Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00124
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
c(RGDyK)
|
|||||
| Structure |
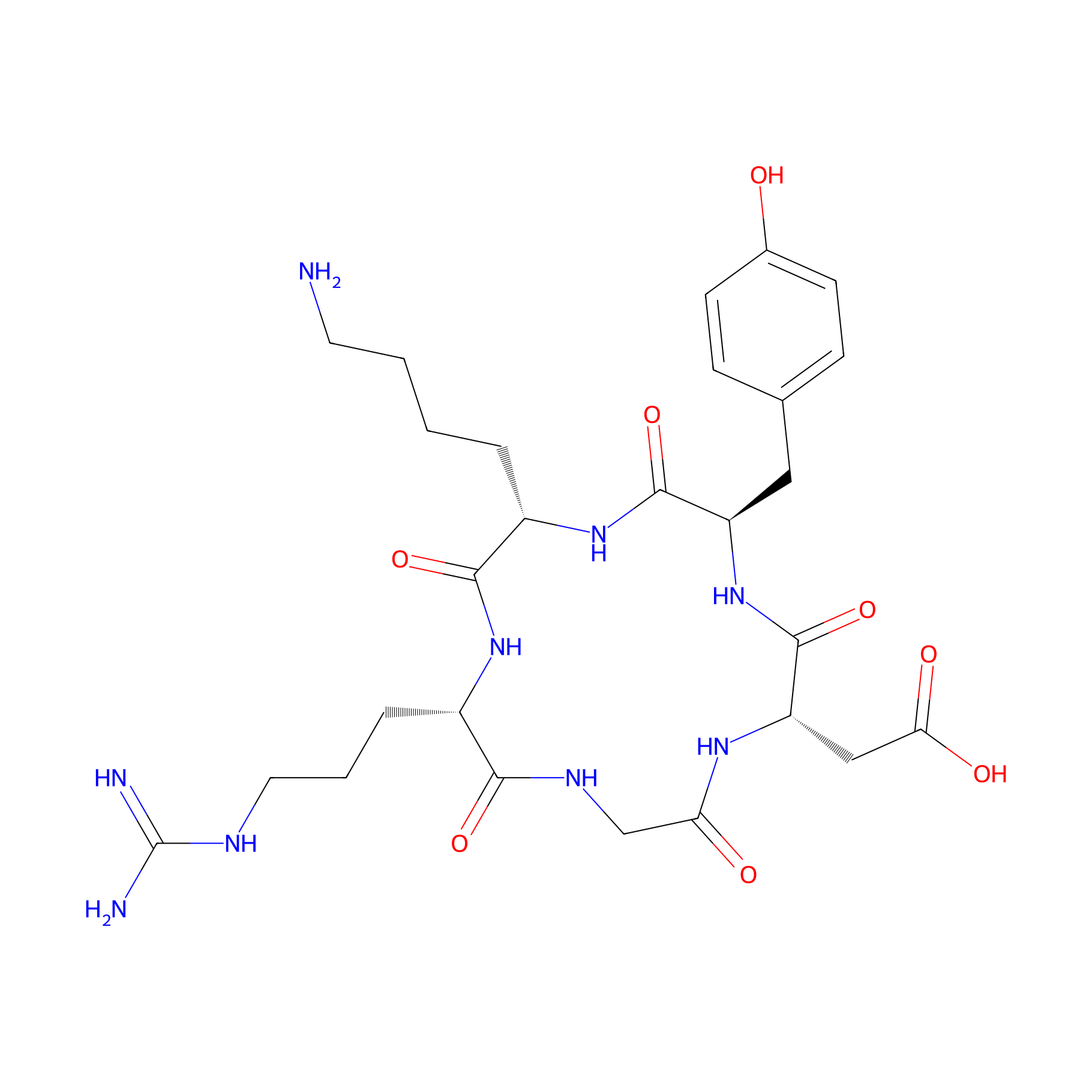
|
|||||
| Sequence |
RGDYK
|
|||||
| Peptide Type |
Cyclic
|
|||||
| Receptor Name |
Integrin alpha-V; Integrin beta-3 (ITGAV; ITGB3)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C27H41N9O8
|
|||||
| Isosmiles |
N=C(N)NCCC[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc2ccc(O)cc2)NC(=O)[C@H](CC(=O)O)NC(=O)CNC1=O
|
|||||
| InChI |
InChI=1S/C27H41N9O8/c28-10-2-1-4-18-24(42)34-17(5-3-11-31-27(29)30)23(41)32-14-21(38)33-20(13-22(39)40)26(44)36-19(25(43)35-18)12-15-6-8-16(37)9-7-15/h6-9,17-20,37H,1-5,10-14,28H2,(H,32,41)(H,33,38)(H,34,42)(H,35,43)(H,36,44)(H,39,40)(H4,29,30,31)/t17-,18-,19+,20-/m0/s1
|
|||||
| InChIKey |
LAROZEWEPNAWMD-HAGHYFMRSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
619.68
|
Polar area
|
290.95
|
||
|
Complexity
|
619.3078093
|
xlogp Value
|
-3.12933
|
|||
|
Heavy Count
|
44
|
Rot Bonds
|
12
|
|||
|
Hbond acc
|
9
|
Hbond Donor
|
11
|
|||
The Activity Data of This Peptide
| Peptide Activity Information 1 | [1] | |||||
| IC50 | > 10000 nM | |||||
|---|---|---|---|---|---|---|
| Binding Affinity Assay |
The activity and selectivity of integrin ligands were determined by a solid-phase binding assay using coated extracellular matrix proteins and soluble integrins.
|
|||||
| Experimental Condition | LAP (TGF-β)(αvβ6) | |||||
| Peptide Activity Information 2 | [1] | |||||
| IC50 | > 10000 nM | |||||
| Binding Affinity Assay |
The activity and selectivity of integrin ligands were determined by a solid-phase binding assay using coated extracellular matrix proteins and soluble integrins.
|
|||||
| Experimental Condition | Human fibrinogen(αIIbβ3) | |||||
| Peptide Activity Information 3 | [1] | |||||
| IC50 | 3.8 ± 0.42 nM | |||||
| Binding Affinity Assay |
The activity and selectivity of integrin ligands were determined by a solid-phase binding assay using coated extracellular matrix proteins and soluble integrins.
|
|||||
| Experimental Condition | Human vitronectin(αvβ3) | |||||
| Peptide Activity Information 4 | [1] | |||||
| IC50 | 86 ± 7 nM | |||||
| Binding Affinity Assay |
The activity and selectivity of integrin ligands were determined by a solid-phase binding assay using coated extracellular matrix proteins and soluble integrins.
|
|||||
| Experimental Condition | Human vitronectin(αvβ5) | |||||
| Peptide Activity Information 5 | [1] | |||||
| IC50 | 236 ± 45 nM | |||||
| Binding Affinity Assay |
The activity and selectivity of integrin ligands were determined by a solid-phase binding assay using coated extracellular matrix proteins and soluble integrins.
|
|||||
| Experimental Condition | LAP (TGF-b)(αvβ8) | |||||
| Peptide Activity Information 6 | [1] | |||||
| IC50 | 503 ± 55 nM | |||||
| Binding Affinity Assay |
The activity and selectivity of integrin ligands were determined by a solid-phase binding assay using coated extracellular matrix proteins and soluble integrins.
|
|||||
| Experimental Condition | Human fibronectin(α5β1) | |||||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
68Ga-NODAGA-E[c(RGDyK)]2 [Phase 3]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Radiochemical purity |
89%-99%
|
|||
| In Vivo Model | Nude mice bearing either human glioblastoma (U87MG)/human neuroendocrine (H727) xenograft tumors. | ||||
64Cu-NODAGA-E[c(RGDyK)]2 [Investigative]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Radiochemical purity |
92%-99%
|
|||
| In Vivo Model | Nude mice bearing either human glioblastoma (U87MG)/human neuroendocrine (H727) xenograft tumors. | ||||
Geo77 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
9 ± 0.4 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | 10-50 min | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Cervical carcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
11 ± 0.3 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Half life period | 10-50 min | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
15 ± 0.7 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 10-50 min | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Chronic myeloid leukemia | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
23 ± 0.5 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Chronic myeloid leukemia | K562 cell | CVCL_0004 | ||
| Half life period | 10-50 min | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
3 ± 0.4 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | 10-50 min | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Cervical carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
9 ± 0.2 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Half life period | 10-50 min | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
10 ± 0.3 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 10-50 min | ||||
| Experiment 8 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Chronic myeloid leukemia | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
17 ± 0.5 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Chronic myeloid leukemia | K562 cell | CVCL_0004 | ||
| Half life period | 10-50 min | ||||
| Experiment 9 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
4 ± 0.4 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | 10-50 min | ||||
| Experiment 10 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Cervical carcinoma | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
5 ± 0.2 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Half life period | 10-50 min | ||||
| Experiment 11 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
8 ± 0.5 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 10-50 min | ||||
| Experiment 12 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Chronic myeloid leukemia | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
12 ± 0.9 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Chronic myeloid leukemia | K562 cell | CVCL_0004 | ||
| Half life period | 10-50 min | ||||
Geo75 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Chronic myeloid leukemia | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
10 ± 0.3 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Chronic myeloid leukemia | K562 cell | CVCL_0004 | ||
| Half life period | 10-50 min | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
10 ± 0.5 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | 10-50 min | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Cervical carcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
11 ± 0.3 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Half life period | 10-50 min | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
16 ± 0.3 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 10-50 min | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
4 ± 0.5 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | 10-50 min | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Cervical carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
11 ± 0.3 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Half life period | 10-50 min | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
11 ± 0.4 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 10-50 min | ||||
| Experiment 8 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Chronic myeloid leukemia | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
12 ± 0.2 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Chronic myeloid leukemia | K562 cell | CVCL_0004 | ||
| Half life period | 10-50 min | ||||
| Experiment 9 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Cervical carcinoma | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
5 ± 0.4 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Half life period | 10-50 min | ||||
| Experiment 10 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Chronic myeloid leukemia | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
5 ± 0.3 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Chronic myeloid leukemia | K562 cell | CVCL_0004 | ||
| Half life period | 10-50 min | ||||
| Experiment 11 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
5 ± 0.5 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | 10-50 min | ||||
| Experiment 12 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
8 ± 0.2 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | 10-50 min | ||||
Geo85 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
64 ± 0.9 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | > 48 h | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Cervical carcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | > 100 µM | |||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Half life period | > 48 h | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Chronic myeloid leukemia | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | > 100 µM | |||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Chronic myeloid leukemia | K562 cell | CVCL_0004 | ||
| Half life period | > 48 h | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | > 100 µM | |||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3 targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates, geo75 and geo77 exhibited antiproliferative effects with low micromolar IC50 values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | > 48 h | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
45 ± 1.0 μM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | > 48 h | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Cervical carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Half life period | > 48 h | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Chronic myeloid leukemia | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Chronic myeloid leukemia | K562 cell | CVCL_0004 | ||
| Half life period | > 48 h | ||||
| Experiment 8 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | > 48 h | ||||
| Experiment 9 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
33 ± 0.9 µM
|
|||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | > 48 h | ||||
| Experiment 10 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Cervical carcinoma | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) | > 100 µM | |||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Half life period | > 48 h | ||||
| Experiment 11 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Chronic myeloid leukemia | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) | > 100 µM | |||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Chronic myeloid leukemia | K562 cell | CVCL_0004 | ||
| Half life period | > 48 h | ||||
| Experiment 12 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) | > 100 µM | |||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Peptide-drug conjugates are delivery systems for selective delivery of cytotoxic agents to target cancer cells. In this work, the optimized synthesis of JH-VII-139-1 and its c(RGDyK) peptide conjugates is presented. The low nanomolar SRPK1 inhibitor, JH-VII-139-1, which is an analogue of Alectinib, was linked to the β3targeting oligopeptide c(RGDyK) through amide, carbamate and urea linkers. The chemostability, cytotoxic and antiangiogenic properties of the synthesized hybrids were thoroughly studied. All conjugates retained mid nanomolar-level inhibitory activity against SRPK1 kinase and two out of four conjugates,geo75andgeo77exhibited antiproliferative effects with low micromolar IC50values against HeLa, K562, MDA-MB231 and MCF7 cancer cells. The activities were strongly related to the stability of the linkers and the release of JH-VII-139-1. In vivo zebrafish screening assays demonstrated the ability of the synthesized conjugates to inhibit the length or width of intersegmental vessels (ISVs). Flow cytometry experiments were used to test the cellular uptake of a fluorescein tagged hybrid in MCF7 and MDA-MB231 cells that revealed a receptor-mediated endocytosis process. In conclusion, most conjugates retained the inhibitory potency against SRPK1 as JH-VII-139-1 and demonstrated antiproliferative and antiangiogenic activities. Further animal model experiments are needed to uncover the full potential of such peptide conjugates in cancer therapy and angiogenesis-related diseases.
Click to Show/Hide
|
||||
| Description |
The cytotoxic activity of JH-VII-139-1 and JH-VII-139-1-c(RGDyK) hybrid compounds was evaluated at different concentrations (0.5-50 uM) over a panel of cell lines including HeLa cervical cancer, MCF7 mammary carcinoma, the triple-negative breast cancer MDA-MB-231 and K562 lymphoblast cells. Integrin receptors are highly overexpressed on the surface of many types of cancer. The metastatic breast cancer cell lines MDA-MB-435 and MCF-7, as well as HeLa cells, express high levels of V3 integrins. On the other hand, K562 cells express very low levels of V3 integrins. The cytotoxic and cytostatic activities of the JH-VII-139-1-c(RGDyK) hybrid compounds were estimated by three concentration-dependent parameters: GI50 (concentration that results in 50% growth inhibition), TGI (concentration that results in total growth inhibition or cytostatic effect), and IC50 (concentration that results in 50% growth cytotoxic effect). JH-VII-139-1 and the hybrid compounds, geo75 and geo77, showed a significant cytostatic (GI50 = 4-12 uM) and cytotoxic (IC50 = 3-18 uM) effect against HeLa, MCF-7, MDA-MB-231 and K562 cancer cells. The most active compound was geo77, showing IC50 and GI50 values of 3 uM and 4 uM, respectively in the most sensitive cell line MCF-7. Geo85 exhibited a slightly cytostatic effect (GI50 = 33 uM), with a minor cytotoxicity (IC50 = 45 uM) only against MCF-7 cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Half life period | > 48 h | ||||
References
