Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00351
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
TPP-DOX-AP2H
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
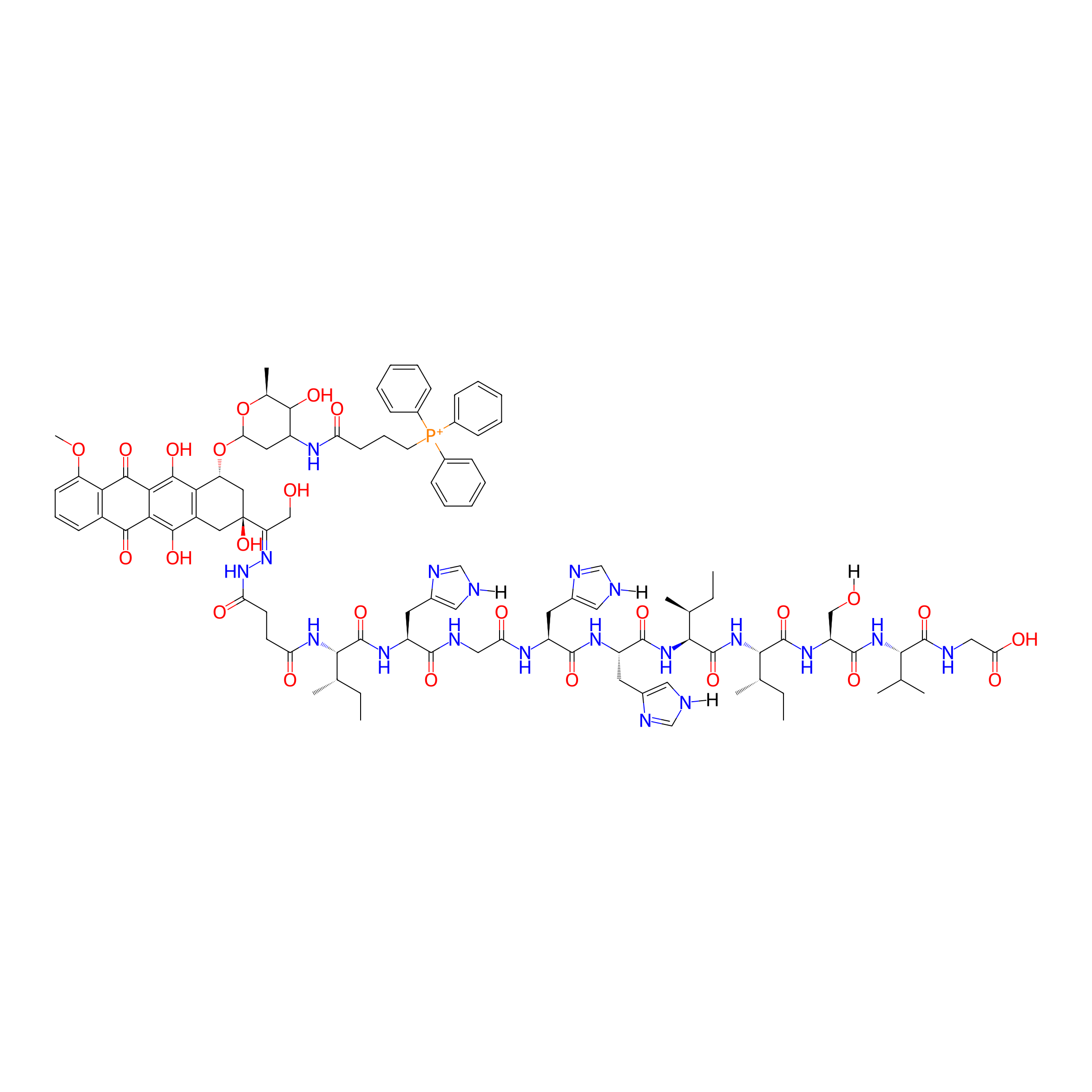
|
|||||
| Peptide Name |
AP2H
|
Peptide Info | ||||
| Receptor Name |
Mitochondria-eating protein (SPATA18)
|
Receptor Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
Hydrazone bond
|
Linker Info | ||||
| Peptide Modified Type |
The modification of binding with chemical macromolecules
|
|||||
| Modified Segment |
Hydrazine group
|
|||||
| Ternimal Modification |
N-terminal modification
|
|||||
| Formula |
C101H129N19O25P+
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 2040.225 | ||||
| Lipid-water partition coefficient (xlogp) | 0.4958 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 22 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 28 | |||||
| Rotatable Bond Count (rotbonds) | 52 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Average tumor volume shrunk |
55%
|
|||
| Administration Time | 18 days | ||||
| Administration Dosage | 20 µM | ||||
| MOA of PDC |
Initiated by the interaction between AP2H and membrane-anchored LAPTM4B protein, PDC specifically recognized and bound cancer cells. The receptor-mediated endocytic pathway then led to the delivery of PDC to lysosomes. The low pH environment in lysosomes efficiently cut the hydrazone bond and released TPP-DOX. The successful escape from lysosomes enabled further transportation of TPP-DOX to mitochondria directed by targeting group TPP. Because TPP-DOX is highly active in producing ROS and mitochondria are prone to oxidative damage, the disruption in the electron transport chain and ATP synthesis finally lead to mitochondrial dysfunction and cell death. With dual-targeting ability, PDC can effectively bypass the interaction with P-gp. Meanwhile, due to the energy-dependent expression feature of P-gp, the damage to mitochondria inhibit P-gp expression, thus inhibit the efflux. (9) The targeted and mitochondria interrupted cell damaging pathway allow PDC to exert toxicity both wild type and drug resistant
Click to Show/Hide
|
||||
| Description |
The average tumor volume shrunk by 55% at day 18 compared with that of the control group (Figure 4c).
|
||||
| In Vivo Model | HepG2 tumor xenograft model. | ||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
17%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 20 µM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Initiated by the interaction between AP2H and membrane-anchored LAPTM4B protein, PDC specifically recognized and bound cancer cells. The receptor-mediated endocytic pathway then led to the delivery of PDC to lysosomes. The low pH environment in lysosomes efficiently cut the hydrazone bond and released TPP-DOX. The successful escape from lysosomes enabled further transportation of TPP-DOX to mitochondria directed by targeting group TPP. Because TPP-DOX is highly active in producing ROS and mitochondria are prone to oxidative damage, the disruption in the electron transport chain and ATP synthesis finally lead to mitochondrial dysfunction and cell death. With dual-targeting ability, PDC can effectively bypass the interaction with P-gp. Meanwhile, due to the energy-dependent expression feature of P-gp, the damage to mitochondria inhibit P-gp expression, thus inhibit the efflux. (9) The targeted and mitochondria interrupted cell damaging pathway allow PDC to exert toxicity both wild type and drug resistant
Click to Show/Hide
|
||||
| Description |
PDC killed most HepG2 cells with a high efficiency of 83% and left HEK293 cells unaffected.
|
||||
| In Vitro Model | Hepatoblastoma | Hep-G2 cell | CVCL_0027 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
40%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 20 µM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Initiated by the interaction between AP2H and membrane-anchored LAPTM4B protein, PDC specifically recognized and bound cancer cells. The receptor-mediated endocytic pathway then led to the delivery of PDC to lysosomes. The low pH environment in lysosomes efficiently cut the hydrazone bond and released TPP-DOX. The successful escape from lysosomes enabled further transportation of TPP-DOX to mitochondria directed by targeting group TPP. Because TPP-DOX is highly active in producing ROS and mitochondria are prone to oxidative damage, the disruption in the electron transport chain and ATP synthesis finally lead to mitochondrial dysfunction and cell death. With dual-targeting ability, PDC can effectively bypass the interaction with P-gp. Meanwhile, due to the energy-dependent expression feature of P-gp, the damage to mitochondria inhibit P-gp expression, thus inhibit the efflux. (9) The targeted and mitochondria interrupted cell damaging pathway allow PDC to exert toxicity both wild type and drug resistant
Click to Show/Hide
|
||||
| Description |
PDC maintained almost the same cytotoxicity against MCF-7/ADR cells and MCF-7/WT cells
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
45%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 20 µM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Initiated by the interaction between AP2H and membrane-anchored LAPTM4B protein, PDC specifically recognized and bound cancer cells. The receptor-mediated endocytic pathway then led to the delivery of PDC to lysosomes. The low pH environment in lysosomes efficiently cut the hydrazone bond and released TPP-DOX. The successful escape from lysosomes enabled further transportation of TPP-DOX to mitochondria directed by targeting group TPP. Because TPP-DOX is highly active in producing ROS and mitochondria are prone to oxidative damage, the disruption in the electron transport chain and ATP synthesis finally lead to mitochondrial dysfunction and cell death. With dual-targeting ability, PDC can effectively bypass the interaction with P-gp. Meanwhile, due to the energy-dependent expression feature of P-gp, the damage to mitochondria inhibit P-gp expression, thus inhibit the efflux. (9) The targeted and mitochondria interrupted cell damaging pathway allow PDC to exert toxicity both wild type and drug resistant
Click to Show/Hide
|
||||
| Description |
PDC maintained almost the same cytotoxicity against MCF-7/ADR cells and MCF-7/WT cells
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF7/ADR cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
100%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 20 µM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Initiated by the interaction between AP2H and membrane-anchored LAPTM4B protein, PDC specifically recognized and bound cancer cells. The receptor-mediated endocytic pathway then led to the delivery of PDC to lysosomes. The low pH environment in lysosomes efficiently cut the hydrazone bond and released TPP-DOX. The successful escape from lysosomes enabled further transportation of TPP-DOX to mitochondria directed by targeting group TPP. Because TPP-DOX is highly active in producing ROS and mitochondria are prone to oxidative damage, the disruption in the electron transport chain and ATP synthesis finally lead to mitochondrial dysfunction and cell death. With dual-targeting ability, PDC can effectively bypass the interaction with P-gp. Meanwhile, due to the energy-dependent expression feature of P-gp, the damage to mitochondria inhibit P-gp expression, thus inhibit the efflux. (9) The targeted and mitochondria interrupted cell damaging pathway allow PDC to exert toxicity both wild type and drug resistant
Click to Show/Hide
|
||||
| Description |
PDC killed most HepG2 cells with a high efficiency of 83% and left HEK293 cells unaffected.
|
||||
| In Vitro Model | Normal | HEK293 cell | CVCL_0045 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
15 µM
|
|||
| MOA of PDC |
Initiated by the interaction between AP2H and membrane-anchored LAPTM4B protein, PDC specifically recognized and bound cancer cells. The receptor-mediated endocytic pathway then led to the delivery of PDC to lysosomes. The low pH environment in lysosomes efficiently cut the hydrazone bond and released TPP-DOX. The successful escape from lysosomes enabled further transportation of TPP-DOX to mitochondria directed by targeting group TPP. Because TPP-DOX is highly active in producing ROS and mitochondria are prone to oxidative damage, the disruption in the electron transport chain and ATP synthesis finally lead to mitochondrial dysfunction and cell death. With dual-targeting ability, PDC can effectively bypass the interaction with P-gp. Meanwhile, due to the energy-dependent expression feature of P-gp, the damage to mitochondria inhibit P-gp expression, thus inhibit the efflux. (9) The targeted and mitochondria interrupted cell damaging pathway allow PDC to exert toxicity both wild type and drug resistant
Click to Show/Hide
|
||||
| Description |
For PDC, its IC50 values against MCF-7/WT (15 uM) and MCF-7/ADR (18 uM) are almost the same, confirming its effectiveness in bypass drug resistance.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
18 µM
|
|||
| MOA of PDC |
Initiated by the interaction between AP2H and membrane-anchored LAPTM4B protein, PDC specifically recognized and bound cancer cells. The receptor-mediated endocytic pathway then led to the delivery of PDC to lysosomes. The low pH environment in lysosomes efficiently cut the hydrazone bond and released TPP-DOX. The successful escape from lysosomes enabled further transportation of TPP-DOX to mitochondria directed by targeting group TPP. Because TPP-DOX is highly active in producing ROS and mitochondria are prone to oxidative damage, the disruption in the electron transport chain and ATP synthesis finally lead to mitochondrial dysfunction and cell death. With dual-targeting ability, PDC can effectively bypass the interaction with P-gp. Meanwhile, due to the energy-dependent expression feature of P-gp, the damage to mitochondria inhibit P-gp expression, thus inhibit the efflux. (9) The targeted and mitochondria interrupted cell damaging pathway allow PDC to exert toxicity both wild type and drug resistant
Click to Show/Hide
|
||||
| Description |
For PDC, its IC50 values against MCF-7/WT (15 uM) and MCF-7/ADR (18 uM) are almost the same, confirming its effectiveness in bypass drug resistance.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF7/ADR cell | CVCL_0031 | ||
References
