Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00241
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
Mipsagargin
|
|||||
| PDC Status |
Discontinued
|
|||||
| Indication |
In total 8 Indication(s)
|
|||||
| Structure |
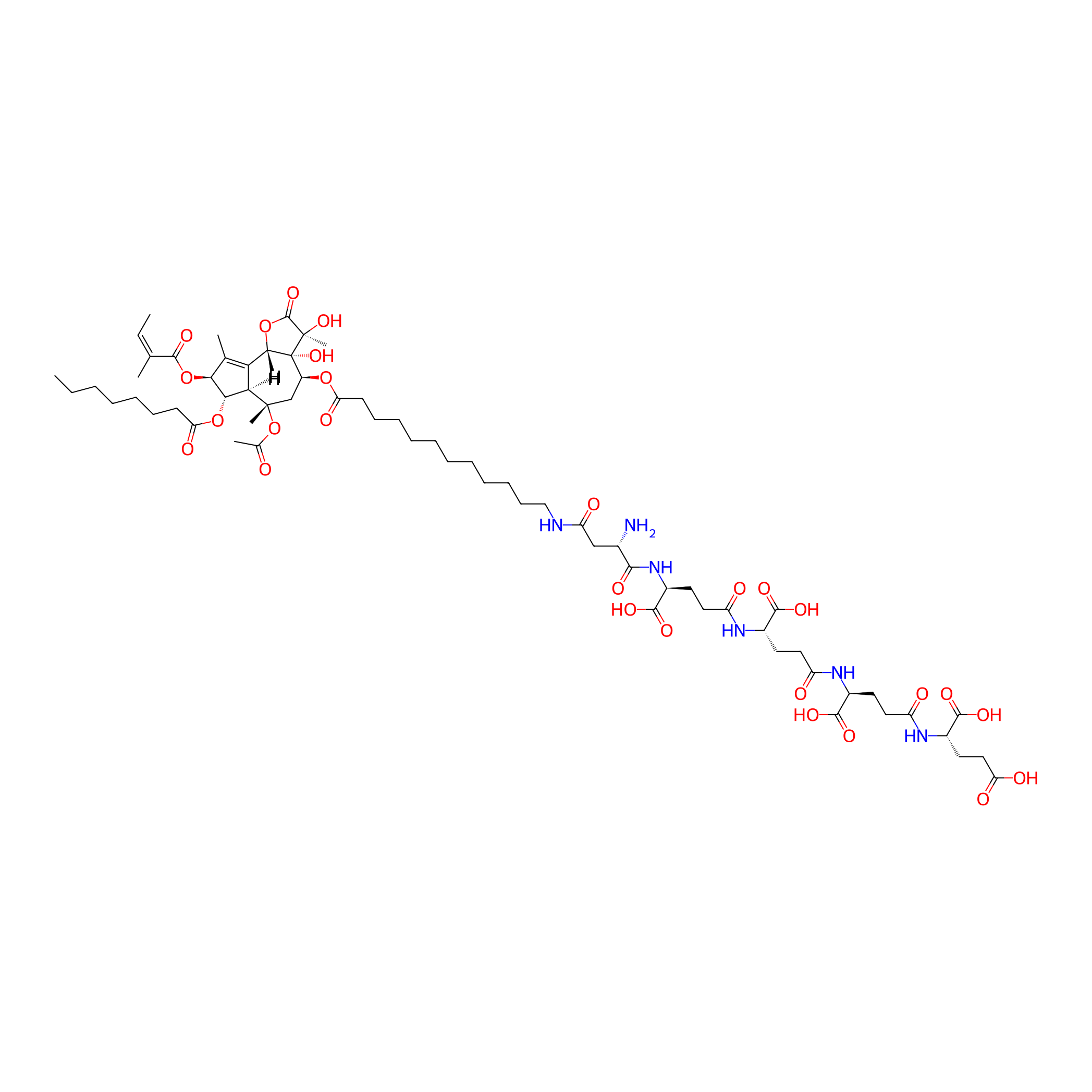
|
|||||
| Peptide Name |
PSMA-specific peptide
|
Peptide Info | ||||
| Receptor Name |
Glutamate carboxypeptidase 2 (FOLH1)
|
Receptor Info | ||||
| Drug Name |
12Adt-asp
|
Drug Info | ||||
| Therapeutic Target |
Calcium-transporting ATPase type 2C member 1 (ATP2C1)
|
Target Info | ||||
| Linker Name |
Amide bond
|
Linker Info | ||||
| Formula |
C66H100N6O27
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 1409.541 | ||||
| Lipid-water partition coefficient (xlogp) | 2.3454 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 13 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 23 | |||||
| Rotatable Bond Count (rotbonds) | 46 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Median overall survival (mOS) |
205
|
|||
| Patients Enrolled |
Patients with hepatocellular carcinoma who progressed on or after treatment with sorafenib or intolerant of sorafenib.
|
||||
| Administration Time | Days 1, 2, and 3 of a 28-day cycle | ||||
| Administration Dosage | 40 mg/m2 | ||||
| Description |
The median TTP was 134.0 days, median PFS was 129.0 days, and median OS was 205.0 days.
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Non-small cell lung cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
52 day
|
|||
| Patients Enrolled |
Patients with non-small cell lung cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 10 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
57 day
|
|||
| Patients Enrolled |
Patients with pancreas cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Adenocarcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
63 day
|
|||
| Patients Enrolled |
Patients with adenocarcinoma cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Advanced solid tumour | ||||
| Efficacy Data | Progression-free survival (PFS) |
77 day
|
|||
| Patients Enrolled |
Patients with endometrial cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40/66.8/66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Cholangiocarcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
89 day
|
|||
| Patients Enrolled |
Patients with cholangiocarcinoma cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 2.5 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
112 day
|
|||
| Patients Enrolled |
Patients with prostate cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 1.2 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 8 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colorectal cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
112 day
|
|||
| Patients Enrolled |
Patients with colorectal cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 5 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 9 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
119 day
|
|||
| Patients Enrolled |
Patients with prostate cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 88 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 10 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Progression-free survival (PFS) |
121 day
|
|||
| Patients Enrolled |
Patients with prostate cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40/66.8/66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 11 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
133 day
|
|||
| Patients Enrolled |
Patients with hepatocellular cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40/66.8/66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 12 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
277 day
|
|||
| Patients Enrolled |
Patients with hepatocellular cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40/66.8/66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 13 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
336 day
|
|||
| Patients Enrolled |
Patients with hepatocellular cancer.
|
||||
| Administration Time | Day 1/2/3 | ||||
| Administration Dosage | Dose level ( mg/m2) 40/66.8/66.8 | ||||
| Description |
Among the 42 patients considered to be evaluable for efficacy, there were no observations of objective response. However, 12 (28.6%) of patients had disease stabilisation. Overall, PFS was a median of 52.0 days (n1/442; censored 8; Q1, Q3: 50.0, 112.0). However, among the 12 patients with SD as best response, progression-free survival (PFS) was considerably higher: 129 days with 8 patients censored at the date of last study visit (Table 5A). Among the 16 efficacy-evaluable patients enrolled in part 2, 5 patients presented to the study with a diagnosis of hepatocellular carcinoma and had progressed on or were intolerant of sorafenib. In these 5 patients, prolonged disease stabilisation with an average PFS of 166 days (6 months) was observed (Table 5B); in patients with HCC who received more than 2 cycles of treatment, the PFS was 209 days, approaching 7.5 months
Click to Show/Hide
|
||||
| Experiment 14 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Progression-free survival (PFS) |
129
|
|||
| Patients Enrolled |
Patients with hepatocellular carcinoma who progressed on or after treatment with sorafenib or intolerant of sorafenib.
|
||||
| Administration Time | Days 1, 2, and 3 of a 28-day cycle | ||||
| Administration Dosage | 40 mg/m2 | ||||
| Description |
The median TTP was 134.0 days, median PFS was 129.0 days, and median OS was 205.0 days.
|
||||
| Experiment 15 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Hepatocellular carcinoma | ||||
| Efficacy Data | Time to disease progression |
134 Day
|
|||
| Patients Enrolled |
Patients with hepatocellular carcinoma who progressed on or after treatment with sorafenib or intolerant of sorafenib.
|
||||
| Administration Time | Days 1, 2, and 3 of a 28-day cycle | ||||
| Administration Dosage | 40 mg/m2 | ||||
| Description |
The median TTP was 134.0 days, median PFS was 129.0 days, and median OS was 205.0 days.
|
||||
References
