Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00153
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
PSMA-specific peptide
|
|||||
| Structure |
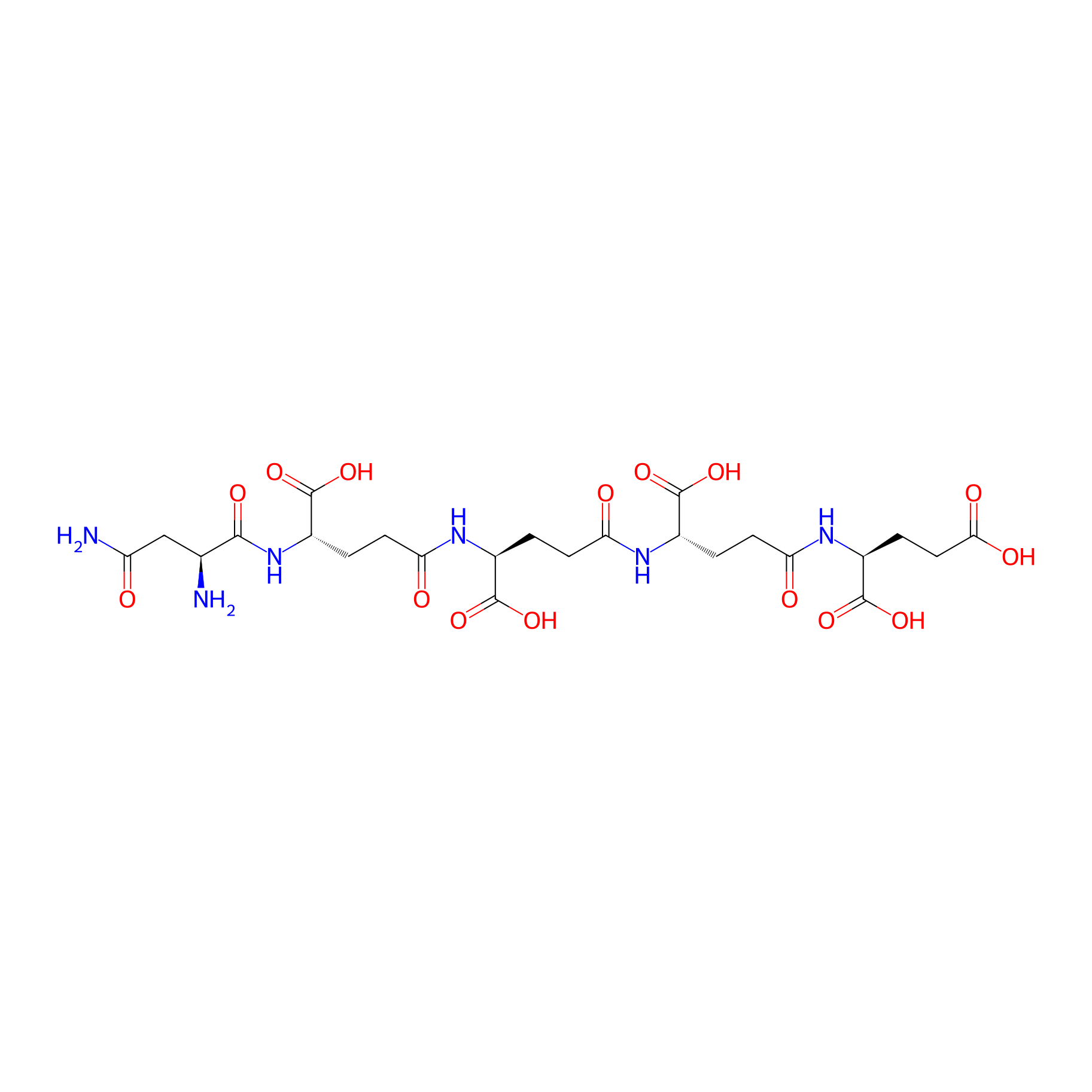
|
|||||
| Sequence |
XXXXE
|
|||||
| Peptide Type |
Linear
|
|||||
| Receptor Name |
Glutamate carboxypeptidase 2 (FOLH1)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C24H36N6O15
|
|||||
| Isosmiles |
NC(=O)C[C@H](N)C(=O)N[C@@H](CCC(=O)N[C@@H](CCC(=O)N[C@@H](CCC(=O)N[C@@H](CCC(=O)O)C(=O)O)C(=O)O)C(=O)O)C(=O)O
|
|||||
| InChI |
InChI=1S/C24H36N6O15/c25-10(9-15(26)31)20(37)30-14(24(44)45)3-7-18(34)28-12(22(40)41)1-5-16(32)27-11(21(38)39)2-6-17(33)29-13(23(42)43)4-8-19(35)36/h10-14H,1-9,25H2,(H2,26,31)(H,27,32)(H,28,34)(H,29,33)(H,30,37)(H,35,36)(H,38,39)(H,40,41)(H,42,43)(H,44,45)/t10-,11-,12-,13-,14-/m0/s1
|
|||||
| InChIKey |
OWCRYJTVWVHTCC-PEDHHIEDSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
648.579
|
Polar area
|
372.01
|
||
|
Complexity
|
648.2238645
|
xlogp Value
|
-4.3279
|
|||
|
Heavy Count
|
45
|
Rot Bonds
|
23
|
|||
|
Hbond acc
|
11
|
Hbond Donor
|
11
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
Mipsagargin [Discontinued]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Time to disease progression |
134 Day
|
|||
| Patients Enrolled |
Patients with hepatocellular carcinoma who progressed on or after treatment with sorafenib or intolerant of sorafenib.
|
||||
| Administration Time | Days 1, 2, and 3 of a 28-day cycle | ||||
| Administration Dosage | 40 mg/m2 | ||||
| Description |
The median TTP was 134.0 days, median PFS was 129.0 days, and median OS was 205.0 days.
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Non-small cell lung cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
52 day
|
|||
| Patients Enrolled |
Patients with non-small cell lung cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 10 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
57 day
|
|||
| Patients Enrolled |
Patients with pancreas cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Adenocarcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
63 day
|
|||
| Patients Enrolled |
Patients with adenocarcinoma cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Advanced solid tumour | ||||
| Efficacy Data | Progression-free survival (PFS) |
77 day
|
|||
| Patients Enrolled |
Patients with endometrial cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40/66.8/66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Cholangiocarcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
89 day
|
|||
| Patients Enrolled |
Patients with cholangiocarcinoma cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 2.5 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
112 day
|
|||
| Patients Enrolled |
Patients with prostate cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 1.2 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 8 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colorectal cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
112 day
|
|||
| Patients Enrolled |
Patients with colorectal cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 5 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 9 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
119 day
|
|||
| Patients Enrolled |
Patients with prostate cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 88 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 10 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
121 day
|
|||
| Patients Enrolled |
Patients with prostate cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40/66.8/66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 11 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
133 day
|
|||
| Patients Enrolled |
Patients with hepatocellular cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40/66.8/66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 12 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
277 day
|
|||
| Patients Enrolled |
Patients with hepatocellular cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40/66.8/66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 13 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
336 day
|
|||
| Patients Enrolled |
Patients with hepatocellular cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40/66.8/66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 14 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
129
|
|||
| Patients Enrolled |
Patients with hepatocellular carcinoma who progressed on or after treatment with sorafenib or intolerant of sorafenib.
|
||||
| Administration Time | Days 1, 2, and 3 of a 28-day cycle | ||||
| Administration Dosage | 40 mg/m2 | ||||
| Description |
The median TTP was 134.0 days, median PFS was 129.0 days, and median OS was 205.0 days.
|
||||
| Experiment 15 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Median overall survival (mOS) |
205
|
|||
| Patients Enrolled |
Patients with hepatocellular carcinoma who progressed on or after treatment with sorafenib or intolerant of sorafenib.
|
||||
| Administration Time | Days 1, 2, and 3 of a 28-day cycle | ||||
| Administration Dosage | 40 mg/m2 | ||||
| Description |
The median TTP was 134.0 days, median PFS was 129.0 days, and median OS was 205.0 days.
|
||||
References
