at 8.75 mg/kg/wk led to a 93% inhibition of HCC-70 tumor growth as compared to 24% for docetaxel alone at an equivalent dose.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 17 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
33.30%
|
|||
| Administration Time | 5 days | ||||
| Administration Dosage | 8.75 mg/kg | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The use of intravenous administration of TH1902 was next investigated in the TNBC-derived MDA-MB-231 xenograft model. Mice subjected to docetaxel treatment were administered with three intravenous injections at 15 mg/kg/wk (MTD), whereas those treated with TH1902 received five injections of an equivalent dose of docetaxel at 35 mg/kg/wk. Similar to what was seen with the intraperitoneal administration protocol, a sustained decrease in tumor size was observed following intravenous administration of TH1902 until day 70, whereas a restart of tumor growth was observed at day 50 for the docetaxel-treated mice. When lower doses, equivalent to the quarter of the MTD, were used of docetaxel (3.75 mg/kg) and TH1902 (8.75 mg/kg), tumor growth as assessed by luminescence intensity was unaffected by intravenous administration of docetaxel, whereas it was significantly inhibited in the TH1902-treated group. This was further confirmed through the measurement of the tumor burden luminescence where TH1902 reduced it significantly in comparison to vehicle- or docetaxel-treated groups. Interestingly, body weight changes remained within endpoint limits in mice on intravenous administration of either docetaxel or TH1902 (data not shown). Similar conclusions were reached on testing another TNBC-derived HCC-70 xenograft model. In fact, administration of TH1902 at 8.75 mg/kg/wk led to a 93% inhibition of HCC-70 tumor growth as compared to 24% for docetaxel alone at an equivalent dose.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 18 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
42.00%
|
|||
| Administration Time | 5 days | ||||
| Administration Dosage | 50mg /kg/wk | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The in vivo efficacy of TH1902 and docetaxel against a TNBC xenograft model was next investigated in vivo. Thus, nude mice were implanted in the right flank with MDA-MB-231-luc cancer cells, and luminescence was measured to monitor tumor growth. Mice were treated with intraperitoneal injections of either docetaxel at the MTD of 15 mg/kg/wk, or with TH1902 at the maximal injectable dose of 50 mg/kg/wk, both for five cycles of treatment. Unlike the vehicle-treated control group, where the average tumor luminescence increased over time, a significant decline in luminescence intensity was observed, starting at day 5 in the TH1902-treated group. The level of luminescence intensity was also significantly lower in the free docetaxel-treated group, when compared to that in the vehicle-treated control group, but remained higher than that for the TH1902-treated group. Interestingly, tumor relapse was observed in the docetaxel-treated group beginning at day 46, whereas a sustained decrease of tumor volume was maintained in the TH1902-treated group until day 74, at which time point a complete disappearance of the tumor was achieved. Representative images of luminescence are shown for vehicle-, docetaxel-, or TH1902-treated mice at day 14 when the control group reached the tumor volume endpoint and at day 74 post-treatment at the end of the experiment. Quantification of the residual tumor burden at days 14 and 74 after docetaxel or TH1902 treatment was performed, and the analysis confirmed a much better in vivo TH1902 efficacy profile than was seen with unconjugated docetaxel. Given the lack of apoptotic or antiproliferative effects of TH19P01, no rationale supports its further assessment in vivo. As TH1902 is considered a new chemical entity, its best control condition therefore is the unconjugated docetaxel molecule itself. Body weight of mice on intraperitoneal administration of either docetaxel or TH1902 remained within endpoint limits (-20%, data not shown).
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 19 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
50.00%
|
|||
| Administration Time | 10 days | ||||
| Administration Dosage | 8.75 mg/kg | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The use of intravenous administration of TH1902 was next investigated in the TNBC-derived MDA-MB-231 xenograft model. Mice subjected to docetaxel treatment were administered with three intravenous injections at 15 mg/kg/wk (MTD), whereas those treated with TH1902 received five injections of an equivalent dose of docetaxel at 35 mg/kg/wk. Similar to what was seen with the intraperitoneal administration protocol, a sustained decrease in tumor size was observed following intravenous administration of TH1902 until day 70, whereas a restart of tumor growth was observed at day 50 for the docetaxel-treated mice. When lower doses, equivalent to the quarter of the MTD, were used of docetaxel (3.75 mg/kg) and TH1902 (8.75 mg/kg), tumor growth as assessed by luminescence intensity was unaffected by intravenous administration of docetaxel, whereas it was significantly inhibited in the TH1902-treated group. This was further confirmed through the measurement of the tumor burden luminescence where TH1902 reduced it significantly in comparison to vehicle- or docetaxel-treated groups. Interestingly, body weight changes remained within endpoint limits in mice on intravenous administration of either docetaxel or TH1902 (data not shown). Similar conclusions were reached on testing another TNBC-derived HCC-70 xenograft model. In fact, administration of TH1902 at 8.75 mg/kg/wk led to a 93% inhibition of HCC-70 tumor growth as compared to 24% for docetaxel alone at an equivalent dose.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 20 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
55.00%
|
|||
| Administration Time | 5 days | ||||
| Administration Dosage | 35 mg/kg/wk | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The use of intravenous administration of TH1902 was next investigated in the TNBC-derived MDA-MB-231 xenograft model. Mice subjected to docetaxel treatment were administered with three intravenous injections at 15 mg/kg/wk (MTD), whereas those treated with TH1902 received five injections of an equivalent dose of docetaxel at 35 mg/kg/wk. Similar to what was seen with the intraperitoneal administration protocol, a sustained decrease in tumor size was observed following intravenous administration of TH1902 until day 70, whereas a restart of tumor growth was observed at day 50 for the docetaxel-treated mice. When lower doses, equivalent to the quarter of the MTD, were used of docetaxel (3.75 mg/kg) and TH1902 (8.75 mg/kg), tumor growth as assessed by luminescence intensity was unaffected by intravenous administration of docetaxel, whereas it was significantly inhibited in the TH1902-treated group. This was further confirmed through the measurement of the tumor burden luminescence where TH1902 reduced it significantly in comparison to vehicle- or docetaxel-treated groups. Interestingly, body weight changes remained within endpoint limits in mice on intravenous administration of either docetaxel or TH1902 (data not shown). Similar conclusions were reached on testing another TNBC-derived HCC-70 xenograft model. In fact, administration of TH1902 at 8.75 mg/kg/wk led to a 93% inhibition of HCC-70 tumor growth as compared to 24% for docetaxel alone at an equivalent dose.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 21 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
66.70%
|
|||
| Administration Time | 15 days | ||||
| Administration Dosage | 8.75 mg/kg | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The use of intravenous administration of TH1902 was next investigated in the TNBC-derived MDA-MB-231 xenograft model. Mice subjected to docetaxel treatment were administered with three intravenous injections at 15 mg/kg/wk (MTD), whereas those treated with TH1902 received five injections of an equivalent dose of docetaxel at 35 mg/kg/wk. Similar to what was seen with the intraperitoneal administration protocol, a sustained decrease in tumor size was observed following intravenous administration of TH1902 until day 70, whereas a restart of tumor growth was observed at day 50 for the docetaxel-treated mice. When lower doses, equivalent to the quarter of the MTD, were used of docetaxel (3.75 mg/kg) and TH1902 (8.75 mg/kg), tumor growth as assessed by luminescence intensity was unaffected by intravenous administration of docetaxel, whereas it was significantly inhibited in the TH1902-treated group. This was further confirmed through the measurement of the tumor burden luminescence where TH1902 reduced it significantly in comparison to vehicle- or docetaxel-treated groups. Interestingly, body weight changes remained within endpoint limits in mice on intravenous administration of either docetaxel or TH1902 (data not shown). Similar conclusions were reached on testing another TNBC-derived HCC-70 xenograft model. In fact, administration of TH1902 at 8.75 mg/kg/wk led to a 93% inhibition of HCC-70 tumor growth as compared to 24% for docetaxel alone at an equivalent dose.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 22 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
70.00%
|
|||
| Administration Time | 10 days | ||||
| Administration Dosage | 50mg /kg/wk | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The in vivo efficacy of TH1902 and docetaxel against a TNBC xenograft model was next investigated in vivo. Thus, nude mice were implanted in the right flank with MDA-MB-231-luc cancer cells, and luminescence was measured to monitor tumor growth. Mice were treated with intraperitoneal injections of either docetaxel at the MTD of 15 mg/kg/wk, or with TH1902 at the maximal injectable dose of 50 mg/kg/wk, both for five cycles of treatment. Unlike the vehicle-treated control group, where the average tumor luminescence increased over time, a significant decline in luminescence intensity was observed, starting at day 5 in the TH1902-treated group. The level of luminescence intensity was also significantly lower in the free docetaxel-treated group, when compared to that in the vehicle-treated control group, but remained higher than that for the TH1902-treated group. Interestingly, tumor relapse was observed in the docetaxel-treated group beginning at day 46, whereas a sustained decrease of tumor volume was maintained in the TH1902-treated group until day 74, at which time point a complete disappearance of the tumor was achieved. Representative images of luminescence are shown for vehicle-, docetaxel-, or TH1902-treated mice at day 14 when the control group reached the tumor volume endpoint and at day 74 post-treatment at the end of the experiment. Quantification of the residual tumor burden at days 14 and 74 after docetaxel or TH1902 treatment was performed, and the analysis confirmed a much better in vivo TH1902 efficacy profile than was seen with unconjugated docetaxel. Given the lack of apoptotic or antiproliferative effects of TH19P01, no rationale supports its further assessment in vivo. As TH1902 is considered a new chemical entity, its best control condition therefore is the unconjugated docetaxel molecule itself. Body weight of mice on intraperitoneal administration of either docetaxel or TH1902 remained within endpoint limits (-20%, data not shown).
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 23 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
78.60%
|
|||
| Administration Time | 15 days | ||||
| Administration Dosage | 35 mg/kg | ||||
| MOA of PDC |
Sortilin (SORT1) is a receptor protein which cycles from the cell surface through intracellular membrane bodies. It binds several circulating proteins and peptides, including progranulin and neurotensin, prior to their rapid intracellular internalization. It is also involved in their intracellular trafficking in endosomal vesicles from the plasma membrane to lysosomes through the endocytic pathway. Deregulation in SORT1 functions has been implicated in cardiovascular disease, Alzheimers disease, and diabetes, whereas its upregulation has been documented in several forms of cancer. Recently, TH1902, a peptide-drug conjugate (PDC) to which two docetaxel molecules were linked to TH19P01 peptide, was generated. TH1902 was demonstrated to recognize and exploit SORT1 functions, and to efficiently inhibit in vitro cell proliferation, and in vivo growth of xenografts from gynecological cancers including triple-negative breast cancer (TNBC)-derived MDA-MB-231 cells, ovarian cancer-derived ES2 and SKOV3 cells, as well as endometrial cancer-derived AN3-CA cells. Interestingly, TH1902 was found to reduce in vitro vasculogenic mimicry (VM) processes in breast and ovarian cancer cells. While some molecular aspects revealing the inter-relationship existence between CSC and VM have been addressed in breast cancer, gastrointestinal cancer, and melanoma, the specific targeting of the CSC subpopulation by TH1902 remains unexplored.
Click to Show/Hide
|
||||
| Description |
In vivo assessment in tumor-bearing animal models classically complements cell cultures data for comparing cancer treatment efficacies. For this reason, hTNBCSC and hOvCSC xenografts were implanted subcutaneously into immunodeficient mice as described in the Methods section with only 1000 cells given their highly tumorigenic nature. Three days later, the animals began receiving weekly IV bolus administration of either vehicle, docetaxel, or TH1902. Docetaxel was administered at a dose (15 mg/kg/week; for 3 cycles) in accordance with the estimated maximal tolerated dose (MTD) for mice, as well as at 1/4 of the MTD (3.75 mg/kg/week). TH1902 was administered at doses (35 and 8.75 mg/kg/week) which contained quantities of docetaxel equivalent to those in the two administrations of free docetaxel. From the size of the hTNBCSC and hOvCSC tumors, it is apparent that docetaxel had little impact on xenografts growth when administered neither at its MTD, for both hTNBCSC and hOvCSC, nor at 1/4 of this dosage as reflected by the growth curves. TH1902, when administered at a dosage equivalent to docetaxel at its MTD, provided greater tumor growth inhibition than did docetaxel for both xenograft models. Furthermore, higher dosage of administered TH1902 (up to 1.5 equivalent of docetaxel MTD) did not generate significant differences in terms of hOvCSC tumor inhibition without affecting mice body weights suggesting that TH1902, even at higher doses, is better tolerated compared to docetaxel. Then, in order to statistically compare the effects of docetaxel and TH1902 on tumor growth, the tumor sizes measured at the vehicle group endpoint were compared and statistically significant differences between the tumor sizes in vehicle-treated animals found in TH1902-treated animals for hTNBCSC and hOvCSC. Mouse body weight was used as an indicator of the morbidity associated with administration of docetaxel or TH1902. Administration of docetaxel at its MTD provoked a weight loss that approached 10% after the treatments, which is often observed in xenograft models with administration of docetaxel at this level. The body weights of animals treated with an equivalent quantity or a 1.5-fold equivalent of TH1902 were maintained at a roughly constant level throughout the experiment. The animals treated with vehicle recorded a slight weight gain (~5%) over this period while animals treated with the lower dosages of docetaxel or TH1902 were similar to the animals treated with vehicle. The body weight data indicates that TH1902 appears to be better tolerated than the equivalent quantity of free docetaxel, in addition to the fact that TH1902 is more efficacious than docetaxel when administered in vivo in the murine models of CSC tested.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian cancer | Ovarian cancer stem cell (hOvCSC) | Homo sapiens | ||
| Experiment 24 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
79%
|
|||
| Administration Time | 15 days | ||||
| Administration Dosage | 42.75 mg/kg | ||||
| MOA of PDC |
Sortilin (SORT1) is a receptor protein which cycles from the cell surface through intracellular membrane bodies. It binds several circulating proteins and peptides, including progranulin and neurotensin, prior to their rapid intracellular internalization. It is also involved in their intracellular trafficking in endosomal vesicles from the plasma membrane to lysosomes through the endocytic pathway. Deregulation in SORT1 functions has been implicated in cardiovascular disease, Alzheimers disease, and diabetes, whereas its upregulation has been documented in several forms of cancer. Recently, TH1902, a peptide-drug conjugate (PDC) to which two docetaxel molecules were linked to TH19P01 peptide, was generated. TH1902 was demonstrated to recognize and exploit SORT1 functions, and to efficiently inhibit in vitro cell proliferation, and in vivo growth of xenografts from gynecological cancers including triple-negative breast cancer (TNBC)-derived MDA-MB-231 cells, ovarian cancer-derived ES2 and SKOV3 cells, as well as endometrial cancer-derived AN3-CA cells. Interestingly, TH1902 was found to reduce in vitro vasculogenic mimicry (VM) processes in breast and ovarian cancer cells. While some molecular aspects revealing the inter-relationship existence between CSC and VM have been addressed in breast cancer, gastrointestinal cancer, and melanoma, the specific targeting of the CSC subpopulation by TH1902 remains unexplored.
Click to Show/Hide
|
||||
| Description |
In vivo assessment in tumor-bearing animal models classically complements cell cultures data for comparing cancer treatment efficacies. For this reason, hTNBCSC and hOvCSC xenografts were implanted subcutaneously into immunodeficient mice as described in the Methods section with only 1000 cells given their highly tumorigenic nature. Three days later, the animals began receiving weekly IV bolus administration of either vehicle, docetaxel, or TH1902. Docetaxel was administered at a dose (15 mg/kg/week; for 3 cycles) in accordance with the estimated maximal tolerated dose (MTD) for mice, as well as at 1/4 of the MTD (3.75 mg/kg/week). TH1902 was administered at doses (35 and 8.75 mg/kg/week) which contained quantities of docetaxel equivalent to those in the two administrations of free docetaxel. From the size of the hTNBCSC and hOvCSC tumors, it is apparent that docetaxel had little impact on xenografts growth when administered neither at its MTD, for both hTNBCSC and hOvCSC, nor at 1/4 of this dosage as reflected by the growth curves. TH1902, when administered at a dosage equivalent to docetaxel at its MTD, provided greater tumor growth inhibition than did docetaxel for both xenograft models. Furthermore, higher dosage of administered TH1902 (up to 1.5 equivalent of docetaxel MTD) did not generate significant differences in terms of hOvCSC tumor inhibition without affecting mice body weights suggesting that TH1902, even at higher doses, is better tolerated compared to docetaxel. Then, in order to statistically compare the effects of docetaxel and TH1902 on tumor growth, the tumor sizes measured at the vehicle group endpoint were compared and statistically significant differences between the tumor sizes in vehicle-treated animals found in TH1902-treated animals for hTNBCSC and hOvCSC. Mouse body weight was used as an indicator of the morbidity associated with administration of docetaxel or TH1902. Administration of docetaxel at its MTD provoked a weight loss that approached 10% after the treatments, which is often observed in xenograft models with administration of docetaxel at this level. The body weights of animals treated with an equivalent quantity or a 1.5-fold equivalent of TH1902 were maintained at a roughly constant level throughout the experiment. The animals treated with vehicle recorded a slight weight gain (~5%) over this period while animals treated with the lower dosages of docetaxel or TH1902 were similar to the animals treated with vehicle. The body weight data indicates that TH1902 appears to be better tolerated than the equivalent quantity of free docetaxel, in addition to the fact that TH1902 is more efficacious than docetaxel when administered in vivo in the murine models of CSC tested.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian cancer | Ovarian cancer stem cell (hOvCSC) | Homo sapiens | ||
| Experiment 25 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
80.00%
|
|||
| Administration Time | 15 days | ||||
| Administration Dosage | 50mg /kg/wk | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The in vivo efficacy of TH1902 and docetaxel against a TNBC xenograft model was next investigated in vivo. Thus, nude mice were implanted in the right flank with MDA-MB-231-luc cancer cells, and luminescence was measured to monitor tumor growth. Mice were treated with intraperitoneal injections of either docetaxel at the MTD of 15 mg/kg/wk, or with TH1902 at the maximal injectable dose of 50 mg/kg/wk, both for five cycles of treatment. Unlike the vehicle-treated control group, where the average tumor luminescence increased over time, a significant decline in luminescence intensity was observed, starting at day 5 in the TH1902-treated group. The level of luminescence intensity was also significantly lower in the free docetaxel-treated group, when compared to that in the vehicle-treated control group, but remained higher than that for the TH1902-treated group. Interestingly, tumor relapse was observed in the docetaxel-treated group beginning at day 46, whereas a sustained decrease of tumor volume was maintained in the TH1902-treated group until day 74, at which time point a complete disappearance of the tumor was achieved. Representative images of luminescence are shown for vehicle-, docetaxel-, or TH1902-treated mice at day 14 when the control group reached the tumor volume endpoint and at day 74 post-treatment at the end of the experiment. Quantification of the residual tumor burden at days 14 and 74 after docetaxel or TH1902 treatment was performed, and the analysis confirmed a much better in vivo TH1902 efficacy profile than was seen with unconjugated docetaxel. Given the lack of apoptotic or antiproliferative effects of TH19P01, no rationale supports its further assessment in vivo. As TH1902 is considered a new chemical entity, its best control condition therefore is the unconjugated docetaxel molecule itself. Body weight of mice on intraperitoneal administration of either docetaxel or TH1902 remained within endpoint limits (-20%, data not shown).
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 26 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
80.00%
|
|||
| Administration Time | 10 days | ||||
| Administration Dosage | 35 mg/kg/wk | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The use of intravenous administration of TH1902 was next investigated in the TNBC-derived MDA-MB-231 xenograft model. Mice subjected to docetaxel treatment were administered with three intravenous injections at 15 mg/kg/wk (MTD), whereas those treated with TH1902 received five injections of an equivalent dose of docetaxel at 35 mg/kg/wk. Similar to what was seen with the intraperitoneal administration protocol, a sustained decrease in tumor size was observed following intravenous administration of TH1902 until day 70, whereas a restart of tumor growth was observed at day 50 for the docetaxel-treated mice. When lower doses, equivalent to the quarter of the MTD, were used of docetaxel (3.75 mg/kg) and TH1902 (8.75 mg/kg), tumor growth as assessed by luminescence intensity was unaffected by intravenous administration of docetaxel, whereas it was significantly inhibited in the TH1902-treated group. This was further confirmed through the measurement of the tumor burden luminescence where TH1902 reduced it significantly in comparison to vehicle- or docetaxel-treated groups. Interestingly, body weight changes remained within endpoint limits in mice on intravenous administration of either docetaxel or TH1902 (data not shown). Similar conclusions were reached on testing another TNBC-derived HCC-70 xenograft model. In fact, administration of TH1902 at 8.75 mg/kg/wk led to a 93% inhibition of HCC-70 tumor growth as compared to 24% for docetaxel alone at an equivalent dose.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 27 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
80.00%
|
|||
| Administration Time | 20 days | ||||
| Administration Dosage | 8.75 mg/kg | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The use of intravenous administration of TH1902 was next investigated in the TNBC-derived MDA-MB-231 xenograft model. Mice subjected to docetaxel treatment were administered with three intravenous injections at 15 mg/kg/wk (MTD), whereas those treated with TH1902 received five injections of an equivalent dose of docetaxel at 35 mg/kg/wk. Similar to what was seen with the intraperitoneal administration protocol, a sustained decrease in tumor size was observed following intravenous administration of TH1902 until day 70, whereas a restart of tumor growth was observed at day 50 for the docetaxel-treated mice. When lower doses, equivalent to the quarter of the MTD, were used of docetaxel (3.75 mg/kg) and TH1902 (8.75 mg/kg), tumor growth as assessed by luminescence intensity was unaffected by intravenous administration of docetaxel, whereas it was significantly inhibited in the TH1902-treated group. This was further confirmed through the measurement of the tumor burden luminescence where TH1902 reduced it significantly in comparison to vehicle- or docetaxel-treated groups. Interestingly, body weight changes remained within endpoint limits in mice on intravenous administration of either docetaxel or TH1902 (data not shown). Similar conclusions were reached on testing another TNBC-derived HCC-70 xenograft model. In fact, administration of TH1902 at 8.75 mg/kg/wk led to a 93% inhibition of HCC-70 tumor growth as compared to 24% for docetaxel alone at an equivalent dose.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 28 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
86.00%
|
|||
| Administration Time | 15 days | ||||
| Administration Dosage | 35 mg/kg/wk | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The use of intravenous administration of TH1902 was next investigated in the TNBC-derived MDA-MB-231 xenograft model. Mice subjected to docetaxel treatment were administered with three intravenous injections at 15 mg/kg/wk (MTD), whereas those treated with TH1902 received five injections of an equivalent dose of docetaxel at 35 mg/kg/wk. Similar to what was seen with the intraperitoneal administration protocol, a sustained decrease in tumor size was observed following intravenous administration of TH1902 until day 70, whereas a restart of tumor growth was observed at day 50 for the docetaxel-treated mice. When lower doses, equivalent to the quarter of the MTD, were used of docetaxel (3.75 mg/kg) and TH1902 (8.75 mg/kg), tumor growth as assessed by luminescence intensity was unaffected by intravenous administration of docetaxel, whereas it was significantly inhibited in the TH1902-treated group. This was further confirmed through the measurement of the tumor burden luminescence where TH1902 reduced it significantly in comparison to vehicle- or docetaxel-treated groups. Interestingly, body weight changes remained within endpoint limits in mice on intravenous administration of either docetaxel or TH1902 (data not shown). Similar conclusions were reached on testing another TNBC-derived HCC-70 xenograft model. In fact, administration of TH1902 at 8.75 mg/kg/wk led to a 93% inhibition of HCC-70 tumor growth as compared to 24% for docetaxel alone at an equivalent dose.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
| Experiment 29 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
87%
|
|||
| Administration Time | 15 days | ||||
| Administration Dosage | 52.5 mg/kg | ||||
| MOA of PDC |
Sortilin (SORT1) is a receptor protein which cycles from the cell surface through intracellular membrane bodies. It binds several circulating proteins and peptides, including progranulin and neurotensin, prior to their rapid intracellular internalization. It is also involved in their intracellular trafficking in endosomal vesicles from the plasma membrane to lysosomes through the endocytic pathway. Deregulation in SORT1 functions has been implicated in cardiovascular disease, Alzheimers disease, and diabetes, whereas its upregulation has been documented in several forms of cancer. Recently, TH1902, a peptide-drug conjugate (PDC) to which two docetaxel molecules were linked to TH19P01 peptide, was generated. TH1902 was demonstrated to recognize and exploit SORT1 functions, and to efficiently inhibit in vitro cell proliferation, and in vivo growth of xenografts from gynecological cancers including triple-negative breast cancer (TNBC)-derived MDA-MB-231 cells, ovarian cancer-derived ES2 and SKOV3 cells, as well as endometrial cancer-derived AN3-CA cells. Interestingly, TH1902 was found to reduce in vitro vasculogenic mimicry (VM) processes in breast and ovarian cancer cells. While some molecular aspects revealing the inter-relationship existence between CSC and VM have been addressed in breast cancer, gastrointestinal cancer, and melanoma, the specific targeting of the CSC subpopulation by TH1902 remains unexplored.
Click to Show/Hide
|
||||
| Description |
In vivo assessment in tumor-bearing animal models classically complements cell cultures data for comparing cancer treatment efficacies. For this reason, hTNBCSC and hOvCSC xenografts were implanted subcutaneously into immunodeficient mice as described in the Methods section with only 1000 cells given their highly tumorigenic nature. Three days later, the animals began receiving weekly IV bolus administration of either vehicle, docetaxel, or TH1902. Docetaxel was administered at a dose (15 mg/kg/week; for 3 cycles) in accordance with the estimated maximal tolerated dose (MTD) for mice, as well as at 1/4 of the MTD (3.75 mg/kg/week). TH1902 was administered at doses (35 and 8.75 mg/kg/week) which contained quantities of docetaxel equivalent to those in the two administrations of free docetaxel. From the size of the hTNBCSC and hOvCSC tumors, it is apparent that docetaxel had little impact on xenografts growth when administered neither at its MTD, for both hTNBCSC and hOvCSC, nor at 1/4 of this dosage as reflected by the growth curves. TH1902, when administered at a dosage equivalent to docetaxel at its MTD, provided greater tumor growth inhibition than did docetaxel for both xenograft models. Furthermore, higher dosage of administered TH1902 (up to 1.5 equivalent of docetaxel MTD) did not generate significant differences in terms of hOvCSC tumor inhibition without affecting mice body weights suggesting that TH1902, even at higher doses, is better tolerated compared to docetaxel. Then, in order to statistically compare the effects of docetaxel and TH1902 on tumor growth, the tumor sizes measured at the vehicle group endpoint were compared and statistically significant differences between the tumor sizes in vehicle-treated animals found in TH1902-treated animals for hTNBCSC and hOvCSC. Mouse body weight was used as an indicator of the morbidity associated with administration of docetaxel or TH1902. Administration of docetaxel at its MTD provoked a weight loss that approached 10% after the treatments, which is often observed in xenograft models with administration of docetaxel at this level. The body weights of animals treated with an equivalent quantity or a 1.5-fold equivalent of TH1902 were maintained at a roughly constant level throughout the experiment. The animals treated with vehicle recorded a slight weight gain (~5%) over this period while animals treated with the lower dosages of docetaxel or TH1902 were similar to the animals treated with vehicle. The body weight data indicates that TH1902 appears to be better tolerated than the equivalent quantity of free docetaxel, in addition to the fact that TH1902 is more efficacious than docetaxel when administered in vivo in the murine models of CSC tested.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian cancer | Ovarian cancer stem cell (hOvCSC) | Homo sapiens | ||
| Experiment 30 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
91.00%
|
|||
| Administration Time | 20 days | ||||
| Administration Dosage | 35 mg/kg/wk | ||||
| MOA of PDC |
Triple-negative breast cancer (TNBC) is a heterogeneous subgroup of cancers which lacks the expression and/or amplification of targetable biomarkers (ie, estrogen receptor, progestrogen receptor, and human epidermal growth factor receptor 2), and is often associated with the worse disease-specific outcomes than other breast cancer subtypes. Here, we report that high expression of the sortilin (SORT1) receptor correlates with the decreased survival in TNBC patients, and more importantly in those bearing lymph node metastases. By exploiting SORT1 function in ligand internalization, a new anticancer treatment strategy was designed to target SORT1-positive TNBC-derived cells both in vitro and in two in vivo tumor xenografts models. A peptide (TH19P01), which requires SORT1 for internalization and to which many anticancer drugs could be conjugated, was developed. In vitro, while the TH19P01 peptide itself did not exert any antiproliferative or apoptotic effects, the docetaxel-TH19P01 conjugate (TH1902) exerted potent antiproliferative and antimigratory activities when tested on TNBC-derived MDA-MB-231 cells. TH1902 triggered faster and more potent apoptotic cell death than did unconjugated docetaxel. The apoptotic and antimigratory effects of TH1902 were both reversed by two SORT1 ligands, neurotensin and progranulin, and on siRNA-mediated silencing of SORT1. TH1902 also altered microtubule polymerization and triggered the downregulation of the anti-apoptotic Bcl-xL biomarker. In vivo, both i.p. and i.v. administrations of TH1902 led to greater tumor regression in two MDA-MB-231 and HCC-70 murine xenograft models than did docetaxel, without inducing neutropenia. Altogether, the data demonstrates the high in vivo efficacy and safety of TH1902 against TNBC through a SORT1 receptor-mediated mechanism. This property allows for selective treatment of SORT1-positive TNBC and makes TH1902 a promising avenue for personalized therapy with the potential of improving the therapeutic window of cytotoxic anticancer drugs such as docetaxel.
Click to Show/Hide
|
||||
| Description |
The use of intravenous administration of TH1902 was next investigated in the TNBC-derived MDA-MB-231 xenograft model. Mice subjected to docetaxel treatment were administered with three intravenous injections at 15 mg/kg/wk (MTD), whereas those treated with TH1902 received five injections of an equivalent dose of docetaxel at 35 mg/kg/wk. Similar to what was seen with the intraperitoneal administration protocol, a sustained decrease in tumor size was observed following intravenous administration of TH1902 until day 70, whereas a restart of tumor growth was observed at day 50 for the docetaxel-treated mice. When lower doses, equivalent to the quarter of the MTD, were used of docetaxel (3.75 mg/kg) and TH1902 (8.75 mg/kg), tumor growth as assessed by luminescence intensity was unaffected by intravenous administration of docetaxel, whereas it was significantly inhibited in the TH1902-treated group. This was further confirmed through the measurement of the tumor burden luminescence where TH1902 reduced it significantly in comparison to vehicle- or docetaxel-treated groups. Interestingly, body weight changes remained within endpoint limits in mice on intravenous administration of either docetaxel or TH1902 (data not shown). Similar conclusions were reached on testing another TNBC-derived HCC-70 xenograft model. In fact, administration of TH1902 at 8.75 mg/kg/wk led to a 93% inhibition of HCC-70 tumor growth as compared to 24% for docetaxel alone at an equivalent dose.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 1.44 h | ||||
References
