Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00111
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
Dau=Aoa-SYSNleEHFRWGKPV-NH2
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
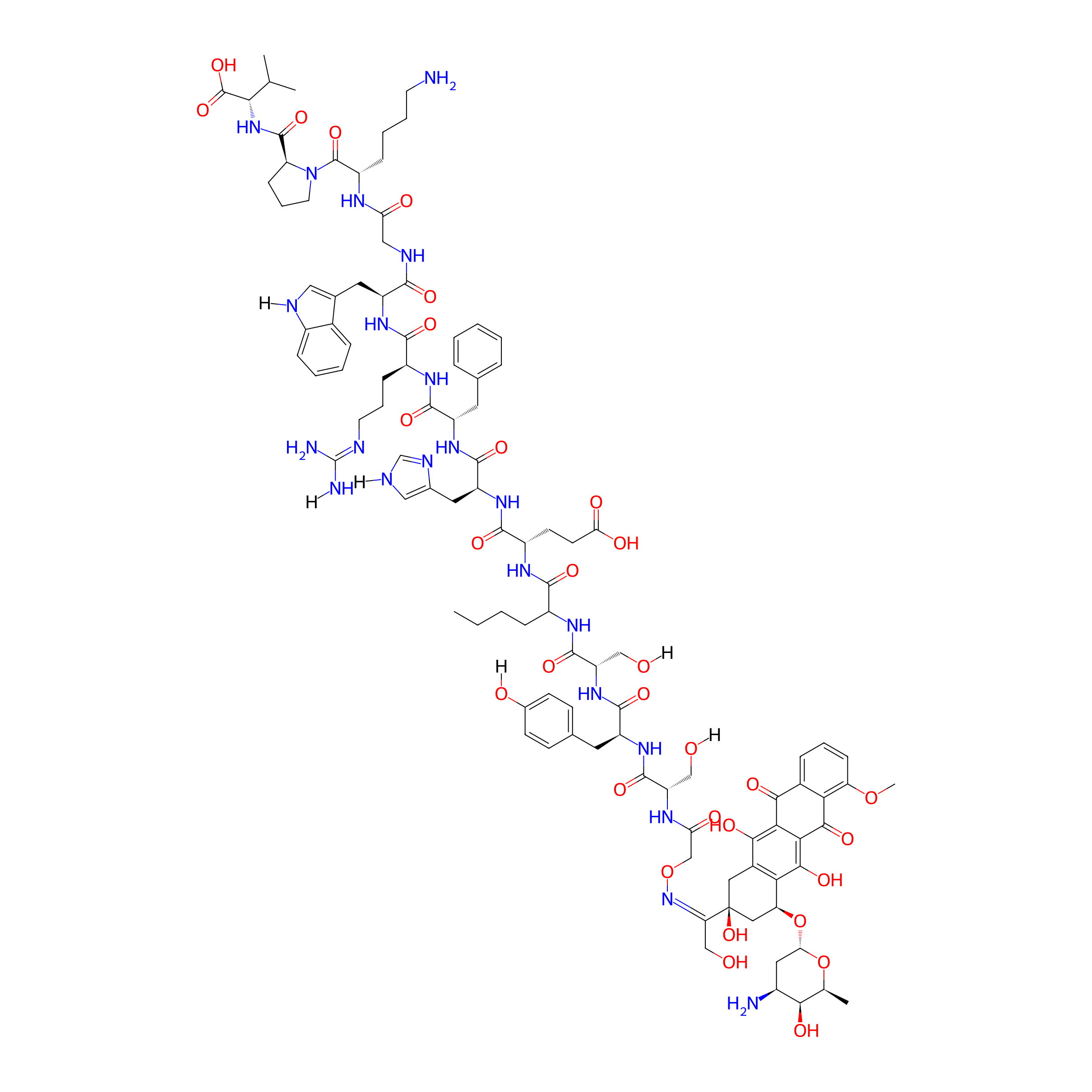
|
|||||
| Peptide Name |
Ac-SYSNleEHFRWGKPV-NH2
|
Peptide Info | ||||
| Receptor Name |
Melanocyte-stimulating hormone receptor (MC1R)
|
Receptor Info | ||||
| Drug Name |
Daunorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
Aminooxyacetic acid
|
Linker Info | ||||
| Formula |
C105H138N22O31
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 2204.382 | ||||
| Lipid-water partition coefficient (xlogp) | -3.9116 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 28 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 34 | |||||
| Rotatable Bond Count (rotbonds) | 61 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
2.9 ± 0.6 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | B16 cell | CVCL_F936 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
9.8 ± 5.4 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | A2058 cell | CVCL_1059 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
9.9 ± 1.5 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | WM983B cell | CVCL_6809 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
12.8 ± 1.6 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | M24 cell | CVCL_D032 | ||
References
