Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_02071
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
SMAC-FRRG-DOX
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
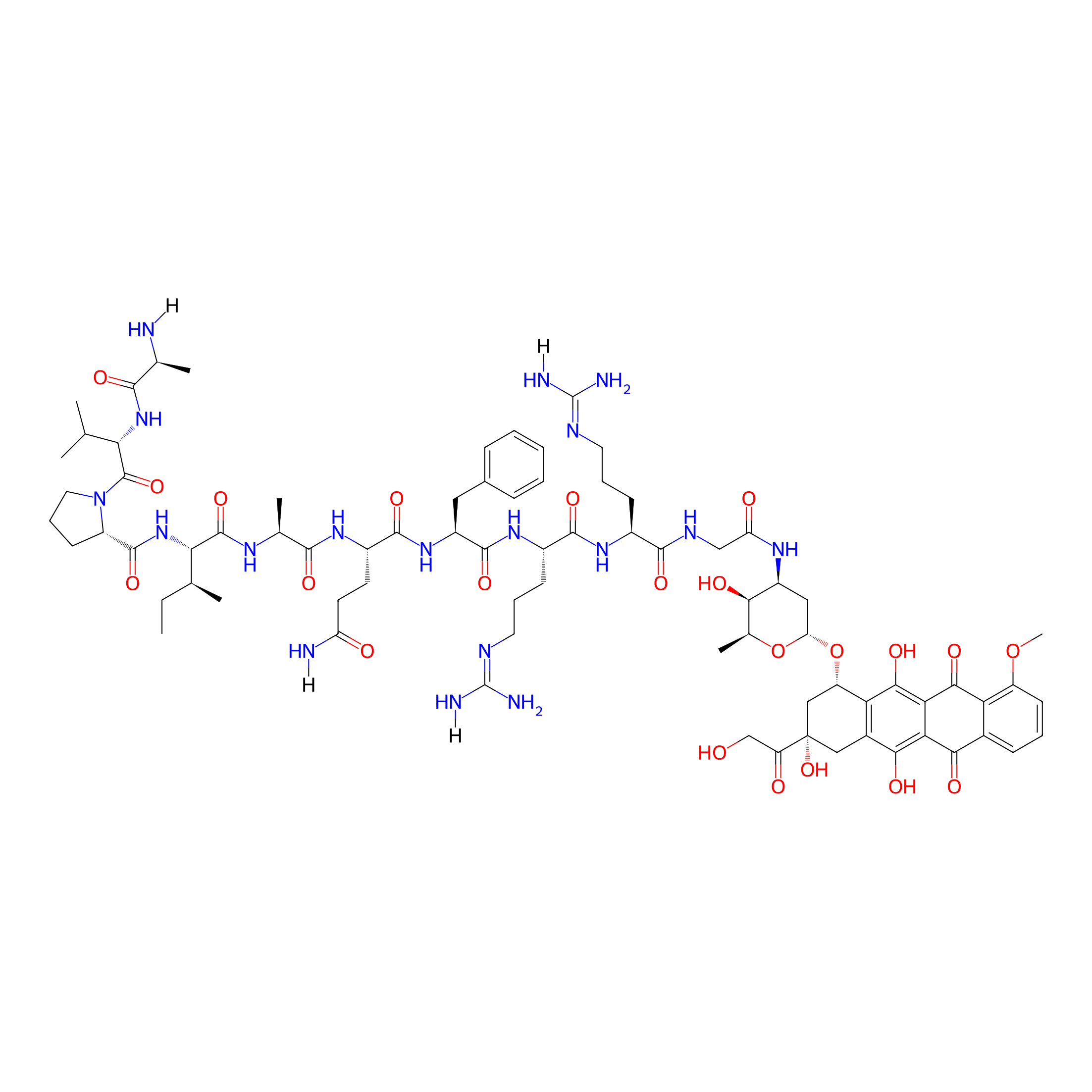
|
|||||
| Peptide Name |
SMAC-FRRG
|
Peptide Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
Amide bond
|
Linker Info | ||||
| Formula |
C77H110N18O22
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 1639.831 | ||||
| Lipid-water partition coefficient (xlogp) | -4.6558 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 20 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 25 | |||||
| Rotatable Bond Count (rotbonds) | 41 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Drug-resistant cancer | ||||
| Efficacy Data | Body weight |
23.5g
|
|||
| Administration Time | 13 days | ||||
| MOA of PDC |
These results suggest that the DD-NPs could improve the in vivo bioavailability and cancer targeting efficiency of SMAC via their stable nanoparticle-derived EPR effect, leading to effective IAPs inhibition in targeted tumor tissues.
|
||||
| In Vivo Model | MCF-7 tumor-bearing mice. | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Drug-resistant cancer | ||||
| Efficacy Data | Tumer volume |
10 mm3
|
|||
| Administration Time | 21 days | ||||
| MOA of PDC |
These results suggest that the DD-NPs could improve the in vivo bioavailability and cancer targeting efficiency of SMAC via their stable nanoparticle-derived EPR effect, leading to effective IAPs inhibition in targeted tumor tissues.
|
||||
| In Vivo Model | MCF-7 tumor-bearing mice. | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Drug-resistant cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
76.00%
|
|||
| Administration Dosage | 1 mg/kg of DOX | ||||
| MOA of PDC |
These results suggest that the DD-NPs could improve the in vivo bioavailability and cancer targeting efficiency of SMAC via their stable nanoparticle-derived EPR effect, leading to effective IAPs inhibition in targeted tumor tissues.
|
||||
| Description |
Finally, the tumor volumes were successfully suppressed from 76% to 89% when the intravenous dose of DD-NPs increased from 1 mg/kg of DOX to 5 mg/kg of DOX.
|
||||
| In Vivo Model | MCF-7 tumor-bearing mice. | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Drug-resistant cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
89%-99%
|
|||
| Administration Dosage | 5 mg/kg | ||||
| MOA of PDC |
These results suggest that the DD-NPs could improve the in vivo bioavailability and cancer targeting efficiency of SMAC via their stable nanoparticle-derived EPR effect, leading to effective IAPs inhibition in targeted tumor tissues.
|
||||
| Description |
Finally, the tumor volumes were successfully suppressed from 76% to 89% when the intravenous dose of DD-NPs increased from 1 mg/kg of DOX to 5 mg/kg of DOX.
|
||||
| In Vivo Model | MCF-7 tumor-bearing mice. | ||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Drug-resistant cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
7.53 μM
|
|||
| MOA of PDC |
These results suggest that the DD-NPs could improve the in vivo bioavailability and cancer targeting efficiency of SMAC via their stable nanoparticle-derived EPR effect, leading to effective IAPs inhibition in targeted tumor tissues.
|
||||
| Description |
When both cells were treated with different concentrations of DD-NPs (0-186 μg/ml), the cytotoxicity was induced only in MCF-7, showing similar cytotoxicity with free DOX at a high concentration (Fig. 2h). However, DD-NPs did not exhibit significant cytotoxicity in H9C2 whereas free DOX showed similar cytotoxicity when treated to MCF-7 (Fig. 2i).
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Drug-resistant cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
9.2 μM
|
|||
| Administration Time | 48 h | ||||
| MOA of PDC |
These results suggest that the DD-NPs could improve the in vivo bioavailability and cancer targeting efficiency of SMAC via their stable nanoparticle-derived EPR effect, leading to effective IAPs inhibition in targeted tumor tissues.
|
||||
| Description |
However, DD-NP-treated group showed similar IC50 in wild-MCF-7 (7.53 μM) and ADR-MCF-7 (9.2 μM), due to the synergetic pro-apoptotic effect of SMAC molecules in DD-NPs (Fig. 3g).
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7/ADR cell | CVCL_0031 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Drug-resistant cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | >> 200 μM | |||
| MOA of PDC |
These results suggest that the DD-NPs could improve the in vivo bioavailability and cancer targeting efficiency of SMAC via their stable nanoparticle-derived EPR effect, leading to effective IAPs inhibition in targeted tumor tissues.
|
||||
| Description |
When both cells were treated with different concentrations of DD-NPs (0-186 μg/ml), the cytotoxicity was induced only in MCF-7, showing similar cytotoxicity with free DOX at a high concentration (Fig. 2h). However, DD-NPs did not exhibit significant cytotoxicity in H9C2 whereas free DOX showed similar cytotoxicity when treated to MCF-7 (Fig. 2i).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | H9c2 cell | CVCL_0286 | ||
References
