Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_02033
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
TP2-cCPP-CBT
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 3 Indication(s)
|
|||||
| Structure |
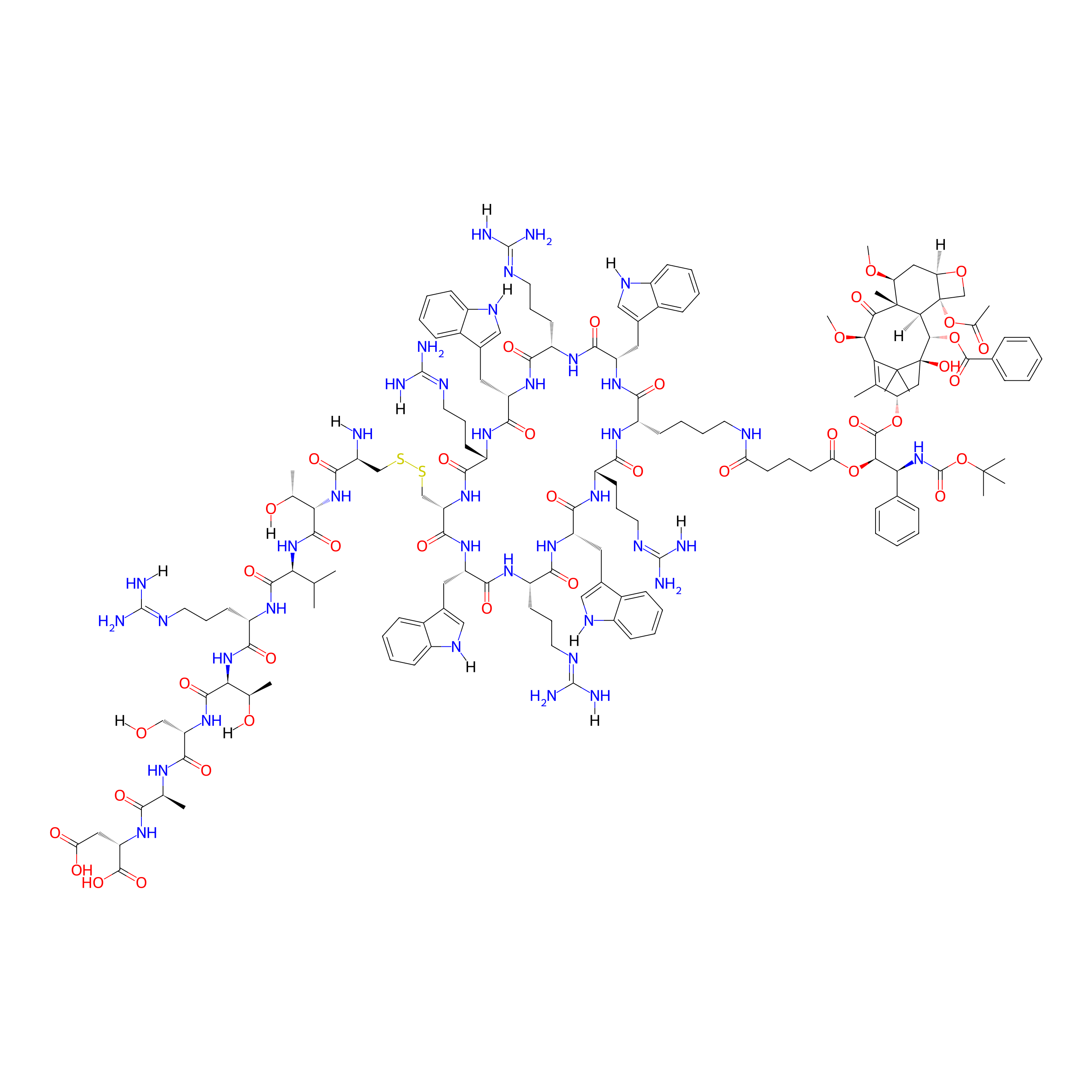
|
|||||
| Peptide Name |
TP2-cCPP
|
Peptide Info | ||||
| Drug Name |
Cabazitaxel
|
Drug Info | ||||
| Therapeutic Target |
Microtubule (MT)
|
Target Info | ||||
| Linker Name |
Glutaric acid
|
Linker Info | ||||
| Peptide Modified Type |
Cyclization modification; The modification of binding with chemical macromolecules
|
|||||
| Modified Segment |
Head-to-tail cyclization, CTVRTSAD
|
|||||
| Formula |
C159H221N39O40S2
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 3382.884 | ||||
| Lipid-water partition coefficient (xlogp) | -3.3075 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 40 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 46 | |||||
| Rotatable Bond Count (rotbonds) | 78 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
162.4 ± 3.2 nM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTS assay | ||||
| MOA of PDC |
The present study explores two different targeting peptides (TPs), RGDC (TP1), and CTVRTSAD (TP2), toward avβ1 integrin and EDB-Fn, respectively. TP1 or TP2 is covalently conjugated via disulfide bond to [C(WR)2K(WR)2]-(CBT) (6) to provide CBT conjugates (11, 16) containing an active targeting ability with increased aqueous solubility without decreasing cellular uptake. Targeting peptides may enable cCPP-CBT to target biomarker overexpressing tumor cells, and cCPP can assist in improving the physicochemical properties of CBT and delivery into the cancer cells. The conjugates in vitro stability was examined in various relevant conditions to mimic the cancerous versus normal physiological environment. Conjugates were evaluated in human plasma at two different glutathione concentrations (GSH, 1 mM and 10 mM) and two different pHs (6.5 and 7.4). The reported GSH level in noncancerous conditions is below 1 mM and approximately 10 mM at the cancer site. The in vitro antiproliferative assay using a biomarker overexpressing cell lines was compared to those without or with moderately expressing cancer cell lines. The study results demonstrate that targeting enhances the efficacy of CBT on cancer cell lines overexpressing extracellular biomarkers.
Click to Show/Hide
|
||||
| Description |
The conjugates therapeutic efficacy with targeting moieties was evaluated using prostate cancer cell lines with and without EDB-Fn overexpression (C4-2 and DU-145). Breast cancer cell lines with and without integrin v1 overexpression (MCF-7 and MDA-MB-231) were also used to evaluate antiproliferative activity. Noncancerous embryonic kidney epithelial cell line (HEK-293) was used as a control since this cell line did not express both biomarkers. All the compounds with and without targeting moieties showed comparable or lower antiproliferative activity than the CBT alone after 72 h incubation. The antiproliferative activity of compound 11 on MCF-7 (IC50 = 160.4 nM) was 1.6 times greater than on MDA-MB-231 (IC50 = 251.6 nM), showing a slight improvement in antiproliferative activity. A similar observation was found with compound 16 that showed 1.45 times higher antiproliferative activity on MCF-7 (IC50 = 162.4 nM) as compared to MDA-MB-231 cells (IC50 = 235.0 nM). Both MCF-7 and MDA-MB-231 are breast cancer cell lines. MDA-MB-231 is triple-negative breast cancer cells, and it showed higher IC50 values for CBT than MCF-7 cells. Later, the antiproliferative activity was compared between MCF-7 and the control cells (HEK-293); it was observed that compound 11 showed 3.33 times higher antiproliferative activity on MCF-7 cells (IC50 for 11 = 160.4 nM) compared to HEK-293 (IC50 for 11 = 534.5 nM), and compound 16 showed 4.6 times higher antiproliferative activity on MCF-7 (IC50 for 16 = 162.4 nM) compared to HEK-293 (IC50 for 16 = 483.3 nM). Thereafter, we wanted to assess the effectiveness of the EDB-Fn targeting moiety by comparing the antiproliferative activities on prostate cancer cell lines with and without EDB-Fn overexpression. The data showed a similar trend to that observed in breast cancer cells. For instance, compound 11 showed 3.4 times higher antiproliferative activity on C4-2 (IC50 for 11 = 181.4 nM) compared to DU-145 (IC50 for 11 = 611.2 nM). Similarly, compound 16 showed 3.7 times higher antiproliferative activity on C4-2 (IC50 for 16 = 202.2 nM) compared to DU-145 (IC50 for 16 = 751.1 nM). Compound 16 (possessing EDB-Fn targeting moiety) showed 1.1 times less antiproliferative activity compared to compound 6 (cCPP-CBT, without targeting moiety) on C4-2 cell lines, but compound 16 showed a much higher difference (3.6 times) in antiproliferative activity when compared with compound 6 on EDB-Fn moderately expressing DU-145 cell line. This data is further corroborated in EDB-Fn nonexpressing HEK-293. For instance, compound 16 showed 4.6 times less antiproliferative activity when compared with compound 6 on HEK-293 cell line. It can be inferred from the data (Table 2) that the EDB-Fn targeting conjugate was significantly effective on EDB-Fn overexpressing cells, while the conjugate has minimal effect on nonexpressing or moderately expressing cells. In conclusion, it is plausible to state that the presence of conjugating moieties on compound 11 (integrin targeting) and compound 16 (EDB-Fn targeting) significantly affects their ability to adhere and deliver the chemotherapeutic agent selectively to biomarker overexpressing cancer cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate carcinoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
202.2 ± 7.9 nM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTS assay | ||||
| MOA of PDC |
The present study explores two different targeting peptides (TPs), RGDC (TP1), and CTVRTSAD (TP2), toward avβ1 integrin and EDB-Fn, respectively. TP1 or TP2 is covalently conjugated via disulfide bond to [C(WR)2K(WR)2]-(CBT) (6) to provide CBT conjugates (11, 16) containing an active targeting ability with increased aqueous solubility without decreasing cellular uptake. Targeting peptides may enable cCPP-CBT to target biomarker overexpressing tumor cells, and cCPP can assist in improving the physicochemical properties of CBT and delivery into the cancer cells. The conjugates in vitro stability was examined in various relevant conditions to mimic the cancerous versus normal physiological environment. Conjugates were evaluated in human plasma at two different glutathione concentrations (GSH, 1 mM and 10 mM) and two different pHs (6.5 and 7.4). The reported GSH level in noncancerous conditions is below 1 mM and approximately 10 mM at the cancer site. The in vitro antiproliferative assay using a biomarker overexpressing cell lines was compared to those without or with moderately expressing cancer cell lines. The study results demonstrate that targeting enhances the efficacy of CBT on cancer cell lines overexpressing extracellular biomarkers.
Click to Show/Hide
|
||||
| Description |
The conjugates therapeutic efficacy with targeting moieties was evaluated using prostate cancer cell lines with and without EDB-Fn overexpression (C4-2 and DU-145). Breast cancer cell lines with and without integrin v1 overexpression (MCF-7 and MDA-MB-231) were also used to evaluate antiproliferative activity. Noncancerous embryonic kidney epithelial cell line (HEK-293) was used as a control since this cell line did not express both biomarkers. All the compounds with and without targeting moieties showed comparable or lower antiproliferative activity than the CBT alone after 72 h incubation. The antiproliferative activity of compound 11 on MCF-7 (IC50 = 160.4 nM) was 1.6 times greater than on MDA-MB-231 (IC50 = 251.6 nM), showing a slight improvement in antiproliferative activity. A similar observation was found with compound 16 that showed 1.45 times higher antiproliferative activity on MCF-7 (IC50 = 162.4 nM) as compared to MDA-MB-231 cells (IC50 = 235.0 nM). Both MCF-7 and MDA-MB-231 are breast cancer cell lines. MDA-MB-231 is triple-negative breast cancer cells, and it showed higher IC50 values for CBT than MCF-7 cells. Later, the antiproliferative activity was compared between MCF-7 and the control cells (HEK-293); it was observed that compound 11 showed 3.33 times higher antiproliferative activity on MCF-7 cells (IC50 for 11 = 160.4 nM) compared to HEK-293 (IC50 for 11 = 534.5 nM), and compound 16 showed 4.6 times higher antiproliferative activity on MCF-7 (IC50 for 16 = 162.4 nM) compared to HEK-293 (IC50 for 16 = 483.3 nM). Thereafter, we wanted to assess the effectiveness of the EDB-Fn targeting moiety by comparing the antiproliferative activities on prostate cancer cell lines with and without EDB-Fn overexpression. The data showed a similar trend to that observed in breast cancer cells. For instance, compound 11 showed 3.4 times higher antiproliferative activity on C4-2 (IC50 for 11 = 181.4 nM) compared to DU-145 (IC50 for 11 = 611.2 nM). Similarly, compound 16 showed 3.7 times higher antiproliferative activity on C4-2 (IC50 for 16 = 202.2 nM) compared to DU-145 (IC50 for 16 = 751.1 nM). Compound 16 (possessing EDB-Fn targeting moiety) showed 1.1 times less antiproliferative activity compared to compound 6 (cCPP-CBT, without targeting moiety) on C4-2 cell lines, but compound 16 showed a much higher difference (3.6 times) in antiproliferative activity when compared with compound 6 on EDB-Fn moderately expressing DU-145 cell line. This data is further corroborated in EDB-Fn nonexpressing HEK-293. For instance, compound 16 showed 4.6 times less antiproliferative activity when compared with compound 6 on HEK-293 cell line. It can be inferred from the data (Table 2) that the EDB-Fn targeting conjugate was significantly effective on EDB-Fn overexpressing cells, while the conjugate has minimal effect on nonexpressing or moderately expressing cells. In conclusion, it is plausible to state that the presence of conjugating moieties on compound 11 (integrin targeting) and compound 16 (EDB-Fn targeting) significantly affects their ability to adhere and deliver the chemotherapeutic agent selectively to biomarker overexpressing cancer cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP C4-2 cell | CVCL_4782 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast adenocarcinoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
235 ± 11.4 nM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTS assay | ||||
| MOA of PDC |
The present study explores two different targeting peptides (TPs), RGDC (TP1), and CTVRTSAD (TP2), toward avβ1 integrin and EDB-Fn, respectively. TP1 or TP2 is covalently conjugated via disulfide bond to [C(WR)2K(WR)2]-(CBT) (6) to provide CBT conjugates (11, 16) containing an active targeting ability with increased aqueous solubility without decreasing cellular uptake. Targeting peptides may enable cCPP-CBT to target biomarker overexpressing tumor cells, and cCPP can assist in improving the physicochemical properties of CBT and delivery into the cancer cells. The conjugates in vitro stability was examined in various relevant conditions to mimic the cancerous versus normal physiological environment. Conjugates were evaluated in human plasma at two different glutathione concentrations (GSH, 1 mM and 10 mM) and two different pHs (6.5 and 7.4). The reported GSH level in noncancerous conditions is below 1 mM and approximately 10 mM at the cancer site. The in vitro antiproliferative assay using a biomarker overexpressing cell lines was compared to those without or with moderately expressing cancer cell lines. The study results demonstrate that targeting enhances the efficacy of CBT on cancer cell lines overexpressing extracellular biomarkers.
Click to Show/Hide
|
||||
| Description |
The conjugates therapeutic efficacy with targeting moieties was evaluated using prostate cancer cell lines with and without EDB-Fn overexpression (C4-2 and DU-145). Breast cancer cell lines with and without integrin v1 overexpression (MCF-7 and MDA-MB-231) were also used to evaluate antiproliferative activity. Noncancerous embryonic kidney epithelial cell line (HEK-293) was used as a control since this cell line did not express both biomarkers. All the compounds with and without targeting moieties showed comparable or lower antiproliferative activity than the CBT alone after 72 h incubation. The antiproliferative activity of compound 11 on MCF-7 (IC50 = 160.4 nM) was 1.6 times greater than on MDA-MB-231 (IC50 = 251.6 nM), showing a slight improvement in antiproliferative activity. A similar observation was found with compound 16 that showed 1.45 times higher antiproliferative activity on MCF-7 (IC50 = 162.4 nM) as compared to MDA-MB-231 cells (IC50 = 235.0 nM). Both MCF-7 and MDA-MB-231 are breast cancer cell lines. MDA-MB-231 is triple-negative breast cancer cells, and it showed higher IC50 values for CBT than MCF-7 cells. Later, the antiproliferative activity was compared between MCF-7 and the control cells (HEK-293); it was observed that compound 11 showed 3.33 times higher antiproliferative activity on MCF-7 cells (IC50 for 11 = 160.4 nM) compared to HEK-293 (IC50 for 11 = 534.5 nM), and compound 16 showed 4.6 times higher antiproliferative activity on MCF-7 (IC50 for 16 = 162.4 nM) compared to HEK-293 (IC50 for 16 = 483.3 nM). Thereafter, we wanted to assess the effectiveness of the EDB-Fn targeting moiety by comparing the antiproliferative activities on prostate cancer cell lines with and without EDB-Fn overexpression. The data showed a similar trend to that observed in breast cancer cells. For instance, compound 11 showed 3.4 times higher antiproliferative activity on C4-2 (IC50 for 11 = 181.4 nM) compared to DU-145 (IC50 for 11 = 611.2 nM). Similarly, compound 16 showed 3.7 times higher antiproliferative activity on C4-2 (IC50 for 16 = 202.2 nM) compared to DU-145 (IC50 for 16 = 751.1 nM). Compound 16 (possessing EDB-Fn targeting moiety) showed 1.1 times less antiproliferative activity compared to compound 6 (cCPP-CBT, without targeting moiety) on C4-2 cell lines, but compound 16 showed a much higher difference (3.6 times) in antiproliferative activity when compared with compound 6 on EDB-Fn moderately expressing DU-145 cell line. This data is further corroborated in EDB-Fn nonexpressing HEK-293. For instance, compound 16 showed 4.6 times less antiproliferative activity when compared with compound 6 on HEK-293 cell line. It can be inferred from the data (Table 2) that the EDB-Fn targeting conjugate was significantly effective on EDB-Fn overexpressing cells, while the conjugate has minimal effect on nonexpressing or moderately expressing cells. In conclusion, it is plausible to state that the presence of conjugating moieties on compound 11 (integrin targeting) and compound 16 (EDB-Fn targeting) significantly affects their ability to adhere and deliver the chemotherapeutic agent selectively to biomarker overexpressing cancer cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
483.3 ± 17.2 nM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTS assay | ||||
| MOA of PDC |
The present study explores two different targeting peptides (TPs), RGDC (TP1), and CTVRTSAD (TP2), toward avβ1 integrin and EDB-Fn, respectively. TP1 or TP2 is covalently conjugated via disulfide bond to [C(WR)2K(WR)2]-(CBT) (6) to provide CBT conjugates (11, 16) containing an active targeting ability with increased aqueous solubility without decreasing cellular uptake. Targeting peptides may enable cCPP-CBT to target biomarker overexpressing tumor cells, and cCPP can assist in improving the physicochemical properties of CBT and delivery into the cancer cells. The conjugates in vitro stability was examined in various relevant conditions to mimic the cancerous versus normal physiological environment. Conjugates were evaluated in human plasma at two different glutathione concentrations (GSH, 1 mM and 10 mM) and two different pHs (6.5 and 7.4). The reported GSH level in noncancerous conditions is below 1 mM and approximately 10 mM at the cancer site. The in vitro antiproliferative assay using a biomarker overexpressing cell lines was compared to those without or with moderately expressing cancer cell lines. The study results demonstrate that targeting enhances the efficacy of CBT on cancer cell lines overexpressing extracellular biomarkers.
Click to Show/Hide
|
||||
| Description |
The conjugates therapeutic efficacy with targeting moieties was evaluated using prostate cancer cell lines with and without EDB-Fn overexpression (C4-2 and DU-145). Breast cancer cell lines with and without integrin v1 overexpression (MCF-7 and MDA-MB-231) were also used to evaluate antiproliferative activity. Noncancerous embryonic kidney epithelial cell line (HEK-293) was used as a control since this cell line did not express both biomarkers. All the compounds with and without targeting moieties showed comparable or lower antiproliferative activity than the CBT alone after 72 h incubation. The antiproliferative activity of compound 11 on MCF-7 (IC50 = 160.4 nM) was 1.6 times greater than on MDA-MB-231 (IC50 = 251.6 nM), showing a slight improvement in antiproliferative activity. A similar observation was found with compound 16 that showed 1.45 times higher antiproliferative activity on MCF-7 (IC50 = 162.4 nM) as compared to MDA-MB-231 cells (IC50 = 235.0 nM). Both MCF-7 and MDA-MB-231 are breast cancer cell lines. MDA-MB-231 is triple-negative breast cancer cells, and it showed higher IC50 values for CBT than MCF-7 cells. Later, the antiproliferative activity was compared between MCF-7 and the control cells (HEK-293); it was observed that compound 11 showed 3.33 times higher antiproliferative activity on MCF-7 cells (IC50 for 11 = 160.4 nM) compared to HEK-293 (IC50 for 11 = 534.5 nM), and compound 16 showed 4.6 times higher antiproliferative activity on MCF-7 (IC50 for 16 = 162.4 nM) compared to HEK-293 (IC50 for 16 = 483.3 nM). Thereafter, we wanted to assess the effectiveness of the EDB-Fn targeting moiety by comparing the antiproliferative activities on prostate cancer cell lines with and without EDB-Fn overexpression. The data showed a similar trend to that observed in breast cancer cells. For instance, compound 11 showed 3.4 times higher antiproliferative activity on C4-2 (IC50 for 11 = 181.4 nM) compared to DU-145 (IC50 for 11 = 611.2 nM). Similarly, compound 16 showed 3.7 times higher antiproliferative activity on C4-2 (IC50 for 16 = 202.2 nM) compared to DU-145 (IC50 for 16 = 751.1 nM). Compound 16 (possessing EDB-Fn targeting moiety) showed 1.1 times less antiproliferative activity compared to compound 6 (cCPP-CBT, without targeting moiety) on C4-2 cell lines, but compound 16 showed a much higher difference (3.6 times) in antiproliferative activity when compared with compound 6 on EDB-Fn moderately expressing DU-145 cell line. This data is further corroborated in EDB-Fn nonexpressing HEK-293. For instance, compound 16 showed 4.6 times less antiproliferative activity when compared with compound 6 on HEK-293 cell line. It can be inferred from the data (Table 2) that the EDB-Fn targeting conjugate was significantly effective on EDB-Fn overexpressing cells, while the conjugate has minimal effect on nonexpressing or moderately expressing cells. In conclusion, it is plausible to state that the presence of conjugating moieties on compound 11 (integrin targeting) and compound 16 (EDB-Fn targeting) significantly affects their ability to adhere and deliver the chemotherapeutic agent selectively to biomarker overexpressing cancer cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | HEK293 cell | CVCL_0045 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate carcinoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
751.1 ± 27.2 nM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTS assay | ||||
| MOA of PDC |
The present study explores two different targeting peptides (TPs), RGDC (TP1), and CTVRTSAD (TP2), toward avβ1 integrin and EDB-Fn, respectively. TP1 or TP2 is covalently conjugated via disulfide bond to [C(WR)2K(WR)2]-(CBT) (6) to provide CBT conjugates (11, 16) containing an active targeting ability with increased aqueous solubility without decreasing cellular uptake. Targeting peptides may enable cCPP-CBT to target biomarker overexpressing tumor cells, and cCPP can assist in improving the physicochemical properties of CBT and delivery into the cancer cells. The conjugates in vitro stability was examined in various relevant conditions to mimic the cancerous versus normal physiological environment. Conjugates were evaluated in human plasma at two different glutathione concentrations (GSH, 1 mM and 10 mM) and two different pHs (6.5 and 7.4). The reported GSH level in noncancerous conditions is below 1 mM and approximately 10 mM at the cancer site. The in vitro antiproliferative assay using a biomarker overexpressing cell lines was compared to those without or with moderately expressing cancer cell lines. The study results demonstrate that targeting enhances the efficacy of CBT on cancer cell lines overexpressing extracellular biomarkers.
Click to Show/Hide
|
||||
| Description |
The conjugates therapeutic efficacy with targeting moieties was evaluated using prostate cancer cell lines with and without EDB-Fn overexpression (C4-2 and DU-145). Breast cancer cell lines with and without integrin v1 overexpression (MCF-7 and MDA-MB-231) were also used to evaluate antiproliferative activity. Noncancerous embryonic kidney epithelial cell line (HEK-293) was used as a control since this cell line did not express both biomarkers. All the compounds with and without targeting moieties showed comparable or lower antiproliferative activity than the CBT alone after 72 h incubation. The antiproliferative activity of compound 11 on MCF-7 (IC50 = 160.4 nM) was 1.6 times greater than on MDA-MB-231 (IC50 = 251.6 nM), showing a slight improvement in antiproliferative activity. A similar observation was found with compound 16 that showed 1.45 times higher antiproliferative activity on MCF-7 (IC50 = 162.4 nM) as compared to MDA-MB-231 cells (IC50 = 235.0 nM). Both MCF-7 and MDA-MB-231 are breast cancer cell lines. MDA-MB-231 is triple-negative breast cancer cells, and it showed higher IC50 values for CBT than MCF-7 cells. Later, the antiproliferative activity was compared between MCF-7 and the control cells (HEK-293); it was observed that compound 11 showed 3.33 times higher antiproliferative activity on MCF-7 cells (IC50 for 11 = 160.4 nM) compared to HEK-293 (IC50 for 11 = 534.5 nM), and compound 16 showed 4.6 times higher antiproliferative activity on MCF-7 (IC50 for 16 = 162.4 nM) compared to HEK-293 (IC50 for 16 = 483.3 nM). Thereafter, we wanted to assess the effectiveness of the EDB-Fn targeting moiety by comparing the antiproliferative activities on prostate cancer cell lines with and without EDB-Fn overexpression. The data showed a similar trend to that observed in breast cancer cells. For instance, compound 11 showed 3.4 times higher antiproliferative activity on C4-2 (IC50 for 11 = 181.4 nM) compared to DU-145 (IC50 for 11 = 611.2 nM). Similarly, compound 16 showed 3.7 times higher antiproliferative activity on C4-2 (IC50 for 16 = 202.2 nM) compared to DU-145 (IC50 for 16 = 751.1 nM). Compound 16 (possessing EDB-Fn targeting moiety) showed 1.1 times less antiproliferative activity compared to compound 6 (cCPP-CBT, without targeting moiety) on C4-2 cell lines, but compound 16 showed a much higher difference (3.6 times) in antiproliferative activity when compared with compound 6 on EDB-Fn moderately expressing DU-145 cell line. This data is further corroborated in EDB-Fn nonexpressing HEK-293. For instance, compound 16 showed 4.6 times less antiproliferative activity when compared with compound 6 on HEK-293 cell line. It can be inferred from the data (Table 2) that the EDB-Fn targeting conjugate was significantly effective on EDB-Fn overexpressing cells, while the conjugate has minimal effect on nonexpressing or moderately expressing cells. In conclusion, it is plausible to state that the presence of conjugating moieties on compound 11 (integrin targeting) and compound 16 (EDB-Fn targeting) significantly affects their ability to adhere and deliver the chemotherapeutic agent selectively to biomarker overexpressing cancer cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | DU145 cell | CVCL_0105 | ||
References
