Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_02073
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
diCPTiRGD
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
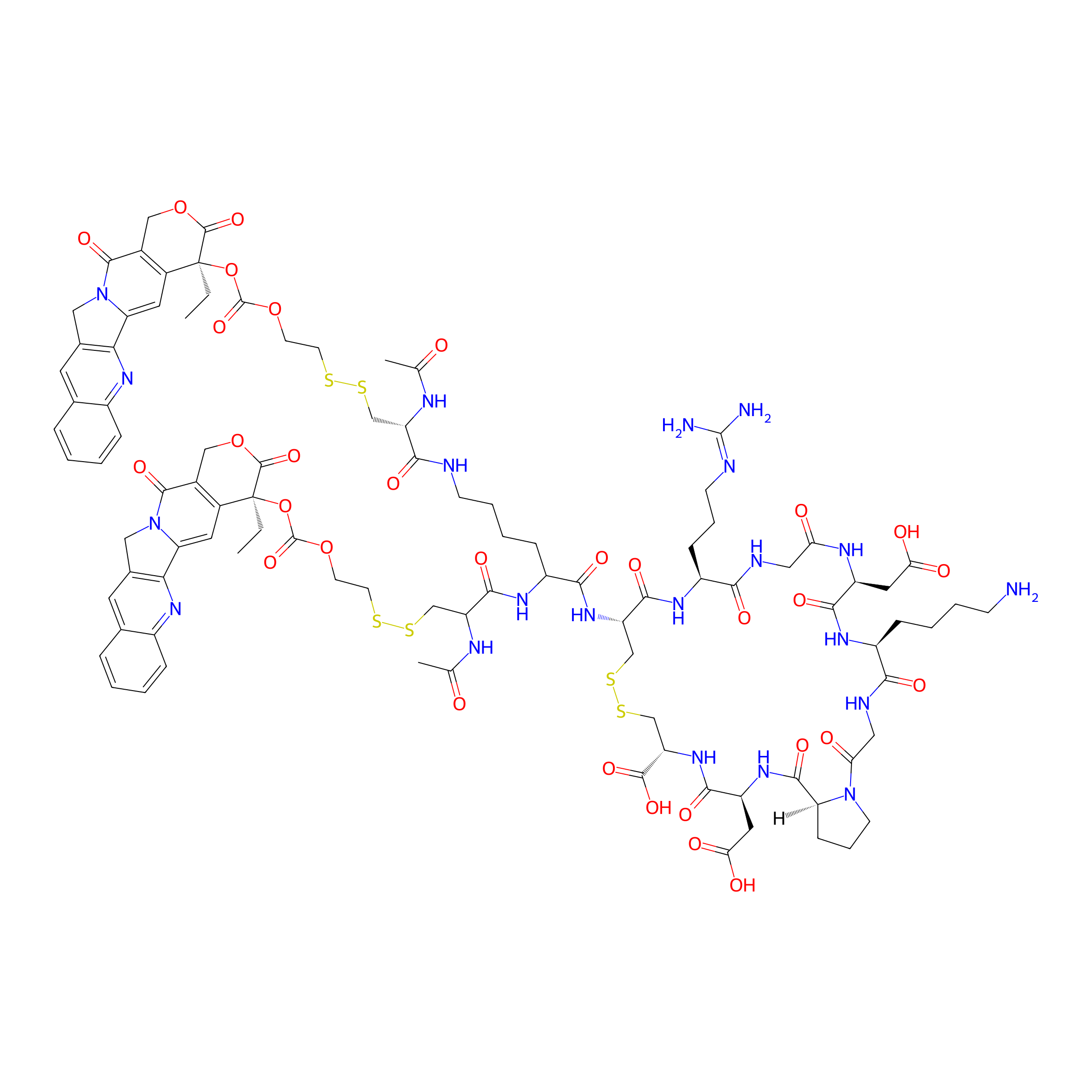
|
|||||
| Peptide Name |
iRGD
|
Peptide Info | ||||
| Drug Name |
Camptothecin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 1 (TOP1)
|
Target Info | ||||
| Linker Name |
2-(Disulfanyl)ethyl hydrogen carbonate
|
Linker Info | ||||
| Peptide Modified Type |
Cyclization modification
|
|||||
| Modified Segment |
Head-to-tail cyclization
|
|||||
| Formula |
C97H119N21O31S6
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 2267.537 | ||||
| Lipid-water partition coefficient (xlogp) | -0.2181 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 18 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 40 | |||||
| Rotatable Bond Count (rotbonds) | 41 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Survival rate |
100.00%
|
|||
| Administration Time | 30 days | ||||
| MOA of PDC |
In this context, we developed a drug-bearing supramolecular hydrogel system to intratumourally (i.t.) deliver CDNs against malignant tumours to achieve cancer chemoimmunotherapy. Our strategy was to chemically conjugate the hydrophilic pep-tide moiety iRGD (a tumour-penetrating peptide that can bind to neuropilin-1 (NRP-1) and trigger tumour tissue penetration) to the hydrophobic anticancer drug CPT to form a self-assembling and self-formulating peptide-drug conjugate (diCPT-iRGD). In aqueous solution, the designed drug amphiphile spontaneously assembles into supramolecular nanotubes (NTs). The negatively charged STING agonist (c-di-AMP (CDA)) can condense on the surface of these positively charged NTs through electrostatic complexations. After injection into the tumour site, the CDA-NT solution can immediately form a hydrogel, functioning as a local reservoir for extended localized release of CDA and CPT to awaken both the innate and adaptive immune systems.
Click to Show/Hide
|
||||
| Description |
When using the diCPT-iRGD NT hydrogel, the CDA-NT treatment led to substantial tumour regression (Fig. 3c-e) and demonstrated a 100% survival rate in mice (Fig. 3f).
|
||||
| In Vivo Model | GL-261 brain cancer C57BL/6 mice. | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Tumer volume |
0 mm3
|
|||
| Administration Time | 30 days | ||||
| MOA of PDC |
In this context, we developed a drug-bearing supramolecular hydrogel system to intratumourally (i.t.) deliver CDNs against malignant tumours to achieve cancer chemoimmunotherapy. Our strategy was to chemically conjugate the hydrophilic pep-tide moiety iRGD (a tumour-penetrating peptide that can bind to neuropilin-1 (NRP-1) and trigger tumour tissue penetration) to the hydrophobic anticancer drug CPT to form a self-assembling and self-formulating peptide-drug conjugate (diCPT-iRGD). In aqueous solution, the designed drug amphiphile spontaneously assembles into supramolecular nanotubes (NTs). The negatively charged STING agonist (c-di-AMP (CDA)) can condense on the surface of these positively charged NTs through electrostatic complexations. After injection into the tumour site, the CDA-NT solution can immediately form a hydrogel, functioning as a local reservoir for extended localized release of CDA and CPT to awaken both the innate and adaptive immune systems.
Click to Show/Hide
|
||||
| Description |
When using the diCPT-iRGD NT hydrogel, the CDA-NT treatment led to substantial tumour regression (Fig. 3c-e) and demonstrated a 100% survival rate in mice (Fig. 3f).
|
||||
| In Vivo Model | GL-261 brain cancer C57BL/6 mice. | ||||
References
