Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00362
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
SBI1301
|
|||||
| PDC Status |
Preclinical
|
|||||
| Indication |
In total 3 Indication(s)
|
|||||
| Structure |
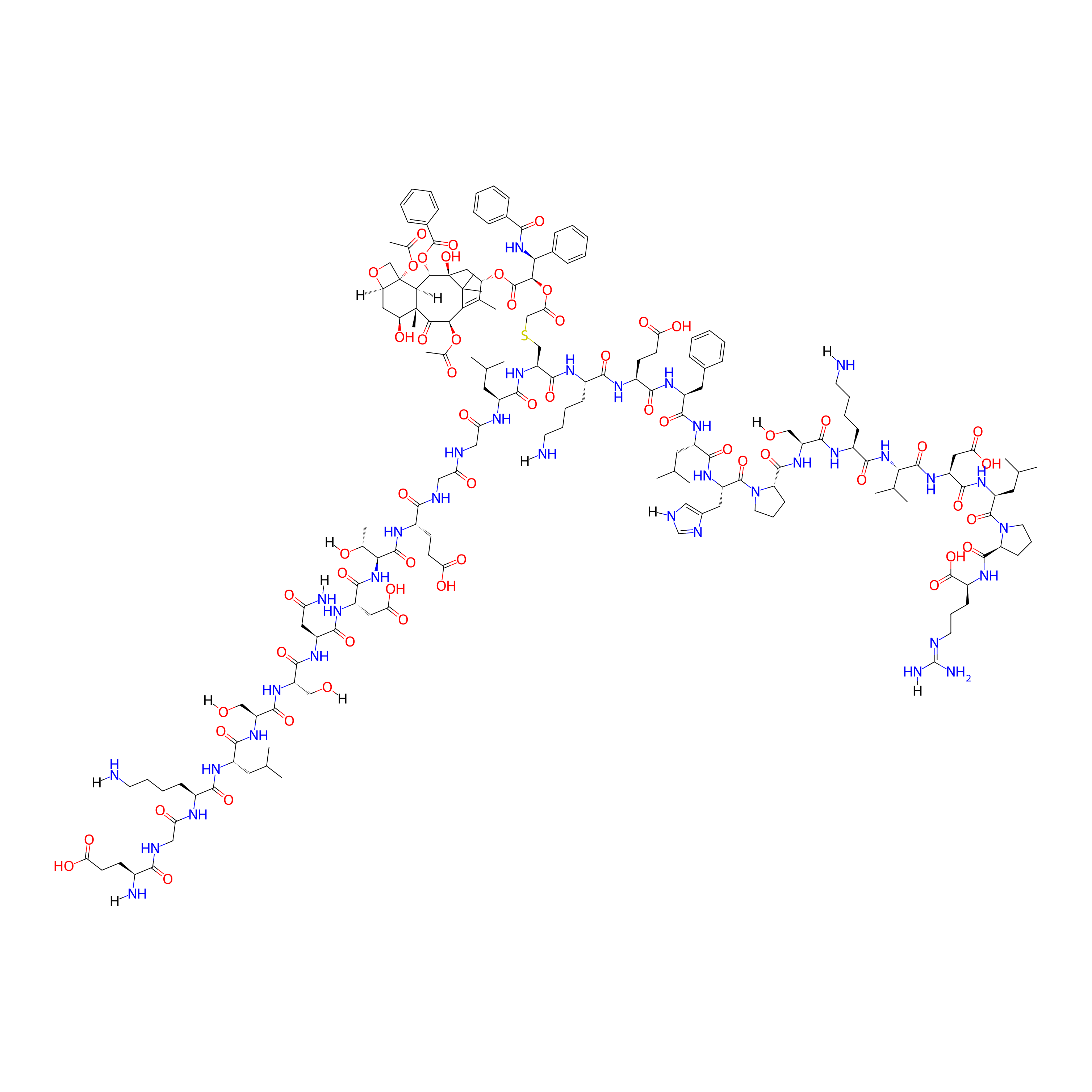
|
|||||
| Peptide Name |
SOR-C27
|
Peptide Info | ||||
| Receptor Name |
Transient receptor potential cation channel subfamily V member 6 (TRPV6)
|
Receptor Info | ||||
| Drug Name |
Paclitaxel
|
Drug Info | ||||
| Therapeutic Target |
Microtubule (MT)
|
Target Info | ||||
| Linker Name |
Thioglycolic acid
|
Linker Info | ||||
| Formula |
C176H257N37O58S
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 3851.26 | ||||
| Lipid-water partition coefficient (xlogp) | -10.7905 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 45 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 59 | |||||
| Rotatable Bond Count (rotbonds) | 122 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
10%
|
|||
| Administration Time | 54 days | ||||
| Administration Dosage | 0.3 mg/kg | ||||
| Evaluation Method | Tumor volume detection | ||||
| Description |
In xenografted prostate cancer in mice, only 3 treatments over 12 days showed complete tumor regression. At the highest dose tested, there were no obvious symptoms of toxicity. After 60 days of observation, the tumor did not regrow.
|
||||
| In Vivo Model | PC-3 mouse xenograft with prostate cancer | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
75%
|
|||
| Administration Time | 54 days | ||||
| Administration Dosage | 0.6 mg/kg | ||||
| Evaluation Method | Tumor volume detection | ||||
| Description |
In xenografted prostate cancer in mice, only 3 treatments over 12 days showed complete tumor regression. At the highest dose tested, there were no obvious symptoms of toxicity. After 60 days of observation, the tumor did not regrow.
|
||||
| In Vivo Model | PC-3 mouse xenograft with prostate cancer | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
100%
|
|||
| Administration Time | 54 days | ||||
| Administration Dosage | 1.2 mg/kg | ||||
| Evaluation Method | Tumor volume detection | ||||
| Description |
In xenografted prostate cancer in mice, only 3 treatments over 12 days showed complete tumor regression. At the highest dose tested, there were no obvious symptoms of toxicity. After 60 days of observation, the tumor did not regrow.
|
||||
| In Vivo Model | PC-3 mouse xenograft with prostate cancer | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
100%
|
|||
| Administration Time | 54 days | ||||
| Administration Dosage | 2.4 mg/kg | ||||
| Evaluation Method | Tumor volume detection | ||||
| Description |
In xenografted prostate cancer in mice, only 3 treatments over 12 days showed complete tumor regression. At the highest dose tested, there were no obvious symptoms of toxicity. After 60 days of observation, the tumor did not regrow.
|
||||
| In Vivo Model | PC-3 mouse xenograft with prostate cancer | ||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
2.3 nM
|
|||
| Description |
In xenografted prostate cancer in mice, only 3 treatments over 12 days showed complete tumor regression. At the highest dose tested, there were no obvious symptoms of toxicity. After 60 days of observation, the tumor did not regrow.
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | BxPC-3 cell | CVCL_0186 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
2.4 nM
|
|||
| Description |
In xenografted prostate cancer in mice, only 3 treatments over 12 days showed complete tumor regression. At the highest dose tested, there were no obvious symptoms of toxicity. After 60 days of observation, the tumor did not regrow.
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
2.6 nM
|
|||
| Description |
In xenografted prostate cancer in mice, only 3 treatments over 12 days showed complete tumor regression. At the highest dose tested, there were no obvious symptoms of toxicity. After 60 days of observation, the tumor did not regrow.
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | MIA PaCa-2 cell | CVCL_0428 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uterine corpus sarcoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
5.7 nM
|
|||
| Description |
In xenografted prostate cancer in mice, only 3 treatments over 12 days showed complete tumor regression. At the highest dose tested, there were no obvious symptoms of toxicity. After 60 days of observation, the tumor did not regrow.
|
||||
| In Vitro Model | Uterine corpus sarcoma | MES-SA cell | CVCL_1404 | ||
