Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00033
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
225Ac-DOTATOC
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
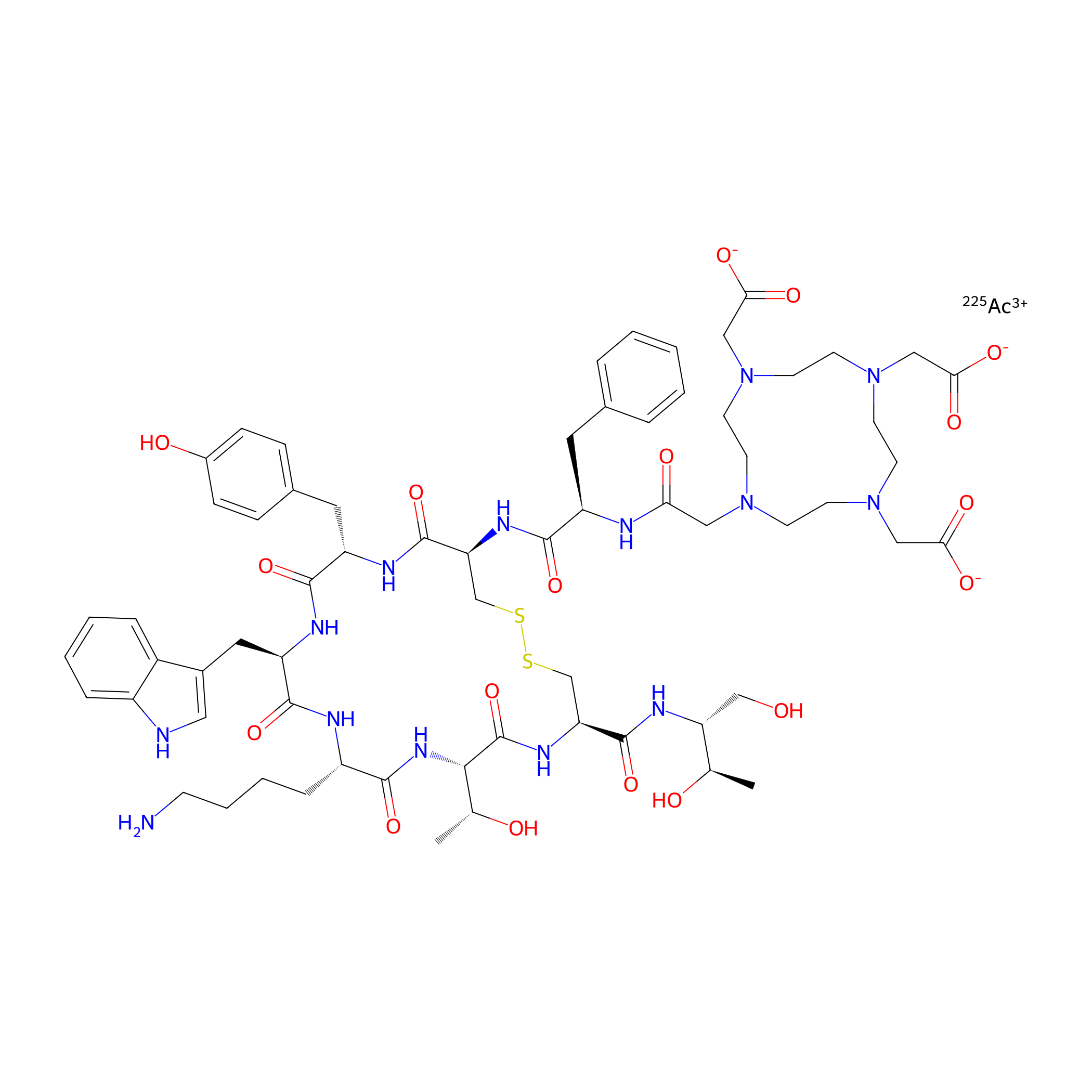
|
|||||
| Peptide Name |
Edotreotide
|
Peptide Info | ||||
| Receptor Name |
Somatostatin receptor type 1 (SSTR1)
|
Receptor Info | ||||
| Drug Name |
Actinium-225
|
Drug Info | ||||
| Therapeutic Target |
Human Deoxyribonucleic acid (hDNA)
|
Target Info | ||||
| Linker Name |
Amide bond
|
Linker Info | ||||
| Peptide Modified Type |
Cyclization modification
|
|||||
| Formula |
C65H89AcN14O18S2
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 1643.66423 | ||||
| Lipid-water partition coefficient (xlogp) | -7.471 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 14 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 25 | |||||
| Rotatable Bond Count (rotbonds) | 26 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Well-differentiated pancreatic neuroendocrine tumor | ||||
| Efficacy Data | Disease control rate (DCR) |
87.50%
|
|||
| Patients Enrolled |
9 patients with paraganglioma.
|
||||
| Administration Time | 2-9 cycles | ||||
| MOA of PDC |
Paragangliomas can metastasize, posing potential challenges in both symptomatic management and disease control. Systemic targeted radiotherapies using 131I-MIBG and 177Lu-DOTATATE are a mainstay in the treatment of metastatic paragangliomas. This clinical scenario and discussion aim to enhance physicians' knowledge of the stepwise approach to treat these patients with paraganglioma targeted radiotherapies. It comprehensively discusses current approaches to selecting paraganglioma patients for targeted radiotherapies and how to choose between the two radiotherapies based on specific patient and tumor characteristics, when either therapy is feasible, or one is superior to another one. The safety, efficacy, toxicity profiles, and optimization of these radiotherapies are also discussed, along with other therapeutic options including radiotherapies, available for patients besides these two therapies. As conclusion, perspectives in radiotherapies of paraganglioma patients are outlined since they hold near future promising approaches that can improve patient outcomes.
Click to Show/Hide
|
||||
| Description |
Recently, a study in 9 patients of metastatic paragangliomas (7 patients received prior 177Lu-DOTATATE and 3 out of 7 patients failed prior 177Lu-DOTATATE) treated with 225Ac-DOTATATE ( particle based radiotherapy targeting SSTRs) and concomitant capecitabine showed a high partial response of 50% (compared to < 25% with particle targeted radiotherapies) and stable disease in 37.5% with a disease control rate of 87.5%, without any grade 3/4 hematotoxicity, nephrotoxicity, or hepatotoxicity.
Click to Show/Hide
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Well-differentiated pancreatic neuroendocrine tumor | ||||
| Efficacy Data | Objective response rate (ORR) |
50%
|
|||
| Patients Enrolled |
9 patients with paraganglioma.
|
||||
| Administration Time | 2-9 cycles | ||||
| MOA of PDC |
Paragangliomas can metastasize, posing potential challenges in both symptomatic management and disease control. Systemic targeted radiotherapies using 131I-MIBG and 177Lu-DOTATATE are a mainstay in the treatment of metastatic paragangliomas. This clinical scenario and discussion aim to enhance physicians' knowledge of the stepwise approach to treat these patients with paraganglioma targeted radiotherapies. It comprehensively discusses current approaches to selecting paraganglioma patients for targeted radiotherapies and how to choose between the two radiotherapies based on specific patient and tumor characteristics, when either therapy is feasible, or one is superior to another one. The safety, efficacy, toxicity profiles, and optimization of these radiotherapies are also discussed, along with other therapeutic options including radiotherapies, available for patients besides these two therapies. As conclusion, perspectives in radiotherapies of paraganglioma patients are outlined since they hold near future promising approaches that can improve patient outcomes.
Click to Show/Hide
|
||||
| Description |
Recently, a study in 9 patients of metastatic paragangliomas (7 patients received prior 177Lu-DOTATATE and 3 out of 7 patients failed prior 177Lu-DOTATATE) treated with 225Ac-DOTATATE ( particle based radiotherapy targeting SSTRs) and concomitant capecitabine showed a high partial response of 50% (compared to < 25% with particle targeted radiotherapies) and stable disease in 37.5% with a disease control rate of 87.5%, without any grade 3/4 hematotoxicity, nephrotoxicity, or hepatotoxicity.
Click to Show/Hide
|
||||
References
