Drug Information
General Information of This Drug
| Drug ID | DRG00010 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Daunorubicin | |||||
| Synonyms |
daunorubicin; Daunomycin; 20830-81-3; Acetyladriamycin; Leukaemomycin C; Rubidomycin; Cerubidine; (+)-Daunomycin; DaunoXome; Daunorubicinum; Daunorubicine; Cerubidin; RP 13057; Rubomycin C; FI 6339; Daunorubicin (INN); NSC-82151; Daunarubicinum; Daunorrubicina; DaunoXome (TN); Daunamycin; FI-6339; FI6339; ZS7284E0ZP; (7S,9S)-9-acetyl-7-[(2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-4-methoxy-8,10-dihydro-7H-tetracene-5,12-dione; (8S-cis)-8-Acetyl-10-((3-amino-2,3,6-trideoxy-alpha-L-lyxo-hexopyrannosyl)oxy)-7,8,9,10-tetrahydro-6,8,11-trihydroxy-1-methoxy-5,12-napthacenedione; DTXSID7022883; CHEBI:41977; RP-13057; 5,12-Naphthacenedione,8-acetyl-10-[(3-amino-2,3,6-trideoxy-a-L-lyxo-hexopyranosyl)oxy]-7,8,9,10-tetrahydro-6,8,11-trihydroxy-1-methoxy-, (8S,10S)-; NCGC00024246-05; Anthracycline; Anthracyline; NDC-0082-4155; DAUNORUBICIN [INN]; Daunorubicinum [INN-Latin]; DTXCID402883; MLS000069508; Daunorubicin [INN:BAN]; NSC-83142; RCRA waste no. U059; (1S,3S)-3-acetyl-3,5,12-trihydroxy-10-methoxy-6,11-dioxo-1,2,3,4,6,11-hexahydrotetracen-1-yl 3-amino-2,3,6-trideoxy-alpha-L-lyxo-hexopyranoside; (8S,10S)-8-acetyl-10-(((2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)-6,8,11-trihydroxy-1-methoxy-7,8,9,10-tetrahydrotetracene-5,12-dione; (8S,10S)-8-acetyl-10-{[(2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy}-6,8,11-trihydroxy-1-methoxy-5,7,8,9,10,12-hexahydrotetracene-5,12-dione; CAS-20830-81-3; NSC82151; SMR000058559; CCRIS 914; SR-01000000033; SR-05000001600; HSDB 5095; NCI-C04693; EINECS 244-069-7; NSC 83142; VS-103; BRN 1445583; Tocris-1467; Daunorubicin(Daunomycin); AI3-52942; Prestwick3_000487; DAUNOMYCIN [IARC]; DAUNORUBICIN [MI]; CHEMBL178; DAUNORUBICIN [HSDB]; SCHEMBL3041; DAUNORUBICIN [VANDF]; EPIRUBICIN IMPURITY D; UNII-ZS7284E0ZP; BSPBio_000353; DAUNORUBICIN [MART.]; 5,12-Naphthacenedione, 8-acetyl-10-((3-amino-2,3,6-trideoxy-alpha-L-lyxo-hexopyranosyl)oxy)-7,8,9,10-tetrahydro-6,8,11-trihydroxy-1-methoxy-, (8S-cis)-; cid_62770; DAUNORUBICIN [WHO-DD]; BPBio1_000389; GTPL7063; BDBM32017; EX-A1337A; Valrubicin impurity, daunorubicin; Daunomycin;RP 13057;Rubidomycin; DAUNORUBICIN [ORANGE BOOK]; HMS2089H04; HMS2091K06; Pharmakon1600-01500223; VYXEOS COMPONENT DAUNORUBICIN; Tox21_110896; BDBM50368352; GR-318; HY-13062A; LMPK13050002; MFCD00866340; NSC756717; Tox21_110896_1; CCG-212559; CS-2004; DB00694; NSC-756717; NCGC00024246-06; NCGC00024246-07; NCGC00024246-09; NCGC00024246-10; NCGC00024246-12; NCGC00024246-15; NCGC00024246-18; NCGC00025173-01; (1S,3S)-3-acetyl-3,5,12-trihydroxy-10-(methyloxy)-6,11-dioxo-1,2,3,4,6,11-hexahydrotetracen-1-yl 3-amino-2,3,6-trideoxy-alpha-L-lyxo-hexopyranoside; (1S,3S)-3-acetyl-3,5,12-trihydroxy-10-methoxy-6,11-dioxo-1,2,3,4,6,11-hexahydrotetracen-1-yl 3-amino-2,3,6-trideoxy-a-L-lyxo-hexopyranoside; (7S,9R)-9-Acetyl-7-[(2S,4S,5S,6S)-4-amino-5-hydroxy-6-methyl-oxan-2-yl]oxy-6,9,11-trihydroxy-4-methoxy-8,10-dihydro-7H-tetracene-5,12-dione; (7S,9S)-9-acetyl-7-[(2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyl-tetrahydropyran-2-yl]oxy-6,9,11-trihydroxy-4-methoxy-8,10-dihydro-7H-tetracene-5,12-dione; 5,12-Naphthacenedione, 8-acetyl-10-((3-amino-2,3,6-trideoxy-alpha-L-lyxo-hexopyranosyl)oxy)-7,8,9,10-tetrahydro-6,8,11-trihydroxy-1-methoxy-, (8S,10S)-; SBI-0206677.P002; AB00514669; C01907; D07776; Epirubicin hydrochloride impurity, daunorubicin-; AB00514669-09; AB01644616_09; AB01644616_10; EN300-7479232; A814957; Q411659; SR-01000000033-4; SR-05000001600-1; SR-05000001600-2; BRD-K43389675-001-01-3; BRD-K43389675-003-02-7; BRD-K43389675-003-03-5; BRD-K43389675-003-20-9; EPIRUBICIN HYDROCHLORIDE IMPURITY D [EP IMPURITY]; VALRUBICIN IMPURITY, DAUNORUBICIN [USP IMPURITY]; DOXORUBICIN HYDROCHLORIDE IMPURITY A [EP IMPURITY]; EPIRUBICIN HYDROCHLORIDE IMPURITY, DAUNORUBICIN- [USP IMPURITY]; (1S,3S)-3-ACETYL-1,2,3,4,6,11-HEXAHYDRO-3,5,12-TRIHYDROXY-10-METHOXY-6,11-DIOXO-1-NAPHTHACENYL 3-AMINO-2,3,6-TRIDEOXY-.ALPHA.-L-LYXO-HEXOPYRANOSIDE; (7S,9S)-7-[(2R,4S,5S,6S)-4-azanyl-6-methyl-5-oxidanyl-oxan-2-yl]oxy-9-ethanoyl-4-methoxy-6,9,11-tris(oxidanyl)-8,10-dihydro-7H-tetracene-5,12-dione;hydrochloride; (7S,9S)-9-acetyl-7-(4-amino-5-hydroxy-6-methyl-tetrahydropyran-2-yl)oxy-6,9,11-trihydroxy-4-methoxy-8,10-dihydro-7H-tetracene-5,12-dione chloride; (7S,9S)-9-acetyl-7-[(2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyl-tetrahydropyran-2-yl]oxy-6,9,11-trihydroxy-4-methoxy-8,10-dihydro-7H-tetracene-5,12-quinone;hydrochloride; (7S,9S)-9-acetyl-7-[(2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-4-methoxy-8,10-dihydro-7H-tetracene-5,12-dione;hydrochloride; (7S,9S)-9-acetyl-7-[[(2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyl-2-oxanyl]oxy]-6,9,11-trihydroxy-4-methoxy-8,10-dihydro-7H-tetracene-5,12-dione;hydrochloride; (8S,10S)-8-ACETYL-10-((3-AMINO-2,3,6-TRIDEOXY-.ALPHA.-L-LYXO-HEXOPYRANOSYL)OXY)-6,8,11-TRIHYDROXY-1-METHOXY-7,8,9,10-TETRAHYDROTETRACENE-5,12-DIONE; (8S-cis)-8-Acetyl-10-[(3-amino-2,3,6-trideoxy-.alpha.-L-lyxo-hexopyranosyl)oxy]-7,8,9,10-tetrahydro--6,8,11-trihydroxy-1-methoxy-5,12-naphthacenedione; 5,12-NAPHTHACENEDIONE, 8-ACETYL-10-((3-AMINO-2,3,6-TRIDEOXY-.ALPHA.-L-LYXO-HEXOPYRANOSYL))OXY)-7,8,9,10-TETRAHYDRO-6,8,11-TRIHYDROXY-1-METHOXY-, (8S-CIS)-
Click to Show/Hide
|
|||||
| Target(s) | DNA topoisomerase 2-alpha (TOP2A) | Target Info | ||||
| Structure |
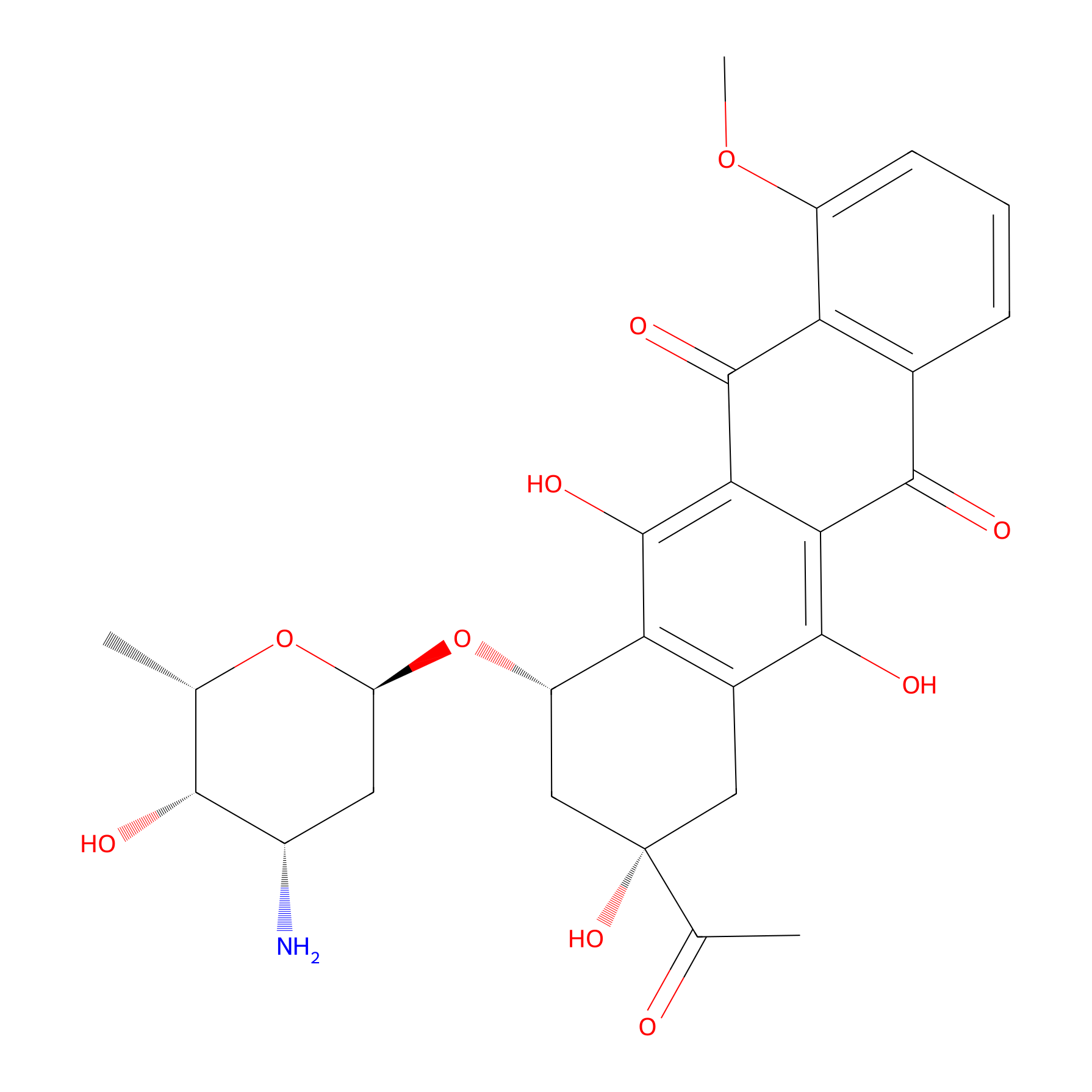
|
|||||
| Formula |
C27H29NO10
|
|||||
| #Ro5 Violations (Lipinski): 2 | Molecular Weight (mw) | 527.5 | ||||
| Lipid-water partition coefficient (xlogp) | 1.8 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 5 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 11 | |||||
| Rotatable Bond Count (rotbonds) | 4 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CC1C(C(CC(O1)OC2CC(CC3=C2C(=C4C(=C3O)C(=O)C5=C(C4=O)C(=CC=C5)OC)O)(C(=O)C)O)N)O
|
|||||
| InChI |
InChI=1S/C27H29NO10/c1-10-22(30)14(28)7-17(37-10)38-16-9-27(35,11(2)29)8-13-19(16)26(34)21-20(24(13)32)23(31)12-5-4-6-15(36-3)18(12)25(21)33/h4-6,10,14,16-17,22,30,32,34-35H,7-9,28H2,1-3H3/t10-,14-,16-,17-,22+,27-/m0/s1
|
|||||
| InChIKey |
STQGQHZAVUOBTE-VGBVRHCVSA-N
|
|||||
| IUPAC Name |
(7S,9S)-9-acetyl-7-[(2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-4-methoxy-8,10-dihydro-7H-tetracene-5,12-dione
|
|||||
The activity data of This Drug
| Standard Type | Value | Administration times | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Half Maximal Inhibitory Concentration (IC50) | 0.026±0.008 µM | 24 h | B16 cell | CVCL_F936 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.04±0.007 µM | 24 h | A2058 cell | CVCL_1059 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.045±0.12 µM | 24 h | SK-MEL-202 cell | CVCL_6106 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.050±0.023 µM | 24 h | WM983B cell | CVCL_6809 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.119±0.025 µM | 24 h | M24 cell | CVCL_D032 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.12±0.07 µM | 24 h | A2058 cell | CVCL_1059 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.21±0.01 µM | 72 h | A2780 cell | CVCL_0134 | [2] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.33±0.05 µM | 24 h | WM983B cell | CVCL_6809 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.56±0.05 µM | 24 h | OCM-3 cell | CVCL_6937 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.73±0.06 µM | 24 h | OCM-1 cell | CVCL_6934 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.75±0.01 µM | 24 h | PC-3 cell | CVCL_0035 | [3] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.81±0.07 µM | 24 h | MDA-MB-453 cell | CVCL_0418 | [3] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.90±0.06 µM | 24 h | MDA-MB-231 cell | CVCL_0062 | [3] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.43±0.58 µM | 72 h | PANC-1 cell | CVCL_0480 | [2] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.19±0.06 nM | 24 h | WM983A cell | CVCL_6808 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 10.4±1.6 nM | 24 h | A2780 cell | CVCL_0134 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 16.3±4.6 nM | 24 h | DU145 cell | CVCL_0105 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 20.9±2.7 nM | 24 h | H1975 cell | CVCL_1511 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 22.9±1.4 nM | 24 h | Hep-G2 cell | CVCL_0027 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 26.0±8.0 nM | 24 h | B16 cell | CVCL_F936 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 27.5±9.1 nM | 24 h | HT168-M1/M9 cell | CVCL_2H39 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 32.7±4.7 nM | 24 h | PC-3 cell | CVCL_0035 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 35.1±14.9 nM | 24 h | A2058 cell | CVCL_1059 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 45.6±33.5 nM | 24 h | PE/CA-PJ41 | CVCL_2680 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 49.8±22.9 nM | 24 h | WM983B cell | CVCL_6809 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 50.3±13.4 nM | 24 h | H1650 cell | CVCL_1483 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 50.5±38.7 nM | 24 h | PE/CA-PJ15 cell | CVCL_2678 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 54.6±7.4 nM | 24 h | MDA-MB-231 cell | CVCL_0062 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 56.0±14.7 nM | 24 h | 4T1 cell | CVCL_0125 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 63.9±21.0 nM | 24 h | MCF-7 cell | CVCL_0031 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 69.3±23.5 nM | 24 h | A-549 cell | CVCL_0023 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 117.5±8.6 nM | 24 h | C-26 cell | CVCL_XC68 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 118.8±25.0 nM | 24 h | M24 cell | CVCL_D032 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 126.4±53.7 nM | 24 h | U-87MG cell | CVCL_0022 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 185.6±99.8 nM | 24 h | OVCAR-8 cell | CVCL_1629 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 202.9±1.0 nM | 24 h | HT29 cell | CVCL_A8EZ | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 287.6±35.1 nM | 24 h | MRC-5 cell | CVCL_0440 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 404.0±9.4 nM | 24 h | OVCAR-3 cell | CVCL_0465 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 525.9±24.7 nM | 24 h | PANC-1 cell | CVCL_0480 | [4] | |
| Half Maximal Effective Dosage (ED50) | 0.3 uM | N.A. | L1210 cell | CVCL_0382 | [5] | |
| Half Maximal Effective Dosage (ED50) | 0.48 uM | N.A. | L1210 cell | CVCL_0382 | [6] | |
| Half Maximal Effective Dosage (ED50) | 0.66 uM | N.A. | L1210 cell | CVCL_0382 | [7] | |
| Half Maximal Growth Inhibition (GI50) | 70 nM | N.A. | HT29 cell | CVCL_A8EZ | [8] | |
| Half Maximal Growth Inhibition (GI50) | 380 nM | N.A. | NCI-H460 cell | CVCL_0459 | [9] | |
| Half Maximal Growth Inhibition (GI50) | 600 nM | N.A. | SF268 cell | CVCL_1689 | [9] | |
| Half Maximal Growth Inhibition (GI50) | 860 nM | N.A. | MCF-7 cell | CVCL_0031 | [9] | |
| Half Maximal Growth Inhibition (GI50) | 1.77 uM | N.A. | Detroit 551 cell | CVCL_2434 | [9] | |
| Half Maximal Infective Dose (ID50) | 9.5 ng/ml | N.A. | P388 cell | CVCL_7222 | [10] | |
| Half Maximal Infective Dose (ID50) | 12.5 ng/ml | N.A. | HeLa cell | CVCL_0030 | [10] | |
| Half Maximal Infective Dose (ID50) | 40 nM | N.A. | CCRF-CEM cell | CVCL_0207 | [11] | |
| Half Maximal Inhibitory Concentration (IC50) | 7 nM | N.A. | KB 3-1 cell | CVCL_2088 | [12] | |
| Half Maximal Inhibitory Concentration (IC50) | 7.4 nM | N.A. | YOSHIDA cell | CVCL_G359 | [13] | |
| Half Maximal Inhibitory Concentration (IC50) | 14 nM | N.A. | MES-SA/Dx5 cell | CVCL_2598 | [14] | |
| Half Maximal Inhibitory Concentration (IC50) | 17 nM | N.A. | MDA-MB-361 cell | CVCL_0620 | [15] | |
| Half Maximal Inhibitory Concentration (IC50) | 21 nM | N.A. | MES-SA cell | CVCL_1404 | [14] | |
| Half Maximal Inhibitory Concentration (IC50) | 21 nM | N.A. | MDA-MB-361 cell | CVCL_0620 | [15] | |
| Half Maximal Inhibitory Concentration (IC50) | 33 nM | N.A. | L1210 cell | CVCL_0382 | [12] | |
| Half Maximal Inhibitory Concentration (IC50) | 36 nM | N.A. | MDA-MB-361 cell | CVCL_0620 | [15] | |
| Half Maximal Inhibitory Concentration (IC50) | 40 nM | N.A. | L1210 cell | CVCL_0382 | [16] | |
| Half Maximal Inhibitory Concentration (IC50) | 60 nM | N.A. | MCF-7 cell | CVCL_0031 | [17] | |
| Half Maximal Inhibitory Concentration (IC50) | 79 nM | N.A. | MDA-MB-435 cell | CVCL_0417 | [18] | |
| Half Maximal Inhibitory Concentration (IC50) | 82.6 nM | N.A. | Jurkat cell | CVCL_0065 | [19] | |
| Half Maximal Inhibitory Concentration (IC50) | 90 nM | N.A. | DU145 cell | CVCL_0105 | [20] | |
| Half Maximal Inhibitory Concentration (IC50) | 129 nM | N.A. | MDA-MB-435 cell | CVCL_0417 | [18] | |
| Half Maximal Inhibitory Concentration (IC50) | 133 nM | N.A. | HT29 cell | CVCL_A8EZ | [21] | |
| Half Maximal Inhibitory Concentration (IC50) | 200 nM | N.A. | HT29 cell | CVCL_A8EZ | [22] | |
| Half Maximal Inhibitory Concentration (IC50) | 380 nM | N.A. | NCI-H460 cell | CVCL_0459 | [23] | |
| Half Maximal Inhibitory Concentration (IC50) | 400 nM | N.A. | MCF-7 cell | CVCL_0031 | [22] | |
| Half Maximal Inhibitory Concentration (IC50) | >400 nM | N.A. | HCT 15 cell | CVCL_0292 | [24] | |
| Half Maximal Inhibitory Concentration (IC50) | 600 nM | N.A. | SF268 cell | CVCL_1689 | [23] | |
| Half Maximal Inhibitory Concentration (IC50) | 880 nM | N.A. | MRC5 cell | CVCL_0440 | [23] | |
| Half Maximal Inhibitory Concentration (IC50) | 977 nM | N.A. | MDA-MB-435 cell | CVCL_0417 | [18] | |
| Half Maximal Inhibitory Concentration (IC50) | >1000 nM | N.A. | H69AR cell | CVCL_3513 | [24] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.01 uM | N.A. | NIH3T3 cell | CVCL_0594 | [25] | |
| Half Maximal Inhibitory Concentration (IC50) | 2 uM | N.A. | MES-SA/Dx5 cell | CVCL_2598 | [14] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.45 uM | N.A. | RD cell | CVCL_1649 | [26] | |
| Half Maximal Inhibitory Concentration (IC50) | 33.9 uM | N.A. | MCF-7 cell | CVCL_0031 | [27] | |
| Half Maximal Lethal Concentration (IC50) | 860 nM | N.A. | MDA-MB-231 cell | CVCL_0062 | [8] | |
| Half Maximal Lethal Concentration (IC50) | >17.2 uM | N.A. | HT29 cell | CVCL_A8EZ | [8] | |
| Tumor Growth Inhibition value (TGI) | 330 nM | N.A. | HT29 cell | CVCL_A8EZ | [8] | |
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
GnRH-III-[2ΔHis, 3D-Tic, 4Lys(Bu), 8Lys(Dau=Aoa)] [Investigative]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor weight decrease | 27.70% | |||
| Administration Dosage | 15 mg/kg Dau content | ||||
| Description |
Based on these tumor weights, we determined that free Dau, 1 and 2 inhibited tumor weight significantly by 40.1, 28.7 and 27.7% in the case of orthotopic human MDA-MB-231 breast tumor model.
|
||||
| In Vivo Model | Orthotopic MDA-MB-231 human breast carcinoma bearing mice model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Colorectal cancer | ||||
| Efficacy Data | Tumor weight | 87.10% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
The obtained data reveal that Dau, 1 and 2 significantly inhibited the tumor growth, whereby the tumor weights were reduced by 84.3, 80.8 and 87.1%, as compared to the control group.
|
||||
| In Vivo Model | Orthotopic HT-29 human colon carcinoma bearing mice model. | ||||
| In Vitro Model | Colon adenocarcinoma | HT-29 cell | CVCL_0320 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor volume decrease | 23.10% | |||
| Administration Dosage | 15 mg/kg Dau content | ||||
| Description |
Apart from that, a significant inhibition of the tumor volume was also obtained in groups which were treated with conjugate 1 (34.1%) and 2 (23.1%).
|
||||
| In Vivo Model | Orthotopic MDA-MB-231 human breast carcinoma bearing mice model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Proliferation index | 25.90% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
It was observed that both GnRH-III conjugates 1 and 2 caused a significant decrease of the proliferation index by 16.3 and 25.9%
|
||||
| In Vivo Model | Orthotopic 4T1 mice breast carcinoma bearing mice model. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Proliferation index | 39.10% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
The obtained data revealed that free Dau and both conjugates (1, 2) significantly inhibited the number of micro-metastases in the lung by 33.7, 43.8 and 49.4%, as compared to the control group. The proliferation index of lung metastases was significantly inhibited by 27.8, 37 and 39.1% in groups that were treated with free Dau, 1 and 2.
|
||||
| In Vivo Model | Orthotopic 4T1 mice breast carcinoma bearing mice model. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Number of macro-metastases decrease | 64.40% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
The number of macro-metastases in peripheral organs, such as spleen, lung, liver and kidneys, was counted, in order to determine the anti-metastatic effect of free Dau and the GnRH-III conjugates on aggressive 4T1 BC orthotopic model (Figure 5A). The number of macro-metastases in spleen was significantly decreased in all treated groups (Dau,1and2) by 64.3, 72.8 and 78.1%. In the lung, the number of macro-metastases was also significantly reduced for all treated groups by 55.4, 55.2 and 64.4%, respectively. The numbers of macro-metastases in the liver and kidneys were decreased under treatments, whereby a significant decrease could be only obtained for conjugate2.
Click to Show/Hide
|
||||
| In Vivo Model | Orthotopic 4T1 mice breast carcinoma bearing mice model. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Number of macro-metastases decrease | 78.10% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
The number of macro-metastases in peripheral organs, such as spleen, lung, liver and kidneys, was counted, in order to determine the anti-metastatic effect of free Dau and the GnRH-III conjugates on aggressive 4T1 BC orthotopic model (Figure 5A). The number of macro-metastases in spleen was significantly decreased in all treated groups (Dau, 1 and 2) by 64.3, 72.8 and 78.1%. In the lung, the number of macro-metastases was also significantly reduced for all treated groups by 55.4, 55.2 and 64.4%, respectively. The numbers of macro-metastases in the liver and kidneys were decreased under treatments, whereby a significant decrease could be only obtained for conjugate 2.
Click to Show/Hide
|
||||
| In Vivo Model | Orthotopic 4T1 mice breast carcinoma bearing mice model. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Micro-metastases decrease | 49.40% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
The obtained data revealed that free Dau and both conjugates (1, 2) significantly inhibited the number of micro-metastases in the lung by 33.7, 43.8 and 49.4%, as compared to the control group. The proliferation index of lung metastases was significantly inhibited by 27.8, 37 and 39.1% in groups that were treated with free Dau, 1 and 2.
|
||||
| In Vivo Model | Orthotopic 4T1 mice breast carcinoma bearing mice model. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Body weigth change | 8.20% | |||
| Administration Dosage | 15 mg/kg Dau content | ||||
| Description |
The body weight of the mice in control group was decreased by 2.7%, while in the groups treated with 1 and 2, it was decreased by 10.1 and 8.2%.
|
||||
| In Vivo Model | Orthotopic MDA-MB-231 human breast carcinoma bearing mice model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 0.14 ± 0.01 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.1 ± 0.2 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | B16 cell | CVCL_F936 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Oral cavity squamous cell carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.7 ± 0.5 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Oral cavity squamous cell carcinoma | PE/CA-PJ41 | CVCL_2680 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.8 ± 0.1 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.9 ± 0.2 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Prostate carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.1 ± 0.2 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | DU145 cell | CVCL_0105 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.1 ± 0.5 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Hepatoblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.2 ± 0.7 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Hepatoblastoma | Hep-G2 cell | CVCL_0027 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.3 ± 0.1 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U-87MG cell | CVCL_0022 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Lung adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.3 ± 0.7 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Lung adenocarcinoma | H1975 cell | CVCL_1511 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Prostate carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.4 ± 0.6 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.49 ± 0.53 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 13 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Normal | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.6 ± 0.7 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | C-26 cell | CVCL_XC68 | ||
| Experiment 14 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.6 ± 0.5 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | A2058 cell | CVCL_1059 | ||
| Experiment 15 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.6 ± 0.6 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | WM983B cell | CVCL_6809 | ||
| Experiment 16 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Amelanotic melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.9 ± 0.6 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | HT168-M1/M9 cell | CVCL_2H39 | ||
| Experiment 17 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Tongue squamous cell carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.9 ± 0.6 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Tongue squamous cell carcinoma | PE/CA-PJ15 cell | CVCL_2678 | ||
| Experiment 18 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.3 ± 0.9 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The efficacy of current chemotherapeutic treatments against most solid tumors is limited by their systemic toxicity, which is partly associated with the cytotoxic properties of agents such as docetaxel or doxorubicin. To avoid or minimize adverse effects from chemotherapeutic molecules, a promising targeted approach is through peptide-drug conjugates (PDCs) that link anticancer molecules to peptides designed to interact with receptors highly expressed on cancer cells, and which can mediate the molecules rapid internalization within those cells. One such receptor is sortilin (SORT1), also known as neurotensin receptor3, a membranebound receptor that belongs to the VPS10P family of receptors. TH19P01 peptide was recently designed to target and exploit SORT1s ligand internalization function. Studies have confirmed that both TH1902 (a docetaxel-TH19P01 conjugate) and TH1904 (a doxorubicin-TH19P01 conjugate) require a SORT1-dependent mechanism of action to exert anticancer activities. In recent preclinical studies performed in immunocompromised animal models, which are unable to produce mature T-cells, TH1902 was effective against several human SORT1-positive xenograft models including triple-negative breast cancer (TNBC), ovarian cancer, and endometrial cancer.
Click to Show/Hide
|
||||
| Description |
In our experiments, GnRH-III was applied as a targeting peptide. The conjugate [8Lys(Dau=Aoa-GFLG)]-GnRH-III, in which Dau was connected to the side chain of Lys in position 8 through an aminooxyacetylated cathepsin B-labile GFLG spacer, showed significant tumor growth inhibition in s.c.-developed fast-growing C26 murine colon cancer-bearing Balb/c mice. The effect highly depended on the treatment schedule. In the first attempt, the conjugate was administered i.p. at a dose of 8.86 umol (5 mg Dau-content/kg body weight) five times on days 7, 9, 11, 14 and 16 after tumor transplantation (Treatment schedule A). The tumor volume inhibition was only 15.7% on day 26 when the experiment finished. A slight enhancement in inhibition (22.3%) was detected when a dose of conjugate corresponding to 15 mg Dau content/kg body weight (26.6 μmol) was injected only once on day 7 (Treatment schedule B). An additional treatment on day 10 (Treatment schedule C) did not result in any further improvements (21.9% on day 29). However, when the treatment schedule was changed to two treatments with the same dose on days 4 and 7, 46.3% inhibition was observed on day 29 (Treatment schedule D). In contrast, the treatments with free Dau at a dose of 2 mg/kg body weight (3.55 umol) on days 7, 9, 11, 14 and 16 showed only 22.6% inhibition on day 26. The median survival rates of the treated animals in comparison to the control group were 1 (A), 1.23 (B), 1.21 (C) and 1.38 (D), respectively, and 0.81 for free Dau. These results indicate the lower toxicity and the higher tumor volume inhibition effect of the conjugates in comparison with those of free Dau, as well as the importance of the treatment schedule. In another experiment, HT-29 human colon cancer was developed s.c. in immunodeficient SCID mice. Dau and two conjugates (with or without a GFLG spacer between the GnRH-III homing peptide and the payload) were used for the treatment. The first i.p. administrations were performed on day 13 after tumor inoculation. All the mice treated once with 2.5 mg/kg body weight (4.43 umol) free Dau died within 10 days. In contrast, the conjugates at a dose of 15 mg Dau content/kg body weight (26.6 umol) that refers to 52 mg/kg [8Lys(Dau=Aoa)]-GnRH-III and 62.5 mg/kg [8Lys(Dau=Aoa-GFLG)]-GnRH-III conjugates, respectively, did not show significant toxicity. The treatment was repeated on days 23 and 30. Because of the significant weight loss in several mice in the control group, the experiment was terminated on day 35. The tumor growth inhibition could be calculated as reductions in the tumor volume by 44.3% and 57.6% and the tumor weight by 41% and 50%, respectively. Interestingly, the conjugates were poorly effective on orthotopically developed tumors. In the case of the C26 colon tumor-bearing female Balb/c mice, only a 7% reduction in tumor weight was detected on day 13 after the two treatments on days 4 and 7 with [8Lys(Dau=Aoa)]-GnRH-III, at a 26.6 umol/kg (15 mg Dau content) dose. The effect of free Dau (2 mg/kg on days 4 and 7) showed a better inhibitory effect (24.4%). Interestingly, our novel developed GnRH-III derivative, in which Ser in position 4 was replaced by Lys(Ac), was much more potent, with 49.3% inhibition. It is worth mentioning that the rate of cellular uptake of the [4Lys(Ac), 8Lys(Dau=Aoa)]-GnRH-III conjugate by the tumor cells was significantly higher than that of the conjugate with the native GnRH-III sequence.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 19 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.31 ± 0.90 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 20 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Amelanotic melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.5 ± 0.6 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | M24 cell | CVCL_D032 | ||
| Experiment 21 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.0 ± 0.8 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 22 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Minimally invasive lung adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.0 ± 0.8 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Minimally invasive lung adenocarcinoma | H1650 cell | CVCL_1483 | ||
| Experiment 23 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Lung adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.3 ± 0.4 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Lung adenocarcinoma | A-549 cell | CVCL_0023 | ||
| Experiment 24 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 7.3 ± 0.3 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 25 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian serous adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8.2 ± 0.5 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | ||
| Experiment 26 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | High grade ovarian serous adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.5 ± 0.8 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | High grade ovarian serous adenocarcinoma | OVCAR-8 cell | CVCL_1629 | ||
| Experiment 27 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Normal | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 19.7 ± 1.2 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MRC-5 cell | CVCL_0440 | ||
| Experiment 28 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 56.4 ± 4.5 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | ||
GnRH-III-[4Lys(Bu), 8Lys(Dau=Aoa)] [Investigative]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor weight decrease | 28.70% | |||
| Administration Dosage | 15 mg/kg Dau content | ||||
| Description |
Based on these tumor weights, we determined that free Dau, 1 and 2 inhibited tumor weight significantly by 40.1, 28.7 and 27.7% in the case of orthotopic human MDA-MB-231 breast tumor model.
|
||||
| In Vivo Model | Orthotopic MDA-MB-231 human breast carcinoma bearing mice model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Colorectal cancer | ||||
| Efficacy Data | Tumor weight | 80.80% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
The obtained data reveal that Dau, 1 and 2 significantly inhibited the tumor growth, whereby the tumor weights were reduced by 84.3, 80.8 and 87.1%, as compared to the control group.
|
||||
| In Vivo Model | Orthotopic HT-29 human colon carcinoma bearing mice model. | ||||
| In Vitro Model | Colon adenocarcinoma | HT-29 cell | CVCL_0320 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor volume decrease | 34.10% | |||
| Administration Dosage | 15 mg/kg Dau content | ||||
| Description |
Apart from that, a significant inhibition of the tumor volume was also obtained in groups which were treated with conjugate 1 (34.1%) and 2 (23.1%).
|
||||
| In Vivo Model | Orthotopic MDA-MB-231 human breast carcinoma bearing mice model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Proliferation index | 16.30% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
It was observed that both GnRH-III conjugates 1 and 2 caused a significant decrease of the proliferation index by 16.3 and 25.9%
|
||||
| In Vivo Model | Orthotopic 4T1 mice breast carcinoma bearing mice model. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Proliferation index | 37% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
The obtained data revealed that free Dau and both conjugates (1, 2) significantly inhibited the number of micro-metastases in the lung by 33.7, 43.8 and 49.4%, as compared to the control group. The proliferation index of lung metastases was significantly inhibited by 27.8, 37 and 39.1% in groups that were treated with free Dau, 1 and 2.
|
||||
| In Vivo Model | Orthotopic 4T1 mice breast carcinoma bearing mice model. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Number of macro-metastases decrease | 55.20% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
The number of macro-metastases in peripheral organs, such as spleen, lung, liver and kidneys, was counted, in order to determine the anti-metastatic effect of free Dau and the GnRH-III conjugates on aggressive 4T1 BC orthotopic model (Figure 5A). The number of macro-metastases in spleen was significantly decreased in all treated groups (Dau,1and2) by 64.3, 72.8 and 78.1%. In the lung, the number of macro-metastases was also significantly reduced for all treated groups by 55.4, 55.2 and 64.4%, respectively. The numbers of macro-metastases in the liver and kidneys were decreased under treatments, whereby a significant decrease could be only obtained for conjugate2.
Click to Show/Hide
|
||||
| In Vivo Model | Orthotopic 4T1 mice breast carcinoma bearing mice model. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Number of macro-metastases decrease | 72.80% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
The number of macro-metastases in peripheral organs, such as spleen, lung, liver and kidneys, was counted, in order to determine the anti-metastatic effect of free Dau and the GnRH-III conjugates on aggressive 4T1 BC orthotopic model (Figure 5A). The number of macro-metastases in spleen was significantly decreased in all treated groups (Dau, 1 and 2) by 64.3, 72.8 and 78.1%. In the lung, the number of macro-metastases was also significantly reduced for all treated groups by 55.4, 55.2 and 64.4%, respectively. The numbers of macro-metastases in the liver and kidneys were decreased under treatments, whereby a significant decrease could be only obtained for conjugate 2.
Click to Show/Hide
|
||||
| In Vivo Model | Orthotopic 4T1 mice breast carcinoma bearing mice model. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Micro-metastases decrease | 43.80% | |||
| Administration Dosage | 10 mg/kg Dau content | ||||
| Description |
The obtained data revealed that free Dau and both conjugates (1, 2) significantly inhibited the number of micro-metastases in the lung by 33.7, 43.8 and 49.4%, as compared to the control group. The proliferation index of lung metastases was significantly inhibited by 27.8, 37 and 39.1% in groups that were treated with free Dau, 1 and 2.
|
||||
| In Vivo Model | Orthotopic 4T1 mice breast carcinoma bearing mice model. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Body weigth change | 10.10% | |||
| Administration Dosage | 15 mg/kg Dau content | ||||
| Description |
The body weight of the mice in control group was decreased by 2.7%, while in the groups treated with 1 and 2, it was decreased by 10.1 and 8.2%.
|
||||
| In Vivo Model | Orthotopic MDA-MB-231 human breast carcinoma bearing mice model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.4 ± 1.1 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.2 ± 0.6 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
In our experiments, GnRH-III was applied as a targeting peptide. The conjugate [8Lys(Dau=Aoa-GFLG)]-GnRH-III, in which Dau was connected to the side chain of Lys in position 8 through an aminooxyacetylated cathepsin B-labile GFLG spacer, showed significant tumor growth inhibition in s.c.-developed fast-growing C26 murine colon cancer-bearing Balb/c mice. The effect highly depended on the treatment schedule. In the first attempt, the conjugate was administered i.p. at a dose of 8.86 umol (5 mg Dau-content/kg body weight) five times on days 7, 9, 11, 14 and 16 after tumor transplantation (Treatment schedule A). The tumor volume inhibition was only 15.7% on day 26 when the experiment finished. A slight enhancement in inhibition (22.3%) was detected when a dose of conjugate corresponding to 15 mg Dau content/kg body weight (26.6 μmol) was injected only once on day 7 (Treatment schedule B). An additional treatment on day 10 (Treatment schedule C) did not result in any further improvements (21.9% on day 29). However, when the treatment schedule was changed to two treatments with the same dose on days 4 and 7, 46.3% inhibition was observed on day 29 (Treatment schedule D). In contrast, the treatments with free Dau at a dose of 2 mg/kg body weight (3.55 umol) on days 7, 9, 11, 14 and 16 showed only 22.6% inhibition on day 26. The median survival rates of the treated animals in comparison to the control group were 1 (A), 1.23 (B), 1.21 (C) and 1.38 (D), respectively, and 0.81 for free Dau. These results indicate the lower toxicity and the higher tumor volume inhibition effect of the conjugates in comparison with those of free Dau, as well as the importance of the treatment schedule. In another experiment, HT-29 human colon cancer was developed s.c. in immunodeficient SCID mice. Dau and two conjugates (with or without a GFLG spacer between the GnRH-III homing peptide and the payload) were used for the treatment. The first i.p. administrations were performed on day 13 after tumor inoculation. All the mice treated once with 2.5 mg/kg body weight (4.43 umol) free Dau died within 10 days. In contrast, the conjugates at a dose of 15 mg Dau content/kg body weight (26.6 umol) that refers to 52 mg/kg [8Lys(Dau=Aoa)]-GnRH-III and 62.5 mg/kg [8Lys(Dau=Aoa-GFLG)]-GnRH-III conjugates, respectively, did not show significant toxicity. The treatment was repeated on days 23 and 30. Because of the significant weight loss in several mice in the control group, the experiment was terminated on day 35. The tumor growth inhibition could be calculated as reductions in the tumor volume by 44.3% and 57.6% and the tumor weight by 41% and 50%, respectively. Interestingly, the conjugates were poorly effective on orthotopically developed tumors. In the case of the C26 colon tumor-bearing female Balb/c mice, only a 7% reduction in tumor weight was detected on day 13 after the two treatments on days 4 and 7 with [8Lys(Dau=Aoa)]-GnRH-III, at a 26.6 umol/kg (15 mg Dau content) dose. The effect of free Dau (2 mg/kg on days 4 and 7) showed a better inhibitory effect (24.4%). Interestingly, our novel developed GnRH-III derivative, in which Ser in position 4 was replaced by Lys(Ac), was much more potent, with 49.3% inhibition. It is worth mentioning that the rate of cellular uptake of the [4Lys(Ac), 8Lys(Dau=Aoa)]-GnRH-III conjugate by the tumor cells was significantly higher than that of the conjugate with the native GnRH-III sequence.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 3 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.36 ± 0.07 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.2 ± 0.8 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | B16 cell | CVCL_F936 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Lung adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.1 ± 0.1 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Lung adenocarcinoma | H1975 cell | CVCL_1511 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Oral cavity squamous cell carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.7 ± 0.8 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Oral cavity squamous cell carcinoma | PE/CA-PJ41 | CVCL_2680 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Prostate carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.3 ± 0.4 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | DU145 cell | CVCL_0105 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | High grade ovarian serous adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.7 ± 0.8 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | High grade ovarian serous adenocarcinoma | OVCAR-8 cell | CVCL_1629 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.8 ± 0.8 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 6.3 ± 0.9 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Prostate carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 6.3 ± 0.3 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Hepatoblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 6.8 ± 0.3 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Hepatoblastoma | Hep-G2 cell | CVCL_0027 | ||
| Experiment 13 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Tongue squamous cell carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 7.4 ± 0.8 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Tongue squamous cell carcinoma | PE/CA-PJ15 cell | CVCL_2678 | ||
| Experiment 14 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8.4 ± 0.3 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | A2058 cell | CVCL_1059 | ||
| Experiment 15 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.0 ± 0.8 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U-87MG cell | CVCL_0022 | ||
| Experiment 16 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.00 ± 1.33µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 17 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Lung adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.7 ± 0.6 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Lung adenocarcinoma | A-549 cell | CVCL_0023 | ||
| Experiment 18 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Minimally invasive lung adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 10.5 ± 1.1 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Minimally invasive lung adenocarcinoma | H1650 cell | CVCL_1483 | ||
| Experiment 19 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Normal | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 10.6 ± 0.2 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | C-26 cell | CVCL_XC68 | ||
| Experiment 20 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 12.7 ± 1.5 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | WM983B cell | CVCL_6809 | ||
| Experiment 21 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Amelanotic melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 13.5 ± 1.1 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | HT168-M1/M9 cell | CVCL_2H39 | ||
| Experiment 22 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 15.5 ± 1.7 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 23 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 15.93 ± 0.99 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 24 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Amelanotic melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 16.2 ± 0.2 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | M24 cell | CVCL_D032 | ||
| Experiment 25 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 16.5 ± 1.2 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 26 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Normal | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 41.9 ± 3.8 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MRC-5 cell | CVCL_0440 | ||
| Experiment 27 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian serous adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 46.0 ± 1.3 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | ||
| Experiment 28 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 24 h | ||||
| Description |
The anti-proliferative effect of the GnRH-III conjugates 1 and 2, as well as free Dau, was tested on wide range of cancer cell lines from different origin and also on MRC-5 (human fibroblast) as non-cancerous control cell line. The data showed that both conjugates possess an anti-proliferative effect on all cell types (Table 1). Except for the ovarian cancer cell lines A2780 and OVCAR-8, conjugate 2 displayed higher anti-proliferative activity than conjugate 1, depending on the type of cancer cells. The lowest activity was measured on PANC-1 pancreatic cancer cells, whereby a high IC50 value was also obtained on MRC-5 cells, showing selectivity of the conjugates for cancer cell lines. The obtained IC50 values of the conjugates vary mostly in the low micromolar range and were one to two order of magnitude higher when compared to free Dau that can enter cells non-specifically by passive diffusion. Moreover, the relative potency was calculated as a ratio of conjugates IC50 and free Daus IC50 in order to show the potency of the conjugates independently from the cell line, due to different activity of free Dau. A lower value of relative potency indicates that the conjugates IC50 value is closer to the free Daus IC50 value, which implies that the targeting capacity of the conjugate as well as its anti-tumor effect is stronger on a particular cell line, as compared to a cell line with higher relative potency. The BC cell lines showed good response to the conjugates by IC50 values, as well as by relative potency. Besides, the conjugates showed high anti-proliferative activity on mice CRC cell line C26, while the conjugates showed a moderate anti-proliferative activity on HT-29 human colon adenocarcinoma, but the relative potency was in the same range as for the BC cells.
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | ||
Dau=Aoa-LRRYfQWAVGHStaL-NH2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Tumor weight inhibition | 16.60% | |||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
Like GnRH and somatostatin, bombesin (BBN) is another example of peptide hor-556 mone which receptors are overexpressed in cancerous tissues. This 14-mer peptide (Glp-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH2), first discovered in the skin of the frog Bombina bombina, is associated with the gastrin-releasing peptide (GRP) as a mammalian counterpart. The bombesin receptor family comprises neuromedin receptor B (NMB-R, or BB1), gastrin-releasing peptide receptor (GRP-R, or BB2) and bombesin receptor subtype 3 (BRS-3, or BB3). Among them, GRP-R has been the most investigated so far and has been proven to be upregulated in breast, prostate, pancreas, small-cell lung cancers, among others, hence representing a suitable target for drug delivery to tumors. Many attempts have been made to modify and shorten the sequence of bombesin to tailor its stability, activity (agonist or antagonist), affinity and selectivity towards this receptor. Several research groups have encouraged the use of their optimized structures as putative drug delivery systems, but they were never directly compared. Moreover, the previous examples of conjugates between GRP-R ligands and anthracyclines display a labile ester bond that could cause the early release of the drug in vivo. Therefore, our group took the leap and produced conjugates based on the most promising bombesin analogs as homing devices attached to Dau via an oxime bond and two cathepsin B-cleavable linkers (LRRY or GFLG). Furthermore, a conjugate bearing a novel developed bombesin analog ([6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14)) was synthesized. The use of oxime-linked Dau as a payload promoted the identification of three conjugates with improved cytostatic activity and cellular uptake by human prostate (PC-3) and breast cancer (MDA-MB-231 and MDA-MB-453) cell lines. The Dau=Aoa-Leu-OH active metabolite was readily released in all the cases in less than 30 min in rat liver lysosomal homogenate, but only L5 (Dau=Aoa-LRRY-[6D-Phe, 13Sta, 14Leu]-BBN(7-14), where Sta is statine) and L6 Dau=Aoa-LRRY-[6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14) demonstrated a satisfactory stability in the mouse plasma. Therefore, only these two conjugates were further investigated in vivo. As mentioned above, their chronic toxicity was assessed in healthy mice by administering the conjugates doses of 5, 10 and 20 mg Dau content/kg body weight. The PDCs were not critically harmful at any dose, although a relatively high mouse weight loss (10-15%) was induced at the highest dose. The in vivo antitumor efficacy was studied in murine xenograft models bearing s.c.-inoculated PC-3 human prostatic adenocarcinoma. The conjugates were administered intraperitoneally every fifth day starting from day 9 after tumor inoculation, for a total of five treatments, at a dose of 10 mg Dau-content/kg body weight, and their tumor growth inhibition values were compared to those of the mice treated with the maximum tolerated dose of free Dau (1 mg/kg) and 0.9% saline solution (control group). On the last day of the experiment (day 33), L5 and L6 revealed reductions in the tumor size by 21.4% and 31.4% and tumor weights by 16.6% and 33.1% compared to those of the control, respectively. On the other hand, the Dau-treated group had to be terminated on day 26 because of severe toxicity, without showing a significant reduction in the tumor volume and weight. To better understand the long-term effect of the newly developed compounds, the tumor doubling time (DT) was calculated; both the PDCs significantly increased the DT compared to the treatment with free Dau.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 12.35 µM | |||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
Like GnRH and somatostatin, bombesin (BBN) is another example of peptide hor-556 mone which receptors are overexpressed in cancerous tissues. This 14-mer peptide (Glp-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH2), first discovered in the skin of the frog Bombina bombina, is associated with the gastrin-releasing peptide (GRP) as a mammalian counterpart. The bombesin receptor family comprises neuromedin receptor B (NMB-R, or BB1), gastrin-releasing peptide receptor (GRP-R, or BB2) and bombesin receptor subtype 3 (BRS-3, or BB3). Among them, GRP-R has been the most investigated so far and has been proven to be upregulated in breast, prostate, pancreas, small-cell lung cancers, among others, hence representing a suitable target for drug delivery to tumors. Many attempts have been made to modify and shorten the sequence of bombesin to tailor its stability, activity (agonist or antagonist), affinity and selectivity towards this receptor. Several research groups have encouraged the use of their optimized structures as putative drug delivery systems, but they were never directly compared. Moreover, the previous examples of conjugates between GRP-R ligands and anthracyclines display a labile ester bond that could cause the early release of the drug in vivo. Therefore, our group took the leap and produced conjugates based on the most promising bombesin analogs as homing devices attached to Dau via an oxime bond and two cathepsin B-cleavable linkers (LRRY or GFLG). Furthermore, a conjugate bearing a novel developed bombesin analog ([6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14)) was synthesized. The use of oxime-linked Dau as a payload promoted the identification of three conjugates with improved cytostatic activity and cellular uptake by human prostate (PC-3) and breast cancer (MDA-MB-231 and MDA-MB-453) cell lines. The Dau=Aoa-Leu-OH active metabolite was readily released in all the cases in less than 30 min in rat liver lysosomal homogenate, but only L5 (Dau=Aoa-LRRY-[6D-Phe, 13Sta, 14Leu]-BBN(7-14), where Sta is statine) and L6 Dau=Aoa-LRRY-[6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14) demonstrated a satisfactory stability in the mouse plasma. Therefore, only these two conjugates were further investigated in vivo. As mentioned above, their chronic toxicity was assessed in healthy mice by administering the conjugates doses of 5, 10 and 20 mg Dau content/kg body weight. The PDCs were not critically harmful at any dose, although a relatively high mouse weight loss (10-15%) was induced at the highest dose. The in vivo antitumor efficacy was studied in murine xenograft models bearing s.c.-inoculated PC-3 human prostatic adenocarcinoma. The conjugates were administered intraperitoneally every fifth day starting from day 9 after tumor inoculation, for a total of five treatments, at a dose of 10 mg Dau-content/kg body weight, and their tumor growth inhibition values were compared to those of the mice treated with the maximum tolerated dose of free Dau (1 mg/kg) and 0.9% saline solution (control group). On the last day of the experiment (day 33), L5 and L6 revealed reductions in the tumor size by 21.4% and 31.4% and tumor weights by 16.6% and 33.1% compared to those of the control, respectively. On the other hand, the Dau-treated group had to be terminated on day 26 because of severe toxicity, without showing a significant reduction in the tumor volume and weight. To better understand the long-term effect of the newly developed compounds, the tumor doubling time (DT) was calculated; both the PDCs significantly increased the DT compared to the treatment with free Dau.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.22 ± 0.19 µM | |||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
Like GnRH and somatostatin, bombesin (BBN) is another example of peptide hor-556 mone which receptors are overexpressed in cancerous tissues. This 14-mer peptide (Glp-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH2), first discovered in the skin of the frog Bombina bombina, is associated with the gastrin-releasing peptide (GRP) as a mammalian counterpart. The bombesin receptor family comprises neuromedin receptor B (NMB-R, or BB1), gastrin-releasing peptide receptor (GRP-R, or BB2) and bombesin receptor subtype 3 (BRS-3, or BB3). Among them, GRP-R has been the most investigated so far and has been proven to be upregulated in breast, prostate, pancreas, small-cell lung cancers, among others, hence representing a suitable target for drug delivery to tumors. Many attempts have been made to modify and shorten the sequence of bombesin to tailor its stability, activity (agonist or antagonist), affinity and selectivity towards this receptor. Several research groups have encouraged the use of their optimized structures as putative drug delivery systems, but they were never directly compared. Moreover, the previous examples of conjugates between GRP-R ligands and anthracyclines display a labile ester bond that could cause the early release of the drug in vivo. Therefore, our group took the leap and produced conjugates based on the most promising bombesin analogs as homing devices attached to Dau via an oxime bond and two cathepsin B-cleavable linkers (LRRY or GFLG). Furthermore, a conjugate bearing a novel developed bombesin analog ([6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14)) was synthesized. The use of oxime-linked Dau as a payload promoted the identification of three conjugates with improved cytostatic activity and cellular uptake by human prostate (PC-3) and breast cancer (MDA-MB-231 and MDA-MB-453) cell lines. The Dau=Aoa-Leu-OH active metabolite was readily released in all the cases in less than 30 min in rat liver lysosomal homogenate, but only L5 (Dau=Aoa-LRRY-[6D-Phe, 13Sta, 14Leu]-BBN(7-14), where Sta is statine) and L6 Dau=Aoa-LRRY-[6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14) demonstrated a satisfactory stability in the mouse plasma. Therefore, only these two conjugates were further investigated in vivo. As mentioned above, their chronic toxicity was assessed in healthy mice by administering the conjugates doses of 5, 10 and 20 mg Dau content/kg body weight. The PDCs were not critically harmful at any dose, although a relatively high mouse weight loss (10-15%) was induced at the highest dose. The in vivo antitumor efficacy was studied in murine xenograft models bearing s.c.-inoculated PC-3 human prostatic adenocarcinoma. The conjugates were administered intraperitoneally every fifth day starting from day 9 after tumor inoculation, for a total of five treatments, at a dose of 10 mg Dau-content/kg body weight, and their tumor growth inhibition values were compared to those of the mice treated with the maximum tolerated dose of free Dau (1 mg/kg) and 0.9% saline solution (control group). On the last day of the experiment (day 33), L5 and L6 revealed reductions in the tumor size by 21.4% and 31.4% and tumor weights by 16.6% and 33.1% compared to those of the control, respectively. On the other hand, the Dau-treated group had to be terminated on day 26 because of severe toxicity, without showing a significant reduction in the tumor volume and weight. To better understand the long-term effect of the newly developed compounds, the tumor doubling time (DT) was calculated; both the PDCs significantly increased the DT compared to the treatment with free Dau.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Drug concentration | 21.40% | |||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
Like GnRH and somatostatin, bombesin (BBN) is another example of peptide hor-556 mone which receptors are overexpressed in cancerous tissues. This 14-mer peptide (Glp-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH2), first discovered in the skin of the frog Bombina bombina, is associated with the gastrin-releasing peptide (GRP) as a mammalian counterpart. The bombesin receptor family comprises neuromedin receptor B (NMB-R, or BB1), gastrin-releasing peptide receptor (GRP-R, or BB2) and bombesin receptor subtype 3 (BRS-3, or BB3). Among them, GRP-R has been the most investigated so far and has been proven to be upregulated in breast, prostate, pancreas, small-cell lung cancers, among others, hence representing a suitable target for drug delivery to tumors. Many attempts have been made to modify and shorten the sequence of bombesin to tailor its stability, activity (agonist or antagonist), affinity and selectivity towards this receptor. Several research groups have encouraged the use of their optimized structures as putative drug delivery systems, but they were never directly compared. Moreover, the previous examples of conjugates between GRP-R ligands and anthracyclines display a labile ester bond that could cause the early release of the drug in vivo. Therefore, our group took the leap and produced conjugates based on the most promising bombesin analogs as homing devices attached to Dau via an oxime bond and two cathepsin B-cleavable linkers (LRRY or GFLG). Furthermore, a conjugate bearing a novel developed bombesin analog ([6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14)) was synthesized. The use of oxime-linked Dau as a payload promoted the identification of three conjugates with improved cytostatic activity and cellular uptake by human prostate (PC-3) and breast cancer (MDA-MB-231 and MDA-MB-453) cell lines. The Dau=Aoa-Leu-OH active metabolite was readily released in all the cases in less than 30 min in rat liver lysosomal homogenate, but only L5 (Dau=Aoa-LRRY-[6D-Phe, 13Sta, 14Leu]-BBN(7-14), where Sta is statine) and L6 Dau=Aoa-LRRY-[6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14) demonstrated a satisfactory stability in the mouse plasma. Therefore, only these two conjugates were further investigated in vivo. As mentioned above, their chronic toxicity was assessed in healthy mice by administering the conjugates doses of 5, 10 and 20 mg Dau content/kg body weight. The PDCs were not critically harmful at any dose, although a relatively high mouse weight loss (10-15%) was induced at the highest dose. The in vivo antitumor efficacy was studied in murine xenograft models bearing s.c.-inoculated PC-3 human prostatic adenocarcinoma. The conjugates were administered intraperitoneally every fifth day starting from day 9 after tumor inoculation, for a total of five treatments, at a dose of 10 mg Dau-content/kg body weight, and their tumor growth inhibition values were compared to those of the mice treated with the maximum tolerated dose of free Dau (1 mg/kg) and 0.9% saline solution (control group). On the last day of the experiment (day 33), L5 and L6 revealed reductions in the tumor size by 21.4% and 31.4% and tumor weights by 16.6% and 33.1% compared to those of the control, respectively. On the other hand, the Dau-treated group had to be terminated on day 26 because of severe toxicity, without showing a significant reduction in the tumor volume and weight. To better understand the long-term effect of the newly developed compounds, the tumor doubling time (DT) was calculated; both the PDCs significantly increased the DT compared to the treatment with free Dau.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
Dau=Aoa-LRRYfQWAVβAlaHStaNle-NH2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Tumor weight inhibition | 33.10% | |||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
Like GnRH and somatostatin, bombesin (BBN) is another example of peptide hor-556 mone which receptors are overexpressed in cancerous tissues. This 14-mer peptide (Glp-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH2), first discovered in the skin of the frog Bombina bombina, is associated with the gastrin-releasing peptide (GRP) as a mammalian counterpart. The bombesin receptor family comprises neuromedin receptor B (NMB-R, or BB1), gastrin-releasing peptide receptor (GRP-R, or BB2) and bombesin receptor subtype 3 (BRS-3, or BB3). Among them, GRP-R has been the most investigated so far and has been proven to be upregulated in breast, prostate, pancreas, small-cell lung cancers, among others, hence representing a suitable target for drug delivery to tumors. Many attempts have been made to modify and shorten the sequence of bombesin to tailor its stability, activity (agonist or antagonist), affinity and selectivity towards this receptor. Several research groups have encouraged the use of their optimized structures as putative drug delivery systems, but they were never directly compared. Moreover, the previous examples of conjugates between GRP-R ligands and anthracyclines display a labile ester bond that could cause the early release of the drug in vivo. Therefore, our group took the leap and produced conjugates based on the most promising bombesin analogs as homing devices attached to Dau via an oxime bond and two cathepsin B-cleavable linkers (LRRY or GFLG). Furthermore, a conjugate bearing a novel developed bombesin analog ([6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14)) was synthesized. The use of oxime-linked Dau as a payload promoted the identification of three conjugates with improved cytostatic activity and cellular uptake by human prostate (PC-3) and breast cancer (MDA-MB-231 and MDA-MB-453) cell lines. The Dau=Aoa-Leu-OH active metabolite was readily released in all the cases in less than 30 min in rat liver lysosomal homogenate, but only L5 (Dau=Aoa-LRRY-[6D-Phe, 13Sta, 14Leu]-BBN(7-14), where Sta is statine) and L6 Dau=Aoa-LRRY-[6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14) demonstrated a satisfactory stability in the mouse plasma. Therefore, only these two conjugates were further investigated in vivo. As mentioned above, their chronic toxicity was assessed in healthy mice by administering the conjugates doses of 5, 10 and 20 mg Dau content/kg body weight. The PDCs were not critically harmful at any dose, although a relatively high mouse weight loss (10-15%) was induced at the highest dose. The in vivo antitumor efficacy was studied in murine xenograft models bearing s.c.-inoculated PC-3 human prostatic adenocarcinoma. The conjugates were administered intraperitoneally every fifth day starting from day 9 after tumor inoculation, for a total of five treatments, at a dose of 10 mg Dau-content/kg body weight, and their tumor growth inhibition values were compared to those of the mice treated with the maximum tolerated dose of free Dau (1 mg/kg) and 0.9% saline solution (control group). On the last day of the experiment (day 33), L5 and L6 revealed reductions in the tumor size by 21.4% and 31.4% and tumor weights by 16.6% and 33.1% compared to those of the control, respectively. On the other hand, the Dau-treated group had to be terminated on day 26 because of severe toxicity, without showing a significant reduction in the tumor volume and weight. To better understand the long-term effect of the newly developed compounds, the tumor doubling time (DT) was calculated; both the PDCs significantly increased the DT compared to the treatment with free Dau.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 16.09 µM | |||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
Like GnRH and somatostatin, bombesin (BBN) is another example of peptide hor-556 mone which receptors are overexpressed in cancerous tissues. This 14-mer peptide (Glp-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH2), first discovered in the skin of the frog Bombina bombina, is associated with the gastrin-releasing peptide (GRP) as a mammalian counterpart. The bombesin receptor family comprises neuromedin receptor B (NMB-R, or BB1), gastrin-releasing peptide receptor (GRP-R, or BB2) and bombesin receptor subtype 3 (BRS-3, or BB3). Among them, GRP-R has been the most investigated so far and has been proven to be upregulated in breast, prostate, pancreas, small-cell lung cancers, among others, hence representing a suitable target for drug delivery to tumors. Many attempts have been made to modify and shorten the sequence of bombesin to tailor its stability, activity (agonist or antagonist), affinity and selectivity towards this receptor. Several research groups have encouraged the use of their optimized structures as putative drug delivery systems, but they were never directly compared. Moreover, the previous examples of conjugates between GRP-R ligands and anthracyclines display a labile ester bond that could cause the early release of the drug in vivo. Therefore, our group took the leap and produced conjugates based on the most promising bombesin analogs as homing devices attached to Dau via an oxime bond and two cathepsin B-cleavable linkers (LRRY or GFLG). Furthermore, a conjugate bearing a novel developed bombesin analog ([6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14)) was synthesized. The use of oxime-linked Dau as a payload promoted the identification of three conjugates with improved cytostatic activity and cellular uptake by human prostate (PC-3) and breast cancer (MDA-MB-231 and MDA-MB-453) cell lines. The Dau=Aoa-Leu-OH active metabolite was readily released in all the cases in less than 30 min in rat liver lysosomal homogenate, but only L5 (Dau=Aoa-LRRY-[6D-Phe, 13Sta, 14Leu]-BBN(7-14), where Sta is statine) and L6 Dau=Aoa-LRRY-[6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14) demonstrated a satisfactory stability in the mouse plasma. Therefore, only these two conjugates were further investigated in vivo. As mentioned above, their chronic toxicity was assessed in healthy mice by administering the conjugates doses of 5, 10 and 20 mg Dau content/kg body weight. The PDCs were not critically harmful at any dose, although a relatively high mouse weight loss (10-15%) was induced at the highest dose. The in vivo antitumor efficacy was studied in murine xenograft models bearing s.c.-inoculated PC-3 human prostatic adenocarcinoma. The conjugates were administered intraperitoneally every fifth day starting from day 9 after tumor inoculation, for a total of five treatments, at a dose of 10 mg Dau-content/kg body weight, and their tumor growth inhibition values were compared to those of the mice treated with the maximum tolerated dose of free Dau (1 mg/kg) and 0.9% saline solution (control group). On the last day of the experiment (day 33), L5 and L6 revealed reductions in the tumor size by 21.4% and 31.4% and tumor weights by 16.6% and 33.1% compared to those of the control, respectively. On the other hand, the Dau-treated group had to be terminated on day 26 because of severe toxicity, without showing a significant reduction in the tumor volume and weight. To better understand the long-term effect of the newly developed compounds, the tumor doubling time (DT) was calculated; both the PDCs significantly increased the DT compared to the treatment with free Dau.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 18.04 ± 3.01 µM | |||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
Like GnRH and somatostatin, bombesin (BBN) is another example of peptide hor-556 mone which receptors are overexpressed in cancerous tissues. This 14-mer peptide (Glp-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH2), first discovered in the skin of the frog Bombina bombina, is associated with the gastrin-releasing peptide (GRP) as a mammalian counterpart. The bombesin receptor family comprises neuromedin receptor B (NMB-R, or BB1), gastrin-releasing peptide receptor (GRP-R, or BB2) and bombesin receptor subtype 3 (BRS-3, or BB3). Among them, GRP-R has been the most investigated so far and has been proven to be upregulated in breast, prostate, pancreas, small-cell lung cancers, among others, hence representing a suitable target for drug delivery to tumors. Many attempts have been made to modify and shorten the sequence of bombesin to tailor its stability, activity (agonist or antagonist), affinity and selectivity towards this receptor. Several research groups have encouraged the use of their optimized structures as putative drug delivery systems, but they were never directly compared. Moreover, the previous examples of conjugates between GRP-R ligands and anthracyclines display a labile ester bond that could cause the early release of the drug in vivo. Therefore, our group took the leap and produced conjugates based on the most promising bombesin analogs as homing devices attached to Dau via an oxime bond and two cathepsin B-cleavable linkers (LRRY or GFLG). Furthermore, a conjugate bearing a novel developed bombesin analog ([6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14)) was synthesized. The use of oxime-linked Dau as a payload promoted the identification of three conjugates with improved cytostatic activity and cellular uptake by human prostate (PC-3) and breast cancer (MDA-MB-231 and MDA-MB-453) cell lines. The Dau=Aoa-Leu-OH active metabolite was readily released in all the cases in less than 30 min in rat liver lysosomal homogenate, but only L5 (Dau=Aoa-LRRY-[6D-Phe, 13Sta, 14Leu]-BBN(7-14), where Sta is statine) and L6 Dau=Aoa-LRRY-[6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14) demonstrated a satisfactory stability in the mouse plasma. Therefore, only these two conjugates were further investigated in vivo. As mentioned above, their chronic toxicity was assessed in healthy mice by administering the conjugates doses of 5, 10 and 20 mg Dau content/kg body weight. The PDCs were not critically harmful at any dose, although a relatively high mouse weight loss (10-15%) was induced at the highest dose. The in vivo antitumor efficacy was studied in murine xenograft models bearing s.c.-inoculated PC-3 human prostatic adenocarcinoma. The conjugates were administered intraperitoneally every fifth day starting from day 9 after tumor inoculation, for a total of five treatments, at a dose of 10 mg Dau-content/kg body weight, and their tumor growth inhibition values were compared to those of the mice treated with the maximum tolerated dose of free Dau (1 mg/kg) and 0.9% saline solution (control group). On the last day of the experiment (day 33), L5 and L6 revealed reductions in the tumor size by 21.4% and 31.4% and tumor weights by 16.6% and 33.1% compared to those of the control, respectively. On the other hand, the Dau-treated group had to be terminated on day 26 because of severe toxicity, without showing a significant reduction in the tumor volume and weight. To better understand the long-term effect of the newly developed compounds, the tumor doubling time (DT) was calculated; both the PDCs significantly increased the DT compared to the treatment with free Dau.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Drug concentration | 31.40% | |||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
Like GnRH and somatostatin, bombesin (BBN) is another example of peptide hor-556 mone which receptors are overexpressed in cancerous tissues. This 14-mer peptide (Glp-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH2), first discovered in the skin of the frog Bombina bombina, is associated with the gastrin-releasing peptide (GRP) as a mammalian counterpart. The bombesin receptor family comprises neuromedin receptor B (NMB-R, or BB1), gastrin-releasing peptide receptor (GRP-R, or BB2) and bombesin receptor subtype 3 (BRS-3, or BB3). Among them, GRP-R has been the most investigated so far and has been proven to be upregulated in breast, prostate, pancreas, small-cell lung cancers, among others, hence representing a suitable target for drug delivery to tumors. Many attempts have been made to modify and shorten the sequence of bombesin to tailor its stability, activity (agonist or antagonist), affinity and selectivity towards this receptor. Several research groups have encouraged the use of their optimized structures as putative drug delivery systems, but they were never directly compared. Moreover, the previous examples of conjugates between GRP-R ligands and anthracyclines display a labile ester bond that could cause the early release of the drug in vivo. Therefore, our group took the leap and produced conjugates based on the most promising bombesin analogs as homing devices attached to Dau via an oxime bond and two cathepsin B-cleavable linkers (LRRY or GFLG). Furthermore, a conjugate bearing a novel developed bombesin analog ([6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14)) was synthesized. The use of oxime-linked Dau as a payload promoted the identification of three conjugates with improved cytostatic activity and cellular uptake by human prostate (PC-3) and breast cancer (MDA-MB-231 and MDA-MB-453) cell lines. The Dau=Aoa-Leu-OH active metabolite was readily released in all the cases in less than 30 min in rat liver lysosomal homogenate, but only L5 (Dau=Aoa-LRRY-[6D-Phe, 13Sta, 14Leu]-BBN(7-14), where Sta is statine) and L6 Dau=Aoa-LRRY-[6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14) demonstrated a satisfactory stability in the mouse plasma. Therefore, only these two conjugates were further investigated in vivo. As mentioned above, their chronic toxicity was assessed in healthy mice by administering the conjugates doses of 5, 10 and 20 mg Dau content/kg body weight. The PDCs were not critically harmful at any dose, although a relatively high mouse weight loss (10-15%) was induced at the highest dose. The in vivo antitumor efficacy was studied in murine xenograft models bearing s.c.-inoculated PC-3 human prostatic adenocarcinoma. The conjugates were administered intraperitoneally every fifth day starting from day 9 after tumor inoculation, for a total of five treatments, at a dose of 10 mg Dau-content/kg body weight, and their tumor growth inhibition values were compared to those of the mice treated with the maximum tolerated dose of free Dau (1 mg/kg) and 0.9% saline solution (control group). On the last day of the experiment (day 33), L5 and L6 revealed reductions in the tumor size by 21.4% and 31.4% and tumor weights by 16.6% and 33.1% compared to those of the control, respectively. On the other hand, the Dau-treated group had to be terminated on day 26 because of severe toxicity, without showing a significant reduction in the tumor volume and weight. To better understand the long-term effect of the newly developed compounds, the tumor doubling time (DT) was calculated; both the PDCs significantly increased the DT compared to the treatment with free Dau.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
PDC-L6 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 4.15 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 15.47 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 16.09 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.64 ± 0.25 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.88 ± 2.82 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 18.04 ± 3.01 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
PDC-L5 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 8.96 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 12.35 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 15.87 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.22 ± 0.19 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.35 ± 0.32 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.86 ± 0.75 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
PDC-G2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 10.5 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 17.04 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.98 ± 2.10 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 7.29 ± 3.41 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 12.72 ± 1.22 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
PDC-L1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 11.63 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 18.67 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 22.92 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.15 ± 0.18 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.38 ± 0.33 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 7.87 ± 0.09 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
PDC-L2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 18.27 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 20.87 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 21.58 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8.57 ± 1.61 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 11.74 ± 0.09 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 19.14 ± 0.49 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
PDC-G4 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 20.22 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 23.33 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.73 ± 0.24 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 7.37 ± 1.28 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 20.98 ± 0.55 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
PDC-G3 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 22.59 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.55 ± 0.76 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8.78 ± 0.97 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.28 ± 0.13 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
Dau=Aoa-LRRYQWAVGHLNle-NH2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 22.92 µM | |||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
Like GnRH and somatostatin, bombesin (BBN) is another example of peptide hor-556 mone which receptors are overexpressed in cancerous tissues. This 14-mer peptide (Glp-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH2), first discovered in the skin of the frog Bombina bombina, is associated with the gastrin-releasing peptide (GRP) as a mammalian counterpart. The bombesin receptor family comprises neuromedin receptor B (NMB-R, or BB1), gastrin-releasing peptide receptor (GRP-R, or BB2) and bombesin receptor subtype 3 (BRS-3, or BB3). Among them, GRP-R has been the most investigated so far and has been proven to be upregulated in breast, prostate, pancreas, small-cell lung cancers, among others, hence representing a suitable target for drug delivery to tumors. Many attempts have been made to modify and shorten the sequence of bombesin to tailor its stability, activity (agonist or antagonist), affinity and selectivity towards this receptor. Several research groups have encouraged the use of their optimized structures as putative drug delivery systems, but they were never directly compared. Moreover, the previous examples of conjugates between GRP-R ligands and anthracyclines display a labile ester bond that could cause the early release of the drug in vivo. Therefore, our group took the leap and produced conjugates based on the most promising bombesin analogs as homing devices attached to Dau via an oxime bond and two cathepsin B-cleavable linkers (LRRY or GFLG). Furthermore, a conjugate bearing a novel developed bombesin analog ([6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14)) was synthesized. The use of oxime-linked Dau as a payload promoted the identification of three conjugates with improved cytostatic activity and cellular uptake by human prostate (PC-3) and breast cancer (MDA-MB-231 and MDA-MB-453) cell lines. The Dau=Aoa-Leu-OH active metabolite was readily released in all the cases in less than 30 min in rat liver lysosomal homogenate, but only L5 (Dau=Aoa-LRRY-[6D-Phe, 13Sta, 14Leu]-BBN(7-14), where Sta is statine) and L6 Dau=Aoa-LRRY-[6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14) demonstrated a satisfactory stability in the mouse plasma. Therefore, only these two conjugates were further investigated in vivo. As mentioned above, their chronic toxicity was assessed in healthy mice by administering the conjugates doses of 5, 10 and 20 mg Dau content/kg body weight. The PDCs were not critically harmful at any dose, although a relatively high mouse weight loss (10-15%) was induced at the highest dose. The in vivo antitumor efficacy was studied in murine xenograft models bearing s.c.-inoculated PC-3 human prostatic adenocarcinoma. The conjugates were administered intraperitoneally every fifth day starting from day 9 after tumor inoculation, for a total of five treatments, at a dose of 10 mg Dau-content/kg body weight, and their tumor growth inhibition values were compared to those of the mice treated with the maximum tolerated dose of free Dau (1 mg/kg) and 0.9% saline solution (control group). On the last day of the experiment (day 33), L5 and L6 revealed reductions in the tumor size by 21.4% and 31.4% and tumor weights by 16.6% and 33.1% compared to those of the control, respectively. On the other hand, the Dau-treated group had to be terminated on day 26 because of severe toxicity, without showing a significant reduction in the tumor volume and weight. To better understand the long-term effect of the newly developed compounds, the tumor doubling time (DT) was calculated; both the PDCs significantly increased the DT compared to the treatment with free Dau.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.38 ± 0.33 µM | |||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
Like GnRH and somatostatin, bombesin (BBN) is another example of peptide hor-556 mone which receptors are overexpressed in cancerous tissues. This 14-mer peptide (Glp-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH2), first discovered in the skin of the frog Bombina bombina, is associated with the gastrin-releasing peptide (GRP) as a mammalian counterpart. The bombesin receptor family comprises neuromedin receptor B (NMB-R, or BB1), gastrin-releasing peptide receptor (GRP-R, or BB2) and bombesin receptor subtype 3 (BRS-3, or BB3). Among them, GRP-R has been the most investigated so far and has been proven to be upregulated in breast, prostate, pancreas, small-cell lung cancers, among others, hence representing a suitable target for drug delivery to tumors. Many attempts have been made to modify and shorten the sequence of bombesin to tailor its stability, activity (agonist or antagonist), affinity and selectivity towards this receptor. Several research groups have encouraged the use of their optimized structures as putative drug delivery systems, but they were never directly compared. Moreover, the previous examples of conjugates between GRP-R ligands and anthracyclines display a labile ester bond that could cause the early release of the drug in vivo. Therefore, our group took the leap and produced conjugates based on the most promising bombesin analogs as homing devices attached to Dau via an oxime bond and two cathepsin B-cleavable linkers (LRRY or GFLG). Furthermore, a conjugate bearing a novel developed bombesin analog ([6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14)) was synthesized. The use of oxime-linked Dau as a payload promoted the identification of three conjugates with improved cytostatic activity and cellular uptake by human prostate (PC-3) and breast cancer (MDA-MB-231 and MDA-MB-453) cell lines. The Dau=Aoa-Leu-OH active metabolite was readily released in all the cases in less than 30 min in rat liver lysosomal homogenate, but only L5 (Dau=Aoa-LRRY-[6D-Phe, 13Sta, 14Leu]-BBN(7-14), where Sta is statine) and L6 Dau=Aoa-LRRY-[6D-Phe, 11-Ala, 13Sta, 14Nle]-BBN(7-14) demonstrated a satisfactory stability in the mouse plasma. Therefore, only these two conjugates were further investigated in vivo. As mentioned above, their chronic toxicity was assessed in healthy mice by administering the conjugates doses of 5, 10 and 20 mg Dau content/kg body weight. The PDCs were not critically harmful at any dose, although a relatively high mouse weight loss (10-15%) was induced at the highest dose. The in vivo antitumor efficacy was studied in murine xenograft models bearing s.c.-inoculated PC-3 human prostatic adenocarcinoma. The conjugates were administered intraperitoneally every fifth day starting from day 9 after tumor inoculation, for a total of five treatments, at a dose of 10 mg Dau-content/kg body weight, and their tumor growth inhibition values were compared to those of the mice treated with the maximum tolerated dose of free Dau (1 mg/kg) and 0.9% saline solution (control group). On the last day of the experiment (day 33), L5 and L6 revealed reductions in the tumor size by 21.4% and 31.4% and tumor weights by 16.6% and 33.1% compared to those of the control, respectively. On the other hand, the Dau-treated group had to be terminated on day 26 because of severe toxicity, without showing a significant reduction in the tumor volume and weight. To better understand the long-term effect of the newly developed compounds, the tumor doubling time (DT) was calculated; both the PDCs significantly increased the DT compared to the treatment with free Dau.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
PDC-L3 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | 24.59 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 18.96 ± 3.23 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 25 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 25 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
PDC-G1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 11.45 ± 1.53 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 11.83 ± 2.50 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 22.80 ± 3.12 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
PDC-G5 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.69 ± 0.17 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 11.62 ± 3.33 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 18.29 ± 1.46 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
PDC-L4 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Uptake Concentration (UC50) | > 25 µM | |||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
We quantified the ability of the produced PDCs to promote the internalisation of daunorubicin after binding to GRP-R by flow cytometry. Each bioconjugate was incubated for 1.5 h in four concentrations (25 uM, 12.5 uM, 6.25 uM and 3.125 uM), with the three cell lines that we have also used for the evaluation of the cytostatic effect. For better comparison, the uptake was described as the necessary concentration to internalise 50% of the compound (UC50). The uptake reflects the GRP-R protein expression: the lower UC50 values, hence the highest uptakes, are noticed in the human breast cancer cell line MDA-MB-453, whereas they are comparable in the two other cell lines. As far as the internalisation of the individual conjugates is concerned, L5 and L6 are the most promising ones in these three cell lines. Notably, both hold the LRRY spacer and the Sta13. L1 and L2 have satisfactory UC50s, too. On the other hand, majority of the conjugates with the GFLG spacer are poorly internalised.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.08 ± 0.09 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.31 ± 0.01 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 21.21 ± 5.36 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
We decided to select some of the described peptides bearing the mentioned substitutions and having affirmed affinity for GRP-R in the low nanomolar range, together with the original BBN, and use them as targeting moiety to deliver a cytotoxic payload, daunorubicin (Dau), to prostate and breast cancers. Compared to the BBN sequence, our targeting peptides are elongated by a D-Phe in position 6 and comprise substitutions in positions 11, 13 and 14. Starting from these variations, we have also generated a new sequence: [D-Phe6, β-Ala11, Sta13, Nle14]BBN. The bombesin analogues have been published in the works of different research groups throughout many years, however, a direct comparison between them in terms of drug delivery has never been done. Once the targeting peptides bind to receptors that are overexpressed on the surface of cancer cells, such as GRP-R, the PDC can be carried inside the cells via receptor-mediated endocytosis. Briefly, the receptor-ligand complex internalises via a special, coated vesicle that later fuses with an early endosome and matures into a late endosome. After the late endosome fuses with a lysosome, the PDC is also digested, the drug is released into the cytoplasm and binds to its target compartment inside the cell. To ensure this release, we have decided to insert cathepsin B cleavable tetrapeptide linkers, namely either GFLG or LRRY, between the homing peptide and Dau. Indeed, cathepsin B is highly expressed by lysosomes and can cleave between Gly-Phe and Leu-Arg, liberating the active metabolites Dau=Aoa-Gly-OH or Dau=Aoa-Leu-OH. Dau is part of the family of anthracyclines and elicits its cytostatic activity by intercalating between DNA base pairs. This prevents the topoisomerase II from resealing the DNA double helix, resulting in reduced cell proliferation. Its attachment to a peptide can be easily performed through an oxime linkage in a very simple and straightforward reaction that occurs between an aminooxy moiety and the C13 ketone on Dau. The high yields of the reaction are advantageous, especially when it comes to the production of an increased amount of conjugate for in vivo studies. Furthermore, thanks to its intrinsic fluorescence, it is possible to evaluate the internalisation of the Dau-conjugates by fluorescence-activated cell sorting (FACS) and confocal laser scanning microscopy (CLSM), without the need to produce alternative peptide conjugates. To the best of our knowledge, so far, the delivery of a payload by bombesin-related peptides has only been studied using fluorescent molecules such as TAMRA, radioligands or nanocarriers. Drugs were attached to the peptides less often, and mainly using the original or truncated bombesin sequence instead of more promising analogues, or ones that are non-selective for GRP-R. Oppositely, investigating features such as the cellular uptake directly, through a conjugate attached to a chemotherapeutic, gives the most realistic picture of the success of a homing peptide acting as a drug carrier. Thus, the generated structures provide valuable tools for the selection of targeting peptides, aiming to the further development of new tumour-selective conjugates containing different linker-payload systems.
Click to Show/Hide
|
||||
| Description |
The cytostatic effect of the conjugates and the free peptides was evaluated on the three mentioned human cancer cell lines expressing GRP-R. The free Dau was used as a positive control for comparison purposes; it displays an IC50 in the high nanomolar range. Overall, the conjugates containing the LRRY spacer have a higher cytostatic effect than the ones with the GFLG spacer. L1, which has the original BBN (7-14) sequence, and L5, bearing a D-Phe in position 6 and the Sta13-Leu14 bond at the C-terminus, shows the best activity in all three cell lines, with IC50 values in the low micromolar range. The Gly11/-Ala11 and Leu13-Met14/Sta13-Nle14 substitutions, which led to a new BBN (6-14) peptide sequence, held by the conjugate L6, affect the activity only slightly. Contrarily, the two free peptides, bearing the sequences of L5 and L6, do not show any activity on any of the cell lines. The lack of toxicity of the conjugates on healthy cells was checked on MRC-5 human fibroblasts, proving that they are non-toxic on non-cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-453 cell | CVCL_0418 | ||
Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 0.13 ± 0.09 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | SK-MEL-202 cell | CVCL_6106 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 0.79 ± 0.05 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | WM983A cell | CVCL_6808 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 0.95 ± 0.12 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | A2058 cell | CVCL_1059 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.00 ± 0.00 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | WM983B cell | CVCL_6809 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.39 ± 0.06 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM-3 cell | CVCL_6937 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.51 ± 0.06 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | OCM-1 cell | CVCL_6934 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.8 ± 0.7 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | B16 cell | CVCL_F936 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.2 ± 0.4 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | A2058 cell | CVCL_1059 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 7.9 ± 0.7 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | WM983B cell | CVCL_6809 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 11.5 ± 0.4 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | M24 cell | CVCL_D032 | ||
DauQ=AoaGFLGGE11PEG [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [30] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.2 µM | |||
| Evaluation Method | xCELLigence SP system assay | ||||
| Administration Time | 48 h | ||||
| MOA of PDC |
As observed, the cathepsin B labile spacer (GFLG), which was incorporated between the targeting peptide and the daunomycin to ensure the efficient release of the active metabolite, highly increases the hydrophobicity of the conjugates. As demonstrated, the solubility problem of these conjugates can be solved by hydrophilic polymer coupling, not only by using the well-known PEG but also by using the amino-monofunctional HbPG. To the best of our knowledge, 1:1 covalent peptide-polymer conjugates with well-defined monofunctional HbPG have been reported here for the first time. The results of the in vitro cell viability and cellular uptake measurements on HT-29 human colon adenocarcinoma cells prove that the HbPG and the PEG highly influenced the biological activity of the drug-peptide-polymer conjugates. In both peptide conjugate series, one GE11 and one D4 targeting peptide-based conjugate were found with outstanding cellular uptake and cytotoxicity values, namely Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG. According to our results, the PEG is suitable for longer targeting peptides (e.g. GE11), but the G5 spacer is not suitable irrespective of the length of the peptide because it may decrease the biological effect by increasing the flexibility of the polymer and shading of the targeting moiety. In contrast, the use of the hydrophilic hyperbranched polyglycerol (HbPG) is advantageous for short targeting peptides (e.g. D4) but only with a G5 spacer, which provides accessibility of the peptide for receptor binding and cellular uptake resulting in outstanding cytotoxicity.
Click to Show/Hide
|
||||
| Description |
The polymers influenced the cellular uptake of the conjugates in a different manner, but in both groups (GE11 and D4 containing conjugates), one of the compounds was outstanding. The HT-29 cells could uptake Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG to the highest extent. The G5 spacer increased the uptake of the D4-HbPG derivative, presumably because the increased distance between the globular HbPG and the very short peptide sequence provides a better receptor binding. In sharp contrast, the G5 spacer decreases the uptake of the conjugates in all other cases. The most significant difference was observed in the case of GE11-PEG, where the G5 spacer completely demolished the internalization. Probably, here the G5 spacer provides more flexibility for the linear PEG chain resulting in decreased receptor binding. There was one outstanding conjugate from each group (Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG) in the cytotoxicity measurements that correlated well with the results of the internalization studies. These conjugates were found to be the most potent ones in the viability measurement and were proved to be taken up by HT-29 cells the most effectively. Depending on the type of polymer, the incorporation of the G5 spacer had an opposite effect on the cytotoxic activity of the conjugates. In the presence of the G5 spacer, the antitumour activity of the PEGylated conjugates decreased, while the cytotoxicity of the HbPG-containing conjugates increased, especially in the case of those with the D4 targeting peptide. We observed that some of the conjugates (Dau[double bond, length as m-dash]Aoa-GFLG-D4-HbPG and Dau[double bond, length as m-dash]Aoa-GFLG-GE11-G5-PEG) could not cause complete cell death, i.e., ˜0% viability value - characteristic for cell-free culturing medium - was not achieved even at the highest concentration, since their dose-response curves reached a plateau in a lower concentration range. In our opinion, this can be explained by the different characteristics of the highly hydrophobic peptide chain and the highly hydrophilic polymer segment. Due to this amphiphilic character, self-aggregation of the conjugates may occur, which then may block the accessibility of the targeting peptide for receptor binding, thereby decreasing the efficiency of the conjugate as well. This assumption is also confirmed by the turbidity results, since the observed low turbidity values may be caused by the possible formation of nanosized aggregates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
Cyclic NGR peptidedaunomycin conjugates 5 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.4 ± 0.1 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.0 ± 0.6 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.7 ± 0.5 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8.7 ± 1.2 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
[8Lys(Dau=Aoa-LRRY)]-GnRH-III [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.8 ± 0.5 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
As a payload for PDCs (and ADCs), analogues of the duocarmycins are attractive. Duocarmycin SA and yatakemycin rank among the most potent natural cytotoxins discovered. The cyclopropyl and prodrug seco forms are both naturally occurring and equipotent in most circumstances. Studies of the binding-driven bonding model of their interaction with DNA suggest that their utility will be enhanced when targeted to tumor cells. In fact, SYD985, an ADC that utilizes a peptide linker for a duocarmycin analogue to trastuzumab, has recently been progressed to phase III clinical trial.
Click to Show/Hide
|
||||
| Description |
The main drug-related toxic side effect of anthracyclines is cardiotoxicity, leading to cardiomyopathy and heart failure. Comparative experiments to determine the cardiotoxicity of peptide-anthracycline conjugates with different linkages might be informative for the other conjugates with different types of drugs as well. For this purpose, human cardiomyocytes (HCM) and human umbilical vein endothelial cells (HUVEC) were used as models. The long-term (0-72 h) cytotoxic effect of sixteen GnRH-based conjugates containing Dox and Dau was determined by real-time impedimetric sensing using the xCELLigence SP system (ACEA Biosciences, San Diego, CA, USA) [75]. The results indicated that the ester-linked GnRH-Dox conjugates, including Zoptarelin Doxorubicin, showed significant toxicity at 100 nM and 1 uM, which was remarkably pronounced on the HCM cells. The cytotoxic effect was comparable to that of the free drug, especially at the highest concentration. In contrast, the conjugates with oxime-linked Dau showed no or only a minor toxicity on both the cell lines (Table 2). These data confirm that the linkage between the payload and homing peptide has a significant influence on early drug release and, consequently, an undesired toxic side effect. We may also conclude that the search for more suitable homing peptides might be more important than the application of cleavable bonds between the drug and the peptide to develop efficient DDSs.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 28.6 ± 5.5 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
The main drug-related toxic side effect of anthracyclines is cardiotoxicity, leading to cardiomyopathy and heart failure. Comparative experiments to determine the cardiotoxicity of peptide-anthracycline conjugates with different linkages might be informative for the other conjugates with different types of drugs as well. For this purpose, human cardiomyocytes (HCM) and human umbilical vein endothelial cells (HUVEC) were used as models. The long-term (0-72 h) cytotoxic effect of sixteen GnRH-based conjugates containing Dox and Dau was determined by real-time impedimetric sensing using the xCELLigence SP system (ACEA Biosciences, San Diego, CA, USA) [75]. The results indicated that the ester-linked GnRH-Dox conjugates, including Zoptarelin Doxorubicin, showed significant toxicity at 100 nM and 1 uM, which was remarkably pronounced on the HCM cells. The cytotoxic effect was comparable to that of the free drug, especially at the highest concentration. In contrast, the conjugates with oxime-linked Dau showed no or only a minor toxicity on both the cell lines (Table 2). These data confirm that the linkage between the payload and homing peptide has a significant influence on early drug release and, consequently, an undesired toxic side effect. We may also conclude that the search for more suitable homing peptides might be more important than the application of cleavable bonds between the drug and the peptide to develop efficient DDSs.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[2ΔHis, 3D-Tic, 7D-Trp 8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.90 ± 0.58 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 7.88 ± 1.24 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 10.70 ± 0.95 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
Cyclic NGR peptidedaunomycin conjugates 2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.0 ± 0.3 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.3 ± 0.9 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 6.8 ± 0.7 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 19.0 ± 1.4 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
Dau=Aoa-SYSNleEHFRWGK(Dau=Aoa)PV-NH2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.0 ± 0.7 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | B16 cell | CVCL_F936 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.0 ± 0.8 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | A2058 cell | CVCL_1059 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.6 ± 0.2 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | WM983B cell | CVCL_6809 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 11.0 ± 0.8 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | M24 cell | CVCL_D032 | ||
Ac-NleEHfRWGK(Dau=Aoa)-NH2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.06 ± 0.10 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | SK-MEL-202 cell | CVCL_6106 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 7.15 ± 0.07 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | A2058 cell | CVCL_1059 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 10.18 ± 0.06 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | WM983B cell | CVCL_6809 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 13.50 ± 0.06 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | WM983A cell | CVCL_6808 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 25.47 ± 0.08 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM-3 cell | CVCL_6937 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 29.68 ± 0.06 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effects of Dau--MSH conjugates were compared with the PrestoBlue assay, which has a higher specificity and efficacy that of the previously used MTT assay, using the same six human melanoma cell lines used in the real-time qPCR assay. According to our results, free daunomycin exhibited the lowest IC50 values, and therefore the highest cytostatic effect, due to the fact that free daunomycin passively diffuses into cells. Our Dau--MSH conjugates showed similar trends in the effectiveness of the tested cell lines. Conj2 and Conj4 resulted in IC50 values in the low uM range. A calculation of the targeting indices showed that compound Conj2 had an over 2-times higher targeting efficiency in most cell lines compared to the A2058 cell line. Interestingly, WM983A showed the lowest TI among the high-MC1R-expressing cell lines compared to A2058, and WM983B exhibited high TI regardless of the relatively low MC1R expression on the mRNA level. In the case of Conj4, the treatment resulted in the highest TIs with OCM-1 and WM983B cell lines.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | OCM-1 cell | CVCL_6934 | ||
GnRH-III-[4Lys(Bu), 8Lys(Dau=Aoa), 10ΔGly-NH-Et] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.23 ± 0.40 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 12.41 ± 2.30 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 17.84 ± 0.08 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.54 ± 0.67µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 6.5 ± 1.8 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The cancer phenotype is commonly associated with aberrant glycosylation patterns. One glycan that is directly linked to cancer is the Thomsen-Friedenreich antigen (TF or CD176). It is a disaccharide composed of a galactose β1-3 N-acetylgalactosamine, O-linked to a glycoprotein through serine or threonine residues and commonly written as Galβ1-3GalNAc--O-Ser/Thr. The TF is therapeutically attractive due to its cryptic nature in normal cells and exposure in embryonic and cancer cells. The expression of the TF has been demonstrated in 90% of primary human carcinomas, including in the lung, the breast, and the pancreas. Additionally, cancer initiating cells or cancer stem cells in the lung, liver, and colon express the TF. The peptide sequence HGRFILPWWYAFSPS (TF-peptide) is known to bind tightly to the TF (Kd = 1.2 M) and has been demonstrated to inhibit processes directly involved in TF accessibility.
Click to Show/Hide
|
||||
| Description |
The main drug-related toxic side effect of anthracyclines is cardiotoxicity, leading to cardiomyopathy and heart failure. Comparative experiments to determine the cardiotoxicity of peptide-anthracycline conjugates with different linkages might be informative for the other conjugates with different types of drugs as well. For this purpose, human cardiomyocytes (HCM) and human umbilical vein endothelial cells (HUVEC) were used as models. The long-term (0-72 h) cytotoxic effect of sixteen GnRH-based conjugates containing Dox and Dau was determined by real-time impedimetric sensing using the xCELLigence SP system (ACEA Biosciences, San Diego, CA, USA) [75]. The results indicated that the ester-linked GnRH-Dox conjugates, including Zoptarelin Doxorubicin, showed significant toxicity at 100 nM and 1 uM, which was remarkably pronounced on the HCM cells. The cytotoxic effect was comparable to that of the free drug, especially at the highest concentration. In contrast, the conjugates with oxime-linked Dau showed no or only a minor toxicity on both the cell lines (Table 2). These data confirm that the linkage between the payload and homing peptide has a significant influence on early drug release and, consequently, an undesired toxic side effect. We may also conclude that the search for more suitable homing peptides might be more important than the application of cleavable bonds between the drug and the peptide to develop efficient DDSs.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8.22 ± 0.13 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 13.89 ± 3.62 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 5 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 14.2 ± 3.2 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
In our experiments, GnRH-III was applied as a targeting peptide. The conjugate [8Lys(Dau=Aoa-GFLG)]-GnRH-III, in which Dau was connected to the side chain of Lys in position 8 through an aminooxyacetylated cathepsin B-labile GFLG spacer, showed significant tumor growth inhibition in s.c.-developed fast-growing C26 murine colon cancer-bearing Balb/c mice. The effect highly depended on the treatment schedule. In the first attempt, the conjugate was administered i.p. at a dose of 8.86 umol (5 mg Dau-content/kg body weight) five times on days 7, 9, 11, 14 and 16 after tumor transplantation (Treatment schedule A). The tumor volume inhibition was only 15.7% on day 26 when the experiment finished. A slight enhancement in inhibition (22.3%) was detected when a dose of conjugate corresponding to 15 mg Dau content/kg body weight (26.6 μmol) was injected only once on day 7 (Treatment schedule B). An additional treatment on day 10 (Treatment schedule C) did not result in any further improvements (21.9% on day 29). However, when the treatment schedule was changed to two treatments with the same dose on days 4 and 7, 46.3% inhibition was observed on day 29 (Treatment schedule D). In contrast, the treatments with free Dau at a dose of 2 mg/kg body weight (3.55 umol) on days 7, 9, 11, 14 and 16 showed only 22.6% inhibition on day 26. The median survival rates of the treated animals in comparison to the control group were 1 (A), 1.23 (B), 1.21 (C) and 1.38 (D), respectively, and 0.81 for free Dau. These results indicate the lower toxicity and the higher tumor volume inhibition effect of the conjugates in comparison with those of free Dau, as well as the importance of the treatment schedule. In another experiment, HT-29 human colon cancer was developed s.c. in immunodeficient SCID mice. Dau and two conjugates (with or without a GFLG spacer between the GnRH-III homing peptide and the payload) were used for the treatment. The first i.p. administrations were performed on day 13 after tumor inoculation. All the mice treated once with 2.5 mg/kg body weight (4.43 umol) free Dau died within 10 days. In contrast, the conjugates at a dose of 15 mg Dau content/kg body weight (26.6 umol) that refers to 52 mg/kg [8Lys(Dau=Aoa)]-GnRH-III and 62.5 mg/kg [8Lys(Dau=Aoa-GFLG)]-GnRH-III conjugates, respectively, did not show significant toxicity. The treatment was repeated on days 23 and 30. Because of the significant weight loss in several mice in the control group, the experiment was terminated on day 35. The tumor growth inhibition could be calculated as reductions in the tumor volume by 44.3% and 57.6% and the tumor weight by 41% and 50%, respectively. Interestingly, the conjugates were poorly effective on orthotopically developed tumors. In the case of the C26 colon tumor-bearing female Balb/c mice, only a 7% reduction in tumor weight was detected on day 13 after the two treatments on days 4 and 7 with [8Lys(Dau=Aoa)]-GnRH-III, at a 26.6 umol/kg (15 mg Dau content) dose. The effect of free Dau (2 mg/kg on days 4 and 7) showed a better inhibitory effect (24.4%). Interestingly, our novel developed GnRH-III derivative, in which Ser in position 4 was replaced by Lys(Ac), was much more potent, with 49.3% inhibition. It is worth mentioning that the rate of cellular uptake of the [4Lys(Ac), 8Lys(Dau=Aoa)]-GnRH-III conjugate by the tumor cells was significantly higher than that of the conjugate with the native GnRH-III sequence.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 6 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 27.8 ± 4.2 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
The main drug-related toxic side effect of anthracyclines is cardiotoxicity, leading to cardiomyopathy and heart failure. Comparative experiments to determine the cardiotoxicity of peptide-anthracycline conjugates with different linkages might be informative for the other conjugates with different types of drugs as well. For this purpose, human cardiomyocytes (HCM) and human umbilical vein endothelial cells (HUVEC) were used as models. The long-term (0-72 h) cytotoxic effect of sixteen GnRH-based conjugates containing Dox and Dau was determined by real-time impedimetric sensing using the xCELLigence SP system (ACEA Biosciences, San Diego, CA, USA) [75]. The results indicated that the ester-linked GnRH-Dox conjugates, including Zoptarelin Doxorubicin, showed significant toxicity at 100 nM and 1 uM, which was remarkably pronounced on the HCM cells. The cytotoxic effect was comparable to that of the free drug, especially at the highest concentration. In contrast, the conjugates with oxime-linked Dau showed no or only a minor toxicity on both the cell lines (Table 2). These data confirm that the linkage between the payload and homing peptide has a significant influence on early drug release and, consequently, an undesired toxic side effect. We may also conclude that the search for more suitable homing peptides might be more important than the application of cleavable bonds between the drug and the peptide to develop efficient DDSs.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[3D-Tic, 4Lys(Bu), 8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.56 ± 0.51 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 12.73 ± 3.10 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[3D-Tic, 4Lys(Bu), 7D-Trp 8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.57 ± 0.47 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 16.83 ± 0.66 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[2ΔHis, 3D-Tic, 8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.75 ± 0.17 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.35 ± 1.93 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 10.32 ± 1.32 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[2ΔHis, 3D-Tic, 4Lys(Bu), 7D-Trp 8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.81 ± 0.04 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8.18 ± 0.18 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 16.55 ± 0.30 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[2ΔHis-3D-Tic, 8Lys(glutaryl-Val-Ala-PABC-Dau conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.85 ± 0.90 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 20.54 ± 1.46 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate cancer | Human prostate cancer cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 24.77 ± 1.73 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Normal human pituitary cell | Homo sapiens | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | ||
GnRH-III-[3D-Tic, 8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.89 ± 0.62 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8.75 ± 0.86 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
[4Lys(Dau=Aoa),8Lys(Dau=Aoa)]-GnRH-III [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.9 ± 0.9 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
As a payload for PDCs (and ADCs), analogues of the duocarmycins are attractive. Duocarmycin SA and yatakemycin rank among the most potent natural cytotoxins discovered. The cyclopropyl and prodrug seco forms are both naturally occurring and equipotent in most circumstances. Studies of the binding-driven bonding model of their interaction with DNA suggest that their utility will be enhanced when targeted to tumor cells. In fact, SYD985, an ADC that utilizes a peptide linker for a duocarmycin analogue to trastuzumab, has recently been progressed to phase III clinical trial.
Click to Show/Hide
|
||||
| Description |
The main drug-related toxic side effect of anthracyclines is cardiotoxicity, leading to cardiomyopathy and heart failure. Comparative experiments to determine the cardiotoxicity of peptide-anthracycline conjugates with different linkages might be informative for the other conjugates with different types of drugs as well. For this purpose, human cardiomyocytes (HCM) and human umbilical vein endothelial cells (HUVEC) were used as models. The long-term (0-72 h) cytotoxic effect of sixteen GnRH-based conjugates containing Dox and Dau was determined by real-time impedimetric sensing using the xCELLigence SP system (ACEA Biosciences, San Diego, CA, USA) [75]. The results indicated that the ester-linked GnRH-Dox conjugates, including Zoptarelin Doxorubicin, showed significant toxicity at 100 nM and 1 uM, which was remarkably pronounced on the HCM cells. The cytotoxic effect was comparable to that of the free drug, especially at the highest concentration. In contrast, the conjugates with oxime-linked Dau showed no or only a minor toxicity on both the cell lines (Table 2). These data confirm that the linkage between the payload and homing peptide has a significant influence on early drug release and, consequently, an undesired toxic side effect. We may also conclude that the search for more suitable homing peptides might be more important than the application of cleavable bonds between the drug and the peptide to develop efficient DDSs.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 6.8 ± 1.0 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
The main drug-related toxic side effect of anthracyclines is cardiotoxicity, leading to cardiomyopathy and heart failure. Comparative experiments to determine the cardiotoxicity of peptide-anthracycline conjugates with different linkages might be informative for the other conjugates with different types of drugs as well. For this purpose, human cardiomyocytes (HCM) and human umbilical vein endothelial cells (HUVEC) were used as models. The long-term (0-72 h) cytotoxic effect of sixteen GnRH-based conjugates containing Dox and Dau was determined by real-time impedimetric sensing using the xCELLigence SP system (ACEA Biosciences, San Diego, CA, USA) [75]. The results indicated that the ester-linked GnRH-Dox conjugates, including Zoptarelin Doxorubicin, showed significant toxicity at 100 nM and 1 uM, which was remarkably pronounced on the HCM cells. The cytotoxic effect was comparable to that of the free drug, especially at the highest concentration. In contrast, the conjugates with oxime-linked Dau showed no or only a minor toxicity on both the cell lines (Table 2). These data confirm that the linkage between the payload and homing peptide has a significant influence on early drug release and, consequently, an undesired toxic side effect. We may also conclude that the search for more suitable homing peptides might be more important than the application of cleavable bonds between the drug and the peptide to develop efficient DDSs.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
Dau=Aoa-SYSNleEHFRWGKPV-NH2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 2.9 ± 0.6 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | B16 cell | CVCL_F936 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.8 ± 5.4 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | A2058 cell | CVCL_1059 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.9 ± 1.5 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Melanoma | WM983B cell | CVCL_6809 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Melanoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 12.8 ± 1.6 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
In this study, the native sequence of -MSH was used, in which Met was replaced by Nle (SYSNleEHFRWGKPV). Daunomycin was attached to the amino functional groups via non-cleavable oxime linkage. These types of conjugates have many excellent properties to study and compare the bioactivity of the compounds. One of the developed conjugates contained drug molecules on both conjugation site (N-terminal and the side chain of Lys). The two additional conjugates were modified by the drug, either on theN-terminus or on the Lys side chain. The in vitro cytostatic effect on mouse melanoma cells did not show any significant differences among them. However, results indicated that the conjugates with Dau on the side chain of Lys could enter the cells more rapidly and efficiently. In contrast, the in vivo tumor growth inhibition was the most pronounced in the case of Ac-SYSNleEHFRWGK(Dau=Aoa)PV-NH2(Conj2). It is worth mentioning that the dose was normalized to Dau content, thus the injected dose ofConj3was half that ofConj 2. Nevertheless,Conj2, with one molecule of the drug, might be superior to the conjugate containing two molecules of daunomycin. The higher tumor growth inhibition effect ofConj2overConj1where the Dau is connected to theN-terminus, confirms our previous results with Dau-GnRH-III (Glp-His-Trp-Lys(Bu)-His-Asp-Trp-Lys(Dau=Aoa)-Pro-Gly-NH2), suggesting that H-Lys(Dau=Aoa)-OH can release easily from this position by the dipeptidyl-peptidase activity of cathepsin B.
Click to Show/Hide
|
||||
| Description |
In vitro studies of the antiproliferative effect showed that melanoma-targeting Dau-conjugates have significantly higher efficacy on B16 murine melanoma cell lines in comparison to human melanoma cell lines (A2058, M24, and WM983B). The IC50 values were detected between 2 and 2.9 μM on B16 cells, where the highest activity was detected in the case of conjugate Conj3, followed by Conj2 and Conj1. Moreover, the relative potencies of conjugates to free Dau were also calculated as independent values from cell lines. A higher value of relative potency indicated the elevated targeting capacity of the conjugate on particular cell lines. Considering relative potencies, the best targeting capacity was shown to be Conj3 on all four cell lines, followed by Conj2 and Conj1. The highest antitumor activity of Conj3 on cells can be explained by the presence of two Dau molecules in this conjugate in comparison with Conj2 and Conj1, which contain only one drug molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | M24 cell | CVCL_D032 | ||
[8Lys(Dau=Aoa-K(Dau=Aoa))]GnRH-III [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.0 ± 0.4 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The cancer phenotype is commonly associated with aberrant glycosylation patterns. One glycan that is directly linked to cancer is the Thomsen-Friedenreich antigen (TF or CD176). It is a disaccharide composed of a galactose β1-3 N-acetylgalactosamine, O-linked to a glycoprotein through serine or threonine residues and commonly written as Galβ1-3GalNAc--O-Ser/Thr. The TF is therapeutically attractive due to its cryptic nature in normal cells and exposure in embryonic and cancer cells. The expression of the TF has been demonstrated in 90% of primary human carcinomas, including in the lung, the breast, and the pancreas. Additionally, cancer initiating cells or cancer stem cells in the lung, liver, and colon express the TF. The peptide sequence HGRFILPWWYAFSPS (TF-peptide) is known to bind tightly to the TF (Kd = 1.2 M) and has been demonstrated to inhibit processes directly involved in TF accessibility.
Click to Show/Hide
|
||||
| Description |
The main drug-related toxic side effect of anthracyclines is cardiotoxicity, leading to cardiomyopathy and heart failure. Comparative experiments to determine the cardiotoxicity of peptide-anthracycline conjugates with different linkages might be informative for the other conjugates with different types of drugs as well. For this purpose, human cardiomyocytes (HCM) and human umbilical vein endothelial cells (HUVEC) were used as models. The long-term (0-72 h) cytotoxic effect of sixteen GnRH-based conjugates containing Dox and Dau was determined by real-time impedimetric sensing using the xCELLigence SP system (ACEA Biosciences, San Diego, CA, USA) [75]. The results indicated that the ester-linked GnRH-Dox conjugates, including Zoptarelin Doxorubicin, showed significant toxicity at 100 nM and 1 uM, which was remarkably pronounced on the HCM cells. The cytotoxic effect was comparable to that of the free drug, especially at the highest concentration. In contrast, the conjugates with oxime-linked Dau showed no or only a minor toxicity on both the cell lines (Table 2). These data confirm that the linkage between the payload and homing peptide has a significant influence on early drug release and, consequently, an undesired toxic side effect. We may also conclude that the search for more suitable homing peptides might be more important than the application of cleavable bonds between the drug and the peptide to develop efficient DDSs.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.6 ± 2.0 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
The main drug-related toxic side effect of anthracyclines is cardiotoxicity, leading to cardiomyopathy and heart failure. Comparative experiments to determine the cardiotoxicity of peptide-anthracycline conjugates with different linkages might be informative for the other conjugates with different types of drugs as well. For this purpose, human cardiomyocytes (HCM) and human umbilical vein endothelial cells (HUVEC) were used as models. The long-term (0-72 h) cytotoxic effect of sixteen GnRH-based conjugates containing Dox and Dau was determined by real-time impedimetric sensing using the xCELLigence SP system (ACEA Biosciences, San Diego, CA, USA) [75]. The results indicated that the ester-linked GnRH-Dox conjugates, including Zoptarelin Doxorubicin, showed significant toxicity at 100 nM and 1 uM, which was remarkably pronounced on the HCM cells. The cytotoxic effect was comparable to that of the free drug, especially at the highest concentration. In contrast, the conjugates with oxime-linked Dau showed no or only a minor toxicity on both the cell lines (Table 2). These data confirm that the linkage between the payload and homing peptide has a significant influence on early drug release and, consequently, an undesired toxic side effect. We may also conclude that the search for more suitable homing peptides might be more important than the application of cleavable bonds between the drug and the peptide to develop efficient DDSs.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
[4Lys(Ac),8Lys(Dau=Aoa)]-GnRH-III [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.1 ± 1.7 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The cancer phenotype is commonly associated with aberrant glycosylation patterns. One glycan that is directly linked to cancer is the Thomsen-Friedenreich antigen (TF or CD176). It is a disaccharide composed of a galactose β1-3 N-acetylgalactosamine, O-linked to a glycoprotein through serine or threonine residues and commonly written as Galβ1-3GalNAc--O-Ser/Thr. The TF is therapeutically attractive due to its cryptic nature in normal cells and exposure in embryonic and cancer cells. The expression of the TF has been demonstrated in 90% of primary human carcinomas, including in the lung, the breast, and the pancreas. Additionally, cancer initiating cells or cancer stem cells in the lung, liver, and colon express the TF. The peptide sequence HGRFILPWWYAFSPS (TF-peptide) is known to bind tightly to the TF (Kd = 1.2 M) and has been demonstrated to inhibit processes directly involved in TF accessibility.
Click to Show/Hide
|
||||
| Description |
The main drug-related toxic side effect of anthracyclines is cardiotoxicity, leading to cardiomyopathy and heart failure. Comparative experiments to determine the cardiotoxicity of peptide-anthracycline conjugates with different linkages might be informative for the other conjugates with different types of drugs as well. For this purpose, human cardiomyocytes (HCM) and human umbilical vein endothelial cells (HUVEC) were used as models. The long-term (0-72 h) cytotoxic effect of sixteen GnRH-based conjugates containing Dox and Dau was determined by real-time impedimetric sensing using the xCELLigence SP system (ACEA Biosciences, San Diego, CA, USA) [75]. The results indicated that the ester-linked GnRH-Dox conjugates, including Zoptarelin Doxorubicin, showed significant toxicity at 100 nM and 1 uM, which was remarkably pronounced on the HCM cells. The cytotoxic effect was comparable to that of the free drug, especially at the highest concentration. In contrast, the conjugates with oxime-linked Dau showed no or only a minor toxicity on both the cell lines (Table 2). These data confirm that the linkage between the payload and homing peptide has a significant influence on early drug release and, consequently, an undesired toxic side effect. We may also conclude that the search for more suitable homing peptides might be more important than the application of cleavable bonds between the drug and the peptide to develop efficient DDSs.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 7.4 ± 2.6 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
In our experiments, GnRH-III was applied as a targeting peptide. The conjugate [8Lys(Dau=Aoa-GFLG)]-GnRH-III, in which Dau was connected to the side chain of Lys in position 8 through an aminooxyacetylated cathepsin B-labile GFLG spacer, showed significant tumor growth inhibition in s.c.-developed fast-growing C26 murine colon cancer-bearing Balb/c mice. The effect highly depended on the treatment schedule. In the first attempt, the conjugate was administered i.p. at a dose of 8.86 umol (5 mg Dau-content/kg body weight) five times on days 7, 9, 11, 14 and 16 after tumor transplantation (Treatment schedule A). The tumor volume inhibition was only 15.7% on day 26 when the experiment finished. A slight enhancement in inhibition (22.3%) was detected when a dose of conjugate corresponding to 15 mg Dau content/kg body weight (26.6 μmol) was injected only once on day 7 (Treatment schedule B). An additional treatment on day 10 (Treatment schedule C) did not result in any further improvements (21.9% on day 29). However, when the treatment schedule was changed to two treatments with the same dose on days 4 and 7, 46.3% inhibition was observed on day 29 (Treatment schedule D). In contrast, the treatments with free Dau at a dose of 2 mg/kg body weight (3.55 umol) on days 7, 9, 11, 14 and 16 showed only 22.6% inhibition on day 26. The median survival rates of the treated animals in comparison to the control group were 1 (A), 1.23 (B), 1.21 (C) and 1.38 (D), respectively, and 0.81 for free Dau. These results indicate the lower toxicity and the higher tumor volume inhibition effect of the conjugates in comparison with those of free Dau, as well as the importance of the treatment schedule. In another experiment, HT-29 human colon cancer was developed s.c. in immunodeficient SCID mice. Dau and two conjugates (with or without a GFLG spacer between the GnRH-III homing peptide and the payload) were used for the treatment. The first i.p. administrations were performed on day 13 after tumor inoculation. All the mice treated once with 2.5 mg/kg body weight (4.43 umol) free Dau died within 10 days. In contrast, the conjugates at a dose of 15 mg Dau content/kg body weight (26.6 umol) that refers to 52 mg/kg [8Lys(Dau=Aoa)]-GnRH-III and 62.5 mg/kg [8Lys(Dau=Aoa-GFLG)]-GnRH-III conjugates, respectively, did not show significant toxicity. The treatment was repeated on days 23 and 30. Because of the significant weight loss in several mice in the control group, the experiment was terminated on day 35. The tumor growth inhibition could be calculated as reductions in the tumor volume by 44.3% and 57.6% and the tumor weight by 41% and 50%, respectively. Interestingly, the conjugates were poorly effective on orthotopically developed tumors. In the case of the C26 colon tumor-bearing female Balb/c mice, only a 7% reduction in tumor weight was detected on day 13 after the two treatments on days 4 and 7 with [8Lys(Dau=Aoa)]-GnRH-III, at a 26.6 umol/kg (15 mg Dau content) dose. The effect of free Dau (2 mg/kg on days 4 and 7) showed a better inhibitory effect (24.4%). Interestingly, our novel developed GnRH-III derivative, in which Ser in position 4 was replaced by Lys(Ac), was much more potent, with 49.3% inhibition. It is worth mentioning that the rate of cellular uptake of the [4Lys(Ac), 8Lys(Dau=Aoa)]-GnRH-III conjugate by the tumor cells was significantly higher than that of the conjugate with the native GnRH-III sequence.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
Cyclic NGR peptidedaunomycin conjugates 3 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.3 ± 0.6 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.7 ± 0.9 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8.0 ± 0.1 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 10.3 ± 2.2 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
GnRH-III-[3D-Tic, 7D-Trp 8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.42 ± 0.39 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 15.34 ± 2.63 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[4Lys(Bu), 6Asp(OMe), 8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.44 ± 0.51 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 18.00 ± 0.13 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[3D-Trp, 8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.60 ± 0.28 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 15.25 ± 2.51 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
DauQ=AoaGFLGD4G5HbPG [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [30] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.8 µM | |||
| Evaluation Method | xCELLigence SP system assay | ||||
| Administration Time | 48 h | ||||
| MOA of PDC |
As observed, the cathepsin B labile spacer (GFLG), which was incorporated between the targeting peptide and the daunomycin to ensure the efficient release of the active metabolite, highly increases the hydrophobicity of the conjugates. As demonstrated, the solubility problem of these conjugates can be solved by hydrophilic polymer coupling, not only by using the well-known PEG but also by using the amino-monofunctional HbPG. To the best of our knowledge, 1:1 covalent peptide-polymer conjugates with well-defined monofunctional HbPG have been reported here for the first time. The results of the in vitro cell viability and cellular uptake measurements on HT-29 human colon adenocarcinoma cells prove that the HbPG and the PEG highly influenced the biological activity of the drug-peptide-polymer conjugates. In both peptide conjugate series, one GE11 and one D4 targeting peptide-based conjugate were found with outstanding cellular uptake and cytotoxicity values, namely Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG. According to our results, the PEG is suitable for longer targeting peptides (e.g. GE11), but the G5 spacer is not suitable irrespective of the length of the peptide because it may decrease the biological effect by increasing the flexibility of the polymer and shading of the targeting moiety. In contrast, the use of the hydrophilic hyperbranched polyglycerol (HbPG) is advantageous for short targeting peptides (e.g. D4) but only with a G5 spacer, which provides accessibility of the peptide for receptor binding and cellular uptake resulting in outstanding cytotoxicity.
Click to Show/Hide
|
||||
| Description |
The polymers influenced the cellular uptake of the conjugates in a different manner, but in both groups (GE11 and D4 containing conjugates), one of the compounds was outstanding. The HT-29 cells could uptake Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG to the highest extent. The G5 spacer increased the uptake of the D4-HbPG derivative, presumably because the increased distance between the globular HbPG and the very short peptide sequence provides a better receptor binding. In sharp contrast, the G5 spacer decreases the uptake of the conjugates in all other cases. The most significant difference was observed in the case of GE11-PEG, where the G5 spacer completely demolished the internalization. Probably, here the G5 spacer provides more flexibility for the linear PEG chain resulting in decreased receptor binding. There was one outstanding conjugate from each group (Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG) in the cytotoxicity measurements that correlated well with the results of the internalization studies. These conjugates were found to be the most potent ones in the viability measurement and were proved to be taken up by HT-29 cells the most effectively. Depending on the type of polymer, the incorporation of the G5 spacer had an opposite effect on the cytotoxic activity of the conjugates. In the presence of the G5 spacer, the antitumour activity of the PEGylated conjugates decreased, while the cytotoxicity of the HbPG-containing conjugates increased, especially in the case of those with the D4 targeting peptide. We observed that some of the conjugates (Dau[double bond, length as m-dash]Aoa-GFLG-D4-HbPG and Dau[double bond, length as m-dash]Aoa-GFLG-GE11-G5-PEG) could not cause complete cell death, i.e., ˜0% viability value - characteristic for cell-free culturing medium - was not achieved even at the highest concentration, since their dose-response curves reached a plateau in a lower concentration range. In our opinion, this can be explained by the different characteristics of the highly hydrophobic peptide chain and the highly hydrophilic polymer segment. Due to this amphiphilic character, self-aggregation of the conjugates may occur, which then may block the accessibility of the targeting peptide for receptor binding, thereby decreasing the efficiency of the conjugate as well. This assumption is also confirmed by the turbidity results, since the observed low turbidity values may be caused by the possible formation of nanosized aggregates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
[8Lys(Dau=Aoa-GFLG)]-GnRH-III [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.9 ± 1.2 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
As a payload for PDCs (and ADCs), analogues of the duocarmycins are attractive. Duocarmycin SA and yatakemycin rank among the most potent natural cytotoxins discovered. The cyclopropyl and prodrug seco forms are both naturally occurring and equipotent in most circumstances. Studies of the binding-driven bonding model of their interaction with DNA suggest that their utility will be enhanced when targeted to tumor cells. In fact, SYD985, an ADC that utilizes a peptide linker for a duocarmycin analogue to trastuzumab, has recently been progressed to phase III clinical trial.
Click to Show/Hide
|
||||
| Description |
The main drug-related toxic side effect of anthracyclines is cardiotoxicity, leading to cardiomyopathy and heart failure. Comparative experiments to determine the cardiotoxicity of peptide-anthracycline conjugates with different linkages might be informative for the other conjugates with different types of drugs as well. For this purpose, human cardiomyocytes (HCM) and human umbilical vein endothelial cells (HUVEC) were used as models. The long-term (0-72 h) cytotoxic effect of sixteen GnRH-based conjugates containing Dox and Dau was determined by real-time impedimetric sensing using the xCELLigence SP system (ACEA Biosciences, San Diego, CA, USA) [75]. The results indicated that the ester-linked GnRH-Dox conjugates, including Zoptarelin Doxorubicin, showed significant toxicity at 100 nM and 1 uM, which was remarkably pronounced on the HCM cells. The cytotoxic effect was comparable to that of the free drug, especially at the highest concentration. In contrast, the conjugates with oxime-linked Dau showed no or only a minor toxicity on both the cell lines (Table 2). These data confirm that the linkage between the payload and homing peptide has a significant influence on early drug release and, consequently, an undesired toxic side effect. We may also conclude that the search for more suitable homing peptides might be more important than the application of cleavable bonds between the drug and the peptide to develop efficient DDSs.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [28] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 19.4 ± 3.1 µM | |||
| Administration Time | 72 h | ||||
| MOA of PDC |
The crucial steps during the synthesis of aminooxyacetylated peptides are the incorporation of aminooxyacetic acid (Aoa), including a protecting group, and the final cleavage and the working-up procedure of the Aoa-containing peptide derivatives. In most cases, Boc-protected Aoa is attached to a peptide chain in the last step of solid-phase peptide synthesis. It has been observed that over-acylation (additional Boc-Aoa-OH connected to the Aoa moiety) may occur as the main side reaction. Many approaches have been investigated to overcome this problem, including carbodiimide-mediated one-pot acylation without a base or the application of Boc-Aoa-OSu active ester as an acylating agent, as well as the use of a high excess (8 equiv) of Boc-Aoa-OH and coupling agents for a short acylation time (10 min). Nevertheless, the coupling of the diBoc-protected Aoa derivative has proved to be the best solution. However, the aminooxyacetyl moiety is very sensitive to molecules containing carbonyl groups, with the partial impact of the peptide sequence. Therefore, the free NH2-O-R group reacts often with these compounds during the working-up procedure after the final cleavage of Aoa-modified peptides from the resin. These carbonyl group-containing derivatives might come from the plastic tubes or residues of acetone used in a laboratory. This cannot be prevented even by using diBoc-protected Aoa or working in argon. We found a highly sensitive peptide to this side reaction; the synthesis of a somatostatin analog developed in Schallys laboratory elongated with Aoa (H-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2) was unsuccessful. After several trials to optimize the reaction conditions, we elaborated the following procedure: The semi-protected peptide H-D-Phe-Cys-Tyr-D-Trp-Lys(Dde)-Val-Cys-Thr-NH2 was cleaved from Rink Amide MBHA resin and reacted with Boc-Aoa-OPcp to incorporate Aoa into the N-terminus in a solution. After the efficient coupling reaction, the Dde-protecting group was removed with 2% hydrazine in DMF. Surprisingly, during the cleavage of Dde, a cyclic peptide also formed (Boc-Aoa-D-Phe-c[Cys-Tyr-D-Trp-Lys-Val-Cys]-Thr-NH2). The Boc group was cleaved in 95% TFA solution in the presence of 10 equiv-free Aoa as a carbonyl capture reagent that could prevent the reaction of the peptide with any carbonyl derivative. The crude product was purified by RP-HPLC, and the solvent was evaporated, followed by direct ligation to daunomycin (Dau) in 0.2 M NaOAc solution at pH 5. This procedure proved to be very efficient to prepare oxime-linked Dau-peptide conjugates.
Click to Show/Hide
|
||||
| Description |
In our experiments, GnRH-III was applied as a targeting peptide. The conjugate [8Lys(Dau=Aoa-GFLG)]-GnRH-III, in which Dau was connected to the side chain of Lys in position 8 through an aminooxyacetylated cathepsin B-labile GFLG spacer, showed significant tumor growth inhibition in s.c.-developed fast-growing C26 murine colon cancer-bearing Balb/c mice. The effect highly depended on the treatment schedule. In the first attempt, the conjugate was administered i.p. at a dose of 8.86 umol (5 mg Dau-content/kg body weight) five times on days 7, 9, 11, 14 and 16 after tumor transplantation (Treatment schedule A). The tumor volume inhibition was only 15.7% on day 26 when the experiment finished. A slight enhancement in inhibition (22.3%) was detected when a dose of conjugate corresponding to 15 mg Dau content/kg body weight (26.6 μmol) was injected only once on day 7 (Treatment schedule B). An additional treatment on day 10 (Treatment schedule C) did not result in any further improvements (21.9% on day 29). However, when the treatment schedule was changed to two treatments with the same dose on days 4 and 7, 46.3% inhibition was observed on day 29 (Treatment schedule D). In contrast, the treatments with free Dau at a dose of 2 mg/kg body weight (3.55 umol) on days 7, 9, 11, 14 and 16 showed only 22.6% inhibition on day 26. The median survival rates of the treated animals in comparison to the control group were 1 (A), 1.23 (B), 1.21 (C) and 1.38 (D), respectively, and 0.81 for free Dau. These results indicate the lower toxicity and the higher tumor volume inhibition effect of the conjugates in comparison with those of free Dau, as well as the importance of the treatment schedule. In another experiment, HT-29 human colon cancer was developed s.c. in immunodeficient SCID mice. Dau and two conjugates (with or without a GFLG spacer between the GnRH-III homing peptide and the payload) were used for the treatment. The first i.p. administrations were performed on day 13 after tumor inoculation. All the mice treated once with 2.5 mg/kg body weight (4.43 umol) free Dau died within 10 days. In contrast, the conjugates at a dose of 15 mg Dau content/kg body weight (26.6 umol) that refers to 52 mg/kg [8Lys(Dau=Aoa)]-GnRH-III and 62.5 mg/kg [8Lys(Dau=Aoa-GFLG)]-GnRH-III conjugates, respectively, did not show significant toxicity. The treatment was repeated on days 23 and 30. Because of the significant weight loss in several mice in the control group, the experiment was terminated on day 35. The tumor growth inhibition could be calculated as reductions in the tumor volume by 44.3% and 57.6% and the tumor weight by 41% and 50%, respectively. Interestingly, the conjugates were poorly effective on orthotopically developed tumors. In the case of the C26 colon tumor-bearing female Balb/c mice, only a 7% reduction in tumor weight was detected on day 13 after the two treatments on days 4 and 7 with [8Lys(Dau=Aoa)]-GnRH-III, at a 26.6 umol/kg (15 mg Dau content) dose. The effect of free Dau (2 mg/kg on days 4 and 7) showed a better inhibitory effect (24.4%). Interestingly, our novel developed GnRH-III derivative, in which Ser in position 4 was replaced by Lys(Ac), was much more potent, with 49.3% inhibition. It is worth mentioning that the rate of cellular uptake of the [4Lys(Ac), 8Lys(Dau=Aoa)]-GnRH-III conjugate by the tumor cells was significantly higher than that of the conjugate with the native GnRH-III sequence.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[2ΔHis-3D-Tic,8Lys(glutaryl-Val-Cit-PABC-Dau)] conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.24 ± 1.09 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 10.97 ± 1.23 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate cancer | Human prostate cancer cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 11.70 ± 0.42 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Normal human pituitary cell | Homo sapiens | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | ||
GnRH-III-[6Asp(OMe), 8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.81 ± 0.72 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 10.66 ± 1.76 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[8Lys(Dau=Aoa), 10ΔGly-NH-Et] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 4.88 ± 0.01 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 14.18 ± 3.59 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 3 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 14.33 ± 1.18 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
DauQ=AoaGFLGD4PEG [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [30] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.1 µM | |||
| Evaluation Method | xCELLigence SP system assay | ||||
| Administration Time | 48 h | ||||
| MOA of PDC |
As observed, the cathepsin B labile spacer (GFLG), which was incorporated between the targeting peptide and the daunomycin to ensure the efficient release of the active metabolite, highly increases the hydrophobicity of the conjugates. As demonstrated, the solubility problem of these conjugates can be solved by hydrophilic polymer coupling, not only by using the well-known PEG but also by using the amino-monofunctional HbPG. To the best of our knowledge, 1:1 covalent peptide-polymer conjugates with well-defined monofunctional HbPG have been reported here for the first time. The results of the in vitro cell viability and cellular uptake measurements on HT-29 human colon adenocarcinoma cells prove that the HbPG and the PEG highly influenced the biological activity of the drug-peptide-polymer conjugates. In both peptide conjugate series, one GE11 and one D4 targeting peptide-based conjugate were found with outstanding cellular uptake and cytotoxicity values, namely Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG. According to our results, the PEG is suitable for longer targeting peptides (e.g. GE11), but the G5 spacer is not suitable irrespective of the length of the peptide because it may decrease the biological effect by increasing the flexibility of the polymer and shading of the targeting moiety. In contrast, the use of the hydrophilic hyperbranched polyglycerol (HbPG) is advantageous for short targeting peptides (e.g. D4) but only with a G5 spacer, which provides accessibility of the peptide for receptor binding and cellular uptake resulting in outstanding cytotoxicity.
Click to Show/Hide
|
||||
| Description |
The polymers influenced the cellular uptake of the conjugates in a different manner, but in both groups (GE11 and D4 containing conjugates), one of the compounds was outstanding. The HT-29 cells could uptake Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG to the highest extent. The G5 spacer increased the uptake of the D4-HbPG derivative, presumably because the increased distance between the globular HbPG and the very short peptide sequence provides a better receptor binding. In sharp contrast, the G5 spacer decreases the uptake of the conjugates in all other cases. The most significant difference was observed in the case of GE11-PEG, where the G5 spacer completely demolished the internalization. Probably, here the G5 spacer provides more flexibility for the linear PEG chain resulting in decreased receptor binding. There was one outstanding conjugate from each group (Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG) in the cytotoxicity measurements that correlated well with the results of the internalization studies. These conjugates were found to be the most potent ones in the viability measurement and were proved to be taken up by HT-29 cells the most effectively. Depending on the type of polymer, the incorporation of the G5 spacer had an opposite effect on the cytotoxic activity of the conjugates. In the presence of the G5 spacer, the antitumour activity of the PEGylated conjugates decreased, while the cytotoxicity of the HbPG-containing conjugates increased, especially in the case of those with the D4 targeting peptide. We observed that some of the conjugates (Dau[double bond, length as m-dash]Aoa-GFLG-D4-HbPG and Dau[double bond, length as m-dash]Aoa-GFLG-GE11-G5-PEG) could not cause complete cell death, i.e., ˜0% viability value - characteristic for cell-free culturing medium - was not achieved even at the highest concentration, since their dose-response curves reached a plateau in a lower concentration range. In our opinion, this can be explained by the different characteristics of the highly hydrophobic peptide chain and the highly hydrophilic polymer segment. Due to this amphiphilic character, self-aggregation of the conjugates may occur, which then may block the accessibility of the targeting peptide for receptor binding, thereby decreasing the efficiency of the conjugate as well. This assumption is also confirmed by the turbidity results, since the observed low turbidity values may be caused by the possible formation of nanosized aggregates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
DauQ=AoaGFLGGE11G5HbPG [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [30] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 6 µM | |||
| Evaluation Method | xCELLigence SP system assay | ||||
| Administration Time | 48 h | ||||
| MOA of PDC |
As observed, the cathepsin B labile spacer (GFLG), which was incorporated between the targeting peptide and the daunomycin to ensure the efficient release of the active metabolite, highly increases the hydrophobicity of the conjugates. As demonstrated, the solubility problem of these conjugates can be solved by hydrophilic polymer coupling, not only by using the well-known PEG but also by using the amino-monofunctional HbPG. To the best of our knowledge, 1:1 covalent peptide-polymer conjugates with well-defined monofunctional HbPG have been reported here for the first time. The results of the in vitro cell viability and cellular uptake measurements on HT-29 human colon adenocarcinoma cells prove that the HbPG and the PEG highly influenced the biological activity of the drug-peptide-polymer conjugates. In both peptide conjugate series, one GE11 and one D4 targeting peptide-based conjugate were found with outstanding cellular uptake and cytotoxicity values, namely Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG. According to our results, the PEG is suitable for longer targeting peptides (e.g. GE11), but the G5 spacer is not suitable irrespective of the length of the peptide because it may decrease the biological effect by increasing the flexibility of the polymer and shading of the targeting moiety. In contrast, the use of the hydrophilic hyperbranched polyglycerol (HbPG) is advantageous for short targeting peptides (e.g. D4) but only with a G5 spacer, which provides accessibility of the peptide for receptor binding and cellular uptake resulting in outstanding cytotoxicity.
Click to Show/Hide
|
||||
| Description |
The polymers influenced the cellular uptake of the conjugates in a different manner, but in both groups (GE11 and D4 containing conjugates), one of the compounds was outstanding. The HT-29 cells could uptake Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG to the highest extent. The G5 spacer increased the uptake of the D4-HbPG derivative, presumably because the increased distance between the globular HbPG and the very short peptide sequence provides a better receptor binding. In sharp contrast, the G5 spacer decreases the uptake of the conjugates in all other cases. The most significant difference was observed in the case of GE11-PEG, where the G5 spacer completely demolished the internalization. Probably, here the G5 spacer provides more flexibility for the linear PEG chain resulting in decreased receptor binding. There was one outstanding conjugate from each group (Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG) in the cytotoxicity measurements that correlated well with the results of the internalization studies. These conjugates were found to be the most potent ones in the viability measurement and were proved to be taken up by HT-29 cells the most effectively. Depending on the type of polymer, the incorporation of the G5 spacer had an opposite effect on the cytotoxic activity of the conjugates. In the presence of the G5 spacer, the antitumour activity of the PEGylated conjugates decreased, while the cytotoxicity of the HbPG-containing conjugates increased, especially in the case of those with the D4 targeting peptide. We observed that some of the conjugates (Dau[double bond, length as m-dash]Aoa-GFLG-D4-HbPG and Dau[double bond, length as m-dash]Aoa-GFLG-GE11-G5-PEG) could not cause complete cell death, i.e., ˜0% viability value - characteristic for cell-free culturing medium - was not achieved even at the highest concentration, since their dose-response curves reached a plateau in a lower concentration range. In our opinion, this can be explained by the different characteristics of the highly hydrophobic peptide chain and the highly hydrophilic polymer segment. Due to this amphiphilic character, self-aggregation of the conjugates may occur, which then may block the accessibility of the targeting peptide for receptor binding, thereby decreasing the efficiency of the conjugate as well. This assumption is also confirmed by the turbidity results, since the observed low turbidity values may be caused by the possible formation of nanosized aggregates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[3D-Trp, 4Lys(Bu), 8Lys(Dau=Aoa)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 6.64 ± 1.58 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [29] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 15.03 ± 2.51 µM | |||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
GnRH-III-[2His-3Trp,8Lys(glutaryl-Val-Ala-PABC-Dau)] conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 7.48 ± 0.66 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 15.46 ± 0.83 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate cancer | Human prostate cancer cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 17.04 ± 1.04 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Normal human pituitary cell | Homo sapiens | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 56.19 ± 17.28 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | ||
Dau=Aoa-TFFYGGSRGK(Dau=Aoa)RNNFKTEEY-OH [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [32] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 7.8 ± 6.3 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The blood-brain barrier (BBB) is a semipermeable system, and, therefore, most of the active substances are poorly transported through this barrier, resulting in decreased therapeutic effects. Angiopep-2 (TFFYGGSRGKRNNFKTEEY) is a peptide ligand of low-density lipoprotein receptor-related protein-1 (LRP1), which can cross the BBB via receptor-mediated transcytosis and simultaneously target glioblastomas. Angiopep-2 contains three amino groups that have previously been used to produce drug-peptide conjugates, although the role and importance of each position have not yet been investigated. Thus, we studied the number and position of drug molecules in Angiopep-2 based conjugates. Conjugates containing one, two, and three daunomycin molecules conjugated via oxime linkage in all possible variations were prepared. The in vitro cytostatic effect and cellular uptake of the conjugates were investigated on U87 human glioblastoma cells. Degradation studies in the presence of rat liver lysosomal homogenates were also performed in order for us to better understand the structure-activity relationship and to determine the smallest metabolites. Conjugates with the best cytostatic effects had a drug molecule at the N-terminus. We demonstrated that the increasing number of drug molecules does not necessarily increase the efficacy of the conjugates, and proved that modification of the different conjugation sites results in differing biological effectiveness.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effect of the synthesized peptide (1) and daunomycin-peptide conjugates (2-8) was investigated on U87 human glioblastoma cells. The cells were treated with the peptide and conjugates at different concentrations (0.05-50 uM) for 24 h, and after a washing step, the cells were incubated for another 48 h at 37 C. The cytostatic effect of the compounds was determined using the MTT test. The measured IC50values are shown in. The free Angiopep-2 did not show any effect on tumor cells up to a 50 μM concentration. However, no clear correlation could be obtained between the number of the daunomycin and the cytostatic efficacy. Among the conjugates with only one Dau, compound3,in which Dau was attached to theN-terminus, showed the highest in vitro cytostatic effect on glioblastoma cells. When the Dau was conjugated to the Lys side chain in position 15 (compound2), the effect decreased significantly, and conjugate4was also not significantly better. In case of conjugates with two drug molecules, a similar tendency was observed. Compound7,with Dau at theN-terminus and on the Lys side chain in position 10, showed the highest activity. When the Lys side chains were used as conjugation sites, the formed compound (6) had moderate activity on U87 cells. Surprisingly, conjugate5,with Dau at theN-terminus and Lys side chain in position 15, showed the lowest efficacy on the U87 cells. Compound8,with three Dau, showed moderate activity. Thus, the effectiveness of Angiopep-2-daunomycin conjugates does not primarily depend on the number of drug molecules, but rather on their position within the molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
GnRH-III-[2His-3Trp,8Lys(glutaryl-Val-Cit-PABC-diamine-PTX)] conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8.61 ± 1.08 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate cancer | Human prostate cancer cells | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.70 ± 1.07 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Normal human pituitary cell | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 67.88 ± 25.36 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | ||
DauQ=AoaGFLGGE11G5PEG [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [30] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.1 µM | |||
| Evaluation Method | xCELLigence SP system assay | ||||
| Administration Time | 48 h | ||||
| MOA of PDC |
As observed, the cathepsin B labile spacer (GFLG), which was incorporated between the targeting peptide and the daunomycin to ensure the efficient release of the active metabolite, highly increases the hydrophobicity of the conjugates. As demonstrated, the solubility problem of these conjugates can be solved by hydrophilic polymer coupling, not only by using the well-known PEG but also by using the amino-monofunctional HbPG. To the best of our knowledge, 1:1 covalent peptide-polymer conjugates with well-defined monofunctional HbPG have been reported here for the first time. The results of the in vitro cell viability and cellular uptake measurements on HT-29 human colon adenocarcinoma cells prove that the HbPG and the PEG highly influenced the biological activity of the drug-peptide-polymer conjugates. In both peptide conjugate series, one GE11 and one D4 targeting peptide-based conjugate were found with outstanding cellular uptake and cytotoxicity values, namely Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG. According to our results, the PEG is suitable for longer targeting peptides (e.g. GE11), but the G5 spacer is not suitable irrespective of the length of the peptide because it may decrease the biological effect by increasing the flexibility of the polymer and shading of the targeting moiety. In contrast, the use of the hydrophilic hyperbranched polyglycerol (HbPG) is advantageous for short targeting peptides (e.g. D4) but only with a G5 spacer, which provides accessibility of the peptide for receptor binding and cellular uptake resulting in outstanding cytotoxicity.
Click to Show/Hide
|
||||
| Description |
The polymers influenced the cellular uptake of the conjugates in a different manner, but in both groups (GE11 and D4 containing conjugates), one of the compounds was outstanding. The HT-29 cells could uptake Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG to the highest extent. The G5 spacer increased the uptake of the D4-HbPG derivative, presumably because the increased distance between the globular HbPG and the very short peptide sequence provides a better receptor binding. In sharp contrast, the G5 spacer decreases the uptake of the conjugates in all other cases. The most significant difference was observed in the case of GE11-PEG, where the G5 spacer completely demolished the internalization. Probably, here the G5 spacer provides more flexibility for the linear PEG chain resulting in decreased receptor binding. There was one outstanding conjugate from each group (Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG) in the cytotoxicity measurements that correlated well with the results of the internalization studies. These conjugates were found to be the most potent ones in the viability measurement and were proved to be taken up by HT-29 cells the most effectively. Depending on the type of polymer, the incorporation of the G5 spacer had an opposite effect on the cytotoxic activity of the conjugates. In the presence of the G5 spacer, the antitumour activity of the PEGylated conjugates decreased, while the cytotoxicity of the HbPG-containing conjugates increased, especially in the case of those with the D4 targeting peptide. We observed that some of the conjugates (Dau[double bond, length as m-dash]Aoa-GFLG-D4-HbPG and Dau[double bond, length as m-dash]Aoa-GFLG-GE11-G5-PEG) could not cause complete cell death, i.e., ˜0% viability value - characteristic for cell-free culturing medium - was not achieved even at the highest concentration, since their dose-response curves reached a plateau in a lower concentration range. In our opinion, this can be explained by the different characteristics of the highly hydrophobic peptide chain and the highly hydrophilic polymer segment. Due to this amphiphilic character, self-aggregation of the conjugates may occur, which then may block the accessibility of the targeting peptide for receptor binding, thereby decreasing the efficiency of the conjugate as well. This assumption is also confirmed by the turbidity results, since the observed low turbidity values may be caused by the possible formation of nanosized aggregates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
DauQ=AoaGFLGGE11HbPG [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [30] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 9.3 µM | |||
| Evaluation Method | xCELLigence SP system assay | ||||
| Administration Time | 48 h | ||||
| MOA of PDC |
As observed, the cathepsin B labile spacer (GFLG), which was incorporated between the targeting peptide and the daunomycin to ensure the efficient release of the active metabolite, highly increases the hydrophobicity of the conjugates. As demonstrated, the solubility problem of these conjugates can be solved by hydrophilic polymer coupling, not only by using the well-known PEG but also by using the amino-monofunctional HbPG. To the best of our knowledge, 1:1 covalent peptide-polymer conjugates with well-defined monofunctional HbPG have been reported here for the first time. The results of the in vitro cell viability and cellular uptake measurements on HT-29 human colon adenocarcinoma cells prove that the HbPG and the PEG highly influenced the biological activity of the drug-peptide-polymer conjugates. In both peptide conjugate series, one GE11 and one D4 targeting peptide-based conjugate were found with outstanding cellular uptake and cytotoxicity values, namely Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG. According to our results, the PEG is suitable for longer targeting peptides (e.g. GE11), but the G5 spacer is not suitable irrespective of the length of the peptide because it may decrease the biological effect by increasing the flexibility of the polymer and shading of the targeting moiety. In contrast, the use of the hydrophilic hyperbranched polyglycerol (HbPG) is advantageous for short targeting peptides (e.g. D4) but only with a G5 spacer, which provides accessibility of the peptide for receptor binding and cellular uptake resulting in outstanding cytotoxicity.
Click to Show/Hide
|
||||
| Description |
The polymers influenced the cellular uptake of the conjugates in a different manner, but in both groups (GE11 and D4 containing conjugates), one of the compounds was outstanding. The HT-29 cells could uptake Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG to the highest extent. The G5 spacer increased the uptake of the D4-HbPG derivative, presumably because the increased distance between the globular HbPG and the very short peptide sequence provides a better receptor binding. In sharp contrast, the G5 spacer decreases the uptake of the conjugates in all other cases. The most significant difference was observed in the case of GE11-PEG, where the G5 spacer completely demolished the internalization. Probably, here the G5 spacer provides more flexibility for the linear PEG chain resulting in decreased receptor binding. There was one outstanding conjugate from each group (Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG) in the cytotoxicity measurements that correlated well with the results of the internalization studies. These conjugates were found to be the most potent ones in the viability measurement and were proved to be taken up by HT-29 cells the most effectively. Depending on the type of polymer, the incorporation of the G5 spacer had an opposite effect on the cytotoxic activity of the conjugates. In the presence of the G5 spacer, the antitumour activity of the PEGylated conjugates decreased, while the cytotoxicity of the HbPG-containing conjugates increased, especially in the case of those with the D4 targeting peptide. We observed that some of the conjugates (Dau[double bond, length as m-dash]Aoa-GFLG-D4-HbPG and Dau[double bond, length as m-dash]Aoa-GFLG-GE11-G5-PEG) could not cause complete cell death, i.e., ˜0% viability value - characteristic for cell-free culturing medium - was not achieved even at the highest concentration, since their dose-response curves reached a plateau in a lower concentration range. In our opinion, this can be explained by the different characteristics of the highly hydrophobic peptide chain and the highly hydrophilic polymer segment. Due to this amphiphilic character, self-aggregation of the conjugates may occur, which then may block the accessibility of the targeting peptide for receptor binding, thereby decreasing the efficiency of the conjugate as well. This assumption is also confirmed by the turbidity results, since the observed low turbidity values may be caused by the possible formation of nanosized aggregates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
Dau=Aoa-TFFYGGSRGKRNNFKTEEY-OH [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [32] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 10.9 ± 2.8 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The blood-brain barrier (BBB) is a semipermeable system, and, therefore, most of the active substances are poorly transported through this barrier, resulting in decreased therapeutic effects. Angiopep-2 (TFFYGGSRGKRNNFKTEEY) is a peptide ligand of low-density lipoprotein receptor-related protein-1 (LRP1), which can cross the BBB via receptor-mediated transcytosis and simultaneously target glioblastomas. Angiopep-2 contains three amino groups that have previously been used to produce drug-peptide conjugates, although the role and importance of each position have not yet been investigated. Thus, we studied the number and position of drug molecules in Angiopep-2 based conjugates. Conjugates containing one, two, and three daunomycin molecules conjugated via oxime linkage in all possible variations were prepared. The in vitro cytostatic effect and cellular uptake of the conjugates were investigated on U87 human glioblastoma cells. Degradation studies in the presence of rat liver lysosomal homogenates were also performed in order for us to better understand the structure-activity relationship and to determine the smallest metabolites. Conjugates with the best cytostatic effects had a drug molecule at the N-terminus. We demonstrated that the increasing number of drug molecules does not necessarily increase the efficacy of the conjugates, and proved that modification of the different conjugation sites results in differing biological effectiveness.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effect of the synthesized peptide (1) and daunomycin-peptide conjugates (2-8) was investigated on U87 human glioblastoma cells. The cells were treated with the peptide and conjugates at different concentrations (0.05-50 uM) for 24 h, and after a washing step, the cells were incubated for another 48 h at 37 C. The cytostatic effect of the compounds was determined using the MTT test. The measured IC50values are shown in. The free Angiopep-2 did not show any effect on tumor cells up to a 50 μM concentration. However, no clear correlation could be obtained between the number of the daunomycin and the cytostatic efficacy. Among the conjugates with only one Dau, compound3,in which Dau was attached to theN-terminus, showed the highest in vitro cytostatic effect on glioblastoma cells. When the Dau was conjugated to the Lys side chain in position 15 (compound2), the effect decreased significantly, and conjugate4was also not significantly better. In case of conjugates with two drug molecules, a similar tendency was observed. Compound7,with Dau at theN-terminus and on the Lys side chain in position 10, showed the highest activity. When the Lys side chains were used as conjugation sites, the formed compound (6) had moderate activity on U87 cells. Surprisingly, conjugate5,with Dau at theN-terminus and Lys side chain in position 15, showed the lowest efficacy on the U87 cells. Compound8,with three Dau, showed moderate activity. Thus, the effectiveness of Angiopep-2-daunomycin conjugates does not primarily depend on the number of drug molecules, but rather on their position within the molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
GnRH-III-[2His-3Trp,8Lys(glutaryl-Val-Cit-PABC-Dau)] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 11.18 ± 0.38 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 13.66 ± 1.87 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate cancer | Human prostate cancer cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 16.12 ± 2.03 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Normal human pituitary cell | Homo sapiens | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 85.57 ± 24.33 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | ||
DauQ=AoaGFLGD4G5PEG [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [30] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 13.5 µM | |||
| Evaluation Method | xCELLigence SP system assay | ||||
| Administration Time | 48 h | ||||
| MOA of PDC |
As observed, the cathepsin B labile spacer (GFLG), which was incorporated between the targeting peptide and the daunomycin to ensure the efficient release of the active metabolite, highly increases the hydrophobicity of the conjugates. As demonstrated, the solubility problem of these conjugates can be solved by hydrophilic polymer coupling, not only by using the well-known PEG but also by using the amino-monofunctional HbPG. To the best of our knowledge, 1:1 covalent peptide-polymer conjugates with well-defined monofunctional HbPG have been reported here for the first time. The results of the in vitro cell viability and cellular uptake measurements on HT-29 human colon adenocarcinoma cells prove that the HbPG and the PEG highly influenced the biological activity of the drug-peptide-polymer conjugates. In both peptide conjugate series, one GE11 and one D4 targeting peptide-based conjugate were found with outstanding cellular uptake and cytotoxicity values, namely Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG. According to our results, the PEG is suitable for longer targeting peptides (e.g. GE11), but the G5 spacer is not suitable irrespective of the length of the peptide because it may decrease the biological effect by increasing the flexibility of the polymer and shading of the targeting moiety. In contrast, the use of the hydrophilic hyperbranched polyglycerol (HbPG) is advantageous for short targeting peptides (e.g. D4) but only with a G5 spacer, which provides accessibility of the peptide for receptor binding and cellular uptake resulting in outstanding cytotoxicity.
Click to Show/Hide
|
||||
| Description |
The polymers influenced the cellular uptake of the conjugates in a different manner, but in both groups (GE11 and D4 containing conjugates), one of the compounds was outstanding. The HT-29 cells could uptake Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG to the highest extent. The G5 spacer increased the uptake of the D4-HbPG derivative, presumably because the increased distance between the globular HbPG and the very short peptide sequence provides a better receptor binding. In sharp contrast, the G5 spacer decreases the uptake of the conjugates in all other cases. The most significant difference was observed in the case of GE11-PEG, where the G5 spacer completely demolished the internalization. Probably, here the G5 spacer provides more flexibility for the linear PEG chain resulting in decreased receptor binding. There was one outstanding conjugate from each group (Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG) in the cytotoxicity measurements that correlated well with the results of the internalization studies. These conjugates were found to be the most potent ones in the viability measurement and were proved to be taken up by HT-29 cells the most effectively. Depending on the type of polymer, the incorporation of the G5 spacer had an opposite effect on the cytotoxic activity of the conjugates. In the presence of the G5 spacer, the antitumour activity of the PEGylated conjugates decreased, while the cytotoxicity of the HbPG-containing conjugates increased, especially in the case of those with the D4 targeting peptide. We observed that some of the conjugates (Dau[double bond, length as m-dash]Aoa-GFLG-D4-HbPG and Dau[double bond, length as m-dash]Aoa-GFLG-GE11-G5-PEG) could not cause complete cell death, i.e., ˜0% viability value - characteristic for cell-free culturing medium - was not achieved even at the highest concentration, since their dose-response curves reached a plateau in a lower concentration range. In our opinion, this can be explained by the different characteristics of the highly hydrophobic peptide chain and the highly hydrophilic polymer segment. Due to this amphiphilic character, self-aggregation of the conjugates may occur, which then may block the accessibility of the targeting peptide for receptor binding, thereby decreasing the efficiency of the conjugate as well. This assumption is also confirmed by the turbidity results, since the observed low turbidity values may be caused by the possible formation of nanosized aggregates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
Cyclic NGR peptidedaunomycin conjugates 4 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 14.5 ± 2.0 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 45.2 ± 13.2 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | » 50 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | » 50 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
Cyclic NGR peptidedaunomycin conjugates 6 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 15.8 ± 4.4 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 43.7 ± 4.1 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | » 50 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | » 50 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
H-TFFYGGSRGK(Dau=Aoa-GFLG)RNNFKTEEY-OH [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [32] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 16.9 ± 5.6 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The blood-brain barrier (BBB) is a semipermeable system, and, therefore, most of the active substances are poorly transported through this barrier, resulting in decreased therapeutic effects. Angiopep-2 (TFFYGGSRGKRNNFKTEEY) is a peptide ligand of low-density lipoprotein receptor-related protein-1 (LRP1), which can cross the BBB via receptor-mediated transcytosis and simultaneously target glioblastomas. Angiopep-2 contains three amino groups that have previously been used to produce drug-peptide conjugates, although the role and importance of each position have not yet been investigated. Thus, we studied the number and position of drug molecules in Angiopep-2 based conjugates. Conjugates containing one, two, and three daunomycin molecules conjugated via oxime linkage in all possible variations were prepared. The in vitro cytostatic effect and cellular uptake of the conjugates were investigated on U87 human glioblastoma cells. Degradation studies in the presence of rat liver lysosomal homogenates were also performed in order for us to better understand the structure-activity relationship and to determine the smallest metabolites. Conjugates with the best cytostatic effects had a drug molecule at the N-terminus. We demonstrated that the increasing number of drug molecules does not necessarily increase the efficacy of the conjugates, and proved that modification of the different conjugation sites results in differing biological effectiveness.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effect and the in vitro cellular uptake of the spacer-containing daunomycin-peptide conjugates (9-11) were studied on U87 human glioblastoma cells as previously described. The measured IC50 values are shown in Table 2, while the measured cellular uptake at 10 μM and 50 μM concentration are given in Figure 4. The fluorescence intensity values showed good correlation with the percentage of daunomycin-positive cells in the case of these conjugates as well. Conjugate 9 showed slight toxicity during the measurements at a concentration of 50 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
H-TFFYGGSRGK(Dau=Aoa-VAGG)RNNFKTEEY-OH [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [32] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 20.2 ± 3.0 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The blood-brain barrier (BBB) is a semipermeable system, and, therefore, most of the active substances are poorly transported through this barrier, resulting in decreased therapeutic effects. Angiopep-2 (TFFYGGSRGKRNNFKTEEY) is a peptide ligand of low-density lipoprotein receptor-related protein-1 (LRP1), which can cross the BBB via receptor-mediated transcytosis and simultaneously target glioblastomas. Angiopep-2 contains three amino groups that have previously been used to produce drug-peptide conjugates, although the role and importance of each position have not yet been investigated. Thus, we studied the number and position of drug molecules in Angiopep-2 based conjugates. Conjugates containing one, two, and three daunomycin molecules conjugated via oxime linkage in all possible variations were prepared. The in vitro cytostatic effect and cellular uptake of the conjugates were investigated on U87 human glioblastoma cells. Degradation studies in the presence of rat liver lysosomal homogenates were also performed in order for us to better understand the structure-activity relationship and to determine the smallest metabolites. Conjugates with the best cytostatic effects had a drug molecule at the N-terminus. We demonstrated that the increasing number of drug molecules does not necessarily increase the efficacy of the conjugates, and proved that modification of the different conjugation sites results in differing biological effectiveness.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effect and the in vitro cellular uptake of the spacer-containing daunomycin-peptide conjugates (9-11) were studied on U87 human glioblastoma cells as previously described. The measured IC50 values are shown in Table 2, while the measured cellular uptake at 10 μM and 50 μM concentration are given in Figure 4. The fluorescence intensity values showed good correlation with the percentage of daunomycin-positive cells in the case of these conjugates as well. Conjugate 9 showed slight toxicity during the measurements at a concentration of 50 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
Cyclic NGR peptidedaunomycin conjugates 1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 20.6 ± 5.6 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 23.8 ± 0.7 µM | |||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | » 50 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [31] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | » 50 µM | |||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
H-TFFYGGSRGK(Dau=Aoa)RNNFK(Dau=Aoa)TEEY-OH [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [32] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 21.6 ± 5.4 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The blood-brain barrier (BBB) is a semipermeable system, and, therefore, most of the active substances are poorly transported through this barrier, resulting in decreased therapeutic effects. Angiopep-2 (TFFYGGSRGKRNNFKTEEY) is a peptide ligand of low-density lipoprotein receptor-related protein-1 (LRP1), which can cross the BBB via receptor-mediated transcytosis and simultaneously target glioblastomas. Angiopep-2 contains three amino groups that have previously been used to produce drug-peptide conjugates, although the role and importance of each position have not yet been investigated. Thus, we studied the number and position of drug molecules in Angiopep-2 based conjugates. Conjugates containing one, two, and three daunomycin molecules conjugated via oxime linkage in all possible variations were prepared. The in vitro cytostatic effect and cellular uptake of the conjugates were investigated on U87 human glioblastoma cells. Degradation studies in the presence of rat liver lysosomal homogenates were also performed in order for us to better understand the structure-activity relationship and to determine the smallest metabolites. Conjugates with the best cytostatic effects had a drug molecule at the N-terminus. We demonstrated that the increasing number of drug molecules does not necessarily increase the efficacy of the conjugates, and proved that modification of the different conjugation sites results in differing biological effectiveness.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effect of the synthesized peptide (1) and daunomycin-peptide conjugates (2-8) was investigated on U87 human glioblastoma cells. The cells were treated with the peptide and conjugates at different concentrations (0.05-50 uM) for 24 h, and after a washing step, the cells were incubated for another 48 h at 37 C. The cytostatic effect of the compounds was determined using the MTT test. The measured IC50values are shown in. The free Angiopep-2 did not show any effect on tumor cells up to a 50 μM concentration. However, no clear correlation could be obtained between the number of the daunomycin and the cytostatic efficacy. Among the conjugates with only one Dau, compound3,in which Dau was attached to theN-terminus, showed the highest in vitro cytostatic effect on glioblastoma cells. When the Dau was conjugated to the Lys side chain in position 15 (compound2), the effect decreased significantly, and conjugate4was also not significantly better. In case of conjugates with two drug molecules, a similar tendency was observed. Compound7,with Dau at theN-terminus and on the Lys side chain in position 10, showed the highest activity. When the Lys side chains were used as conjugation sites, the formed compound (6) had moderate activity on U87 cells. Surprisingly, conjugate5,with Dau at theN-terminus and Lys side chain in position 15, showed the lowest efficacy on the U87 cells. Compound8,with three Dau, showed moderate activity. Thus, the effectiveness of Angiopep-2-daunomycin conjugates does not primarily depend on the number of drug molecules, but rather on their position within the molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
Dau=Aoa-TFFYGGSRGK(Dau=Aoa)RNNFK(Dau=Aoa)TEEY-OH [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [32] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 23.6 ± 6.3 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The blood-brain barrier (BBB) is a semipermeable system, and, therefore, most of the active substances are poorly transported through this barrier, resulting in decreased therapeutic effects. Angiopep-2 (TFFYGGSRGKRNNFKTEEY) is a peptide ligand of low-density lipoprotein receptor-related protein-1 (LRP1), which can cross the BBB via receptor-mediated transcytosis and simultaneously target glioblastomas. Angiopep-2 contains three amino groups that have previously been used to produce drug-peptide conjugates, although the role and importance of each position have not yet been investigated. Thus, we studied the number and position of drug molecules in Angiopep-2 based conjugates. Conjugates containing one, two, and three daunomycin molecules conjugated via oxime linkage in all possible variations were prepared. The in vitro cytostatic effect and cellular uptake of the conjugates were investigated on U87 human glioblastoma cells. Degradation studies in the presence of rat liver lysosomal homogenates were also performed in order for us to better understand the structure-activity relationship and to determine the smallest metabolites. Conjugates with the best cytostatic effects had a drug molecule at the N-terminus. We demonstrated that the increasing number of drug molecules does not necessarily increase the efficacy of the conjugates, and proved that modification of the different conjugation sites results in differing biological effectiveness.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effect of the synthesized peptide (1) and daunomycin-peptide conjugates (2-8) was investigated on U87 human glioblastoma cells. The cells were treated with the peptide and conjugates at different concentrations (0.05-50 uM) for 24 h, and after a washing step, the cells were incubated for another 48 h at 37 C. The cytostatic effect of the compounds was determined using the MTT test. The measured IC50values are shown in. The free Angiopep-2 did not show any effect on tumor cells up to a 50 μM concentration. However, no clear correlation could be obtained between the number of the daunomycin and the cytostatic efficacy. Among the conjugates with only one Dau, compound3,in which Dau was attached to theN-terminus, showed the highest in vitro cytostatic effect on glioblastoma cells. When the Dau was conjugated to the Lys side chain in position 15 (compound2), the effect decreased significantly, and conjugate4was also not significantly better. In case of conjugates with two drug molecules, a similar tendency was observed. Compound7,with Dau at theN-terminus and on the Lys side chain in position 10, showed the highest activity. When the Lys side chains were used as conjugation sites, the formed compound (6) had moderate activity on U87 cells. Surprisingly, conjugate5,with Dau at theN-terminus and Lys side chain in position 15, showed the lowest efficacy on the U87 cells. Compound8,with three Dau, showed moderate activity. Thus, the effectiveness of Angiopep-2-daunomycin conjugates does not primarily depend on the number of drug molecules, but rather on their position within the molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
H-TFFYGGSRGK(Dau=Aoa)RNNFKTEEY-OH [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [32] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 24.0 ± 5.9 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The blood-brain barrier (BBB) is a semipermeable system, and, therefore, most of the active substances are poorly transported through this barrier, resulting in decreased therapeutic effects. Angiopep-2 (TFFYGGSRGKRNNFKTEEY) is a peptide ligand of low-density lipoprotein receptor-related protein-1 (LRP1), which can cross the BBB via receptor-mediated transcytosis and simultaneously target glioblastomas. Angiopep-2 contains three amino groups that have previously been used to produce drug-peptide conjugates, although the role and importance of each position have not yet been investigated. Thus, we studied the number and position of drug molecules in Angiopep-2 based conjugates. Conjugates containing one, two, and three daunomycin molecules conjugated via oxime linkage in all possible variations were prepared. The in vitro cytostatic effect and cellular uptake of the conjugates were investigated on U87 human glioblastoma cells. Degradation studies in the presence of rat liver lysosomal homogenates were also performed in order for us to better understand the structure-activity relationship and to determine the smallest metabolites. Conjugates with the best cytostatic effects had a drug molecule at the N-terminus. We demonstrated that the increasing number of drug molecules does not necessarily increase the efficacy of the conjugates, and proved that modification of the different conjugation sites results in differing biological effectiveness.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effect of the synthesized peptide (1) and daunomycin-peptide conjugates (2-8) was investigated on U87 human glioblastoma cells. The cells were treated with the peptide and conjugates at different concentrations (0.05-50 uM) for 24 h, and after a washing step, the cells were incubated for another 48 h at 37 C. The cytostatic effect of the compounds was determined using the MTT test. The measured IC50values are shown in. The free Angiopep-2 did not show any effect on tumor cells up to a 50 μM concentration. However, no clear correlation could be obtained between the number of the daunomycin and the cytostatic efficacy. Among the conjugates with only one Dau, compound3,in which Dau was attached to theN-terminus, showed the highest in vitro cytostatic effect on glioblastoma cells. When the Dau was conjugated to the Lys side chain in position 15 (compound2), the effect decreased significantly, and conjugate4was also not significantly better. In case of conjugates with two drug molecules, a similar tendency was observed. Compound7,with Dau at theN-terminus and on the Lys side chain in position 10, showed the highest activity. When the Lys side chains were used as conjugation sites, the formed compound (6) had moderate activity on U87 cells. Surprisingly, conjugate5,with Dau at theN-terminus and Lys side chain in position 15, showed the lowest efficacy on the U87 cells. Compound8,with three Dau, showed moderate activity. Thus, the effectiveness of Angiopep-2-daunomycin conjugates does not primarily depend on the number of drug molecules, but rather on their position within the molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
H-TFFYGGSRGKRNNFK(Dau=Aoa)TEEY-OH [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [32] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 30.2 ± 6.4 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The blood-brain barrier (BBB) is a semipermeable system, and, therefore, most of the active substances are poorly transported through this barrier, resulting in decreased therapeutic effects. Angiopep-2 (TFFYGGSRGKRNNFKTEEY) is a peptide ligand of low-density lipoprotein receptor-related protein-1 (LRP1), which can cross the BBB via receptor-mediated transcytosis and simultaneously target glioblastomas. Angiopep-2 contains three amino groups that have previously been used to produce drug-peptide conjugates, although the role and importance of each position have not yet been investigated. Thus, we studied the number and position of drug molecules in Angiopep-2 based conjugates. Conjugates containing one, two, and three daunomycin molecules conjugated via oxime linkage in all possible variations were prepared. The in vitro cytostatic effect and cellular uptake of the conjugates were investigated on U87 human glioblastoma cells. Degradation studies in the presence of rat liver lysosomal homogenates were also performed in order for us to better understand the structure-activity relationship and to determine the smallest metabolites. Conjugates with the best cytostatic effects had a drug molecule at the N-terminus. We demonstrated that the increasing number of drug molecules does not necessarily increase the efficacy of the conjugates, and proved that modification of the different conjugation sites results in differing biological effectiveness.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effect of the synthesized peptide (1) and daunomycin-peptide conjugates (2-8) was investigated on U87 human glioblastoma cells. The cells were treated with the peptide and conjugates at different concentrations (0.05-50 uM) for 24 h, and after a washing step, the cells were incubated for another 48 h at 37 C. The cytostatic effect of the compounds was determined using the MTT test. The measured IC50values are shown in. The free Angiopep-2 did not show any effect on tumor cells up to a 50 μM concentration. However, no clear correlation could be obtained between the number of the daunomycin and the cytostatic efficacy. Among the conjugates with only one Dau, compound3,in which Dau was attached to theN-terminus, showed the highest in vitro cytostatic effect on glioblastoma cells. When the Dau was conjugated to the Lys side chain in position 15 (compound2), the effect decreased significantly, and conjugate4was also not significantly better. In case of conjugates with two drug molecules, a similar tendency was observed. Compound7,with Dau at theN-terminus and on the Lys side chain in position 10, showed the highest activity. When the Lys side chains were used as conjugation sites, the formed compound (6) had moderate activity on U87 cells. Surprisingly, conjugate5,with Dau at theN-terminus and Lys side chain in position 15, showed the lowest efficacy on the U87 cells. Compound8,with three Dau, showed moderate activity. Thus, the effectiveness of Angiopep-2-daunomycin conjugates does not primarily depend on the number of drug molecules, but rather on their position within the molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
Dau=Aoa-TFFYGGSRGKRNNFK(Dau=Aoa)TEEY-OH [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [32] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 32.3 ± 8.1 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The blood-brain barrier (BBB) is a semipermeable system, and, therefore, most of the active substances are poorly transported through this barrier, resulting in decreased therapeutic effects. Angiopep-2 (TFFYGGSRGKRNNFKTEEY) is a peptide ligand of low-density lipoprotein receptor-related protein-1 (LRP1), which can cross the BBB via receptor-mediated transcytosis and simultaneously target glioblastomas. Angiopep-2 contains three amino groups that have previously been used to produce drug-peptide conjugates, although the role and importance of each position have not yet been investigated. Thus, we studied the number and position of drug molecules in Angiopep-2 based conjugates. Conjugates containing one, two, and three daunomycin molecules conjugated via oxime linkage in all possible variations were prepared. The in vitro cytostatic effect and cellular uptake of the conjugates were investigated on U87 human glioblastoma cells. Degradation studies in the presence of rat liver lysosomal homogenates were also performed in order for us to better understand the structure-activity relationship and to determine the smallest metabolites. Conjugates with the best cytostatic effects had a drug molecule at the N-terminus. We demonstrated that the increasing number of drug molecules does not necessarily increase the efficacy of the conjugates, and proved that modification of the different conjugation sites results in differing biological effectiveness.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effect of the synthesized peptide (1) and daunomycin-peptide conjugates (2-8) was investigated on U87 human glioblastoma cells. The cells were treated with the peptide and conjugates at different concentrations (0.05-50 uM) for 24 h, and after a washing step, the cells were incubated for another 48 h at 37 C. The cytostatic effect of the compounds was determined using the MTT test. The measured IC50values are shown in. The free Angiopep-2 did not show any effect on tumor cells up to a 50 μM concentration. However, no clear correlation could be obtained between the number of the daunomycin and the cytostatic efficacy. Among the conjugates with only one Dau, compound3,in which Dau was attached to theN-terminus, showed the highest in vitro cytostatic effect on glioblastoma cells. When the Dau was conjugated to the Lys side chain in position 15 (compound2), the effect decreased significantly, and conjugate4was also not significantly better. In case of conjugates with two drug molecules, a similar tendency was observed. Compound7,with Dau at theN-terminus and on the Lys side chain in position 10, showed the highest activity. When the Lys side chains were used as conjugation sites, the formed compound (6) had moderate activity on U87 cells. Surprisingly, conjugate5,with Dau at theN-terminus and Lys side chain in position 15, showed the lowest efficacy on the U87 cells. Compound8,with three Dau, showed moderate activity. Thus, the effectiveness of Angiopep-2-daunomycin conjugates does not primarily depend on the number of drug molecules, but rather on their position within the molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
GnRH-III-[2ΔHis-3D-Tic,8Lys(glutaryl-Val-Cit-PABC-diamine-PTX)] conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 36.29 ± 3.17 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Normal human pituitary cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 42.67 ± 7.04 µM | |||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
Human pituitary and human prostate cancer tissues have been used to evaluate the binding affinities of the new GnRH-III-drug conjugates to GnRH-R. Therefore, increasing compound concentrations were applied and the displacement of radiolabeled [125I]-triptorelin from GnRH-Rs was detected. The obtained results were compared with the binding affinities of the oxime bond-linked GnRH-III-Dau conjugates (I, II). All compounds bind to the receptors with high affinities in the low nanomolar range, while GnRH unrelated peptides such as somatostatin or bombesin were not able to displace the radio-labelled triptorelin. However, in comparison to the GnRH-III-homing peptide (I), the self-immolative linker conjugate exhibited a 3- to 10-times reduced affinity to the GnRH receptors. Interestingly, most of the PTX-containing cleavable compounds possessed a slightly higher binding affinity than the corresponding Dau-equivalent, even if the targeting sequence and the cathepsin cleavage site remained the same.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate cancer | Human prostate cancer cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 48.14 ± 0.47 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Evaluation Method | GraphPad prism assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
Drug delivery systems (DDS) are promising tools for targeted tumor therapy providing the selective delivery of cytotoxic drugs to malignant cells, while side-effects and systemic toxicity are reduced. In addition to monoclonal antibodies (mAb), peptide ligands with a high affinity for tumor-specific cell surface compartments (e.g., receptors) can be used as carriers for cytotoxic payloads, as they provide beneficial features such as good tissue penetration, low immunogenicity and structural simplicity which enables their cost-efficient production by chemical synthesi. Receptors for the human gonadotropin releasing hormone (GnRH-I, <EHWSYGLRPG-NH2, <E is pyroglutamic acid) were not only identified in the pituitary, but also in various reproductive system-related cancers, such as breast, prostate and ovarian cancers, as well as non-reproductive cancers, such as colon and lung cancer. Thus, GnRH-related peptides are promising homing devices to deliver cytotoxic drugs selectively to cancer cells. A natural isoform of GnRH-I is the sea lamprey analog GnRH-III. This weak GnRH agonist binds to GnRH receptors (GnRH-R) on cancer cells and induces, like GnRH-I, a direct antitumor activity on several cancer cell lines, but its gonadotropin releasing activity is 500-1000 times lower in vitro and in vivo. Due to the direct anticancer activity and the low endocrine effect, GnRH-III and its derivatives have been successfully used as homing devices in in vitro and in vivo experiments. Encouraged by these promising findings, we report on the synthesis and biochemical characterization of eight cleavable self-immolative linker containing GnRH-III-drug conjugates. Of particular interest was the comparison of (1) two GnRH-III targeting moieties (GnRH-III-[4Lys(Bu)] (I) and GnRH-III-[2His,3D-Tic,4Lys(Bu)] (II)), (2) two cathepsin B-cleavable dipeptidyl-PABC linkers (Val-Ala and Val-Cit) and (3) two traditional anticancer drugs with different modes of action (Dau and PTX). For a better comparison and to demonstrate the proof of concept, four corresponding non-cleavable GnRH-III-Dau and -PTX conjugates have been developed and analyzed. The 8Lys of the targeting peptide was used as the ligation site. In the case of the Dau conjugates, the amino group of the daunosamine sugar has been used for attachment to the linker, while in the case of PTX, the C2-OH group was exploited for this purpose. All synthesized GnRH-III-Dau and -PTX conjugates were studied for their anticancer activity on A2780 ovarian and Panc-1 pancreatic cancer cells. Furthermore, the release of the drug by lysosomal enzymes and the GnRH-R binding affinities of the SMDC were examined.
Click to Show/Hide
|
||||
| Description |
To investigate the anticancer activity of the GnRH-III drug conjugates, cell viability studies have been performed on A2780 ovarian cancer and Panc-1 pancreatic cancer cells. The GnRH-R expression of these cell lines was determined by Western blot studies. In the case of the A2780 cells, a distinct band at 38 kDa could be detected which corresponds to the full-length human GnRH-R. In contrast, the signal intensity of the 38 kDa band was much lower for Panc-1 pancreatic cancer cells being in line with our previous results. Thus, the antiproliferative activity of the GnRH-drug conjugates was studied on high-GnRH-R-expressing A2780 cells and low-GnRH-R-expressing Panc-1 cells. Since the release of free Dau and PTX can be assumed, both drugs were used as controls. The cells were treated for either 24 h (Dau conjugates) or six hours (PTX compounds), followed by additional incubation with fresh growth medium until 72 h after treatment initiation. The obtained results reveal, on the one hand, that the non-cleavable linker-containing conjugates possess a reduced anticancer activity in comparison to the cleavable conjugates and, on the other hand, that the activity of the all GnRH-III-drug conjugates was substantially reduced compared to the free drug. Moreover, all compounds displayed a lower biological activity on Panc-1 cells than on A2780. In the case of the cleavable GnRH-III-Dau conjugates, the IC50 values varied between 2.85-11.18 μM on A2780 cells, whereby the best activity was obtained for compound 13 (2.85 μM) which contained the cathepsin B-cleavage site Val-Ala and the GnRH-III-[2His-3D-Tic-4Lys(Bu)] peptide carrier. Apart from that, the IC50 values of the cleavable PTX conjugates on A2780 cells are in the same sub-micromolar range and vary between 0.51-0.77 μM, while the activity of these conjugates was approximately 10 times lower on Panc-1 cells (5.03-8.15 μM).
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | ||
H-TFFYGGSRGK(Dau=Aoa-VA)RNNFKTEEY-OH [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [32] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 50 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The blood-brain barrier (BBB) is a semipermeable system, and, therefore, most of the active substances are poorly transported through this barrier, resulting in decreased therapeutic effects. Angiopep-2 (TFFYGGSRGKRNNFKTEEY) is a peptide ligand of low-density lipoprotein receptor-related protein-1 (LRP1), which can cross the BBB via receptor-mediated transcytosis and simultaneously target glioblastomas. Angiopep-2 contains three amino groups that have previously been used to produce drug-peptide conjugates, although the role and importance of each position have not yet been investigated. Thus, we studied the number and position of drug molecules in Angiopep-2 based conjugates. Conjugates containing one, two, and three daunomycin molecules conjugated via oxime linkage in all possible variations were prepared. The in vitro cytostatic effect and cellular uptake of the conjugates were investigated on U87 human glioblastoma cells. Degradation studies in the presence of rat liver lysosomal homogenates were also performed in order for us to better understand the structure-activity relationship and to determine the smallest metabolites. Conjugates with the best cytostatic effects had a drug molecule at the N-terminus. We demonstrated that the increasing number of drug molecules does not necessarily increase the efficacy of the conjugates, and proved that modification of the different conjugation sites results in differing biological effectiveness.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effect and the in vitro cellular uptake of the spacer-containing daunomycin-peptide conjugates (9-11) were studied on U87 human glioblastoma cells as previously described. The measured IC50 values are shown in Table 2, while the measured cellular uptake at 10 μM and 50 μM concentration are given in Figure 4. The fluorescence intensity values showed good correlation with the percentage of daunomycin-positive cells in the case of these conjugates as well. Conjugate 9 showed slight toxicity during the measurements at a concentration of 50 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
DauQ=AoaGFLGD4HbPG [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [30] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 450 µM | |||
| Evaluation Method | xCELLigence SP system assay | ||||
| Administration Time | 48 h | ||||
| MOA of PDC |
As observed, the cathepsin B labile spacer (GFLG), which was incorporated between the targeting peptide and the daunomycin to ensure the efficient release of the active metabolite, highly increases the hydrophobicity of the conjugates. As demonstrated, the solubility problem of these conjugates can be solved by hydrophilic polymer coupling, not only by using the well-known PEG but also by using the amino-monofunctional HbPG. To the best of our knowledge, 1:1 covalent peptide-polymer conjugates with well-defined monofunctional HbPG have been reported here for the first time. The results of the in vitro cell viability and cellular uptake measurements on HT-29 human colon adenocarcinoma cells prove that the HbPG and the PEG highly influenced the biological activity of the drug-peptide-polymer conjugates. In both peptide conjugate series, one GE11 and one D4 targeting peptide-based conjugate were found with outstanding cellular uptake and cytotoxicity values, namely Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG. According to our results, the PEG is suitable for longer targeting peptides (e.g. GE11), but the G5 spacer is not suitable irrespective of the length of the peptide because it may decrease the biological effect by increasing the flexibility of the polymer and shading of the targeting moiety. In contrast, the use of the hydrophilic hyperbranched polyglycerol (HbPG) is advantageous for short targeting peptides (e.g. D4) but only with a G5 spacer, which provides accessibility of the peptide for receptor binding and cellular uptake resulting in outstanding cytotoxicity.
Click to Show/Hide
|
||||
| Description |
The polymers influenced the cellular uptake of the conjugates in a different manner, but in both groups (GE11 and D4 containing conjugates), one of the compounds was outstanding. The HT-29 cells could uptake Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG to the highest extent. The G5 spacer increased the uptake of the D4-HbPG derivative, presumably because the increased distance between the globular HbPG and the very short peptide sequence provides a better receptor binding. In sharp contrast, the G5 spacer decreases the uptake of the conjugates in all other cases. The most significant difference was observed in the case of GE11-PEG, where the G5 spacer completely demolished the internalization. Probably, here the G5 spacer provides more flexibility for the linear PEG chain resulting in decreased receptor binding. There was one outstanding conjugate from each group (Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG) in the cytotoxicity measurements that correlated well with the results of the internalization studies. These conjugates were found to be the most potent ones in the viability measurement and were proved to be taken up by HT-29 cells the most effectively. Depending on the type of polymer, the incorporation of the G5 spacer had an opposite effect on the cytotoxic activity of the conjugates. In the presence of the G5 spacer, the antitumour activity of the PEGylated conjugates decreased, while the cytotoxicity of the HbPG-containing conjugates increased, especially in the case of those with the D4 targeting peptide. We observed that some of the conjugates (Dau[double bond, length as m-dash]Aoa-GFLG-D4-HbPG and Dau[double bond, length as m-dash]Aoa-GFLG-GE11-G5-PEG) could not cause complete cell death, i.e., ˜0% viability value - characteristic for cell-free culturing medium - was not achieved even at the highest concentration, since their dose-response curves reached a plateau in a lower concentration range. In our opinion, this can be explained by the different characteristics of the highly hydrophobic peptide chain and the highly hydrophilic polymer segment. Due to this amphiphilic character, self-aggregation of the conjugates may occur, which then may block the accessibility of the targeting peptide for receptor binding, thereby decreasing the efficiency of the conjugate as well. This assumption is also confirmed by the turbidity results, since the observed low turbidity values may be caused by the possible formation of nanosized aggregates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
Dau=Aoa-GFLG-c[DKP-RGD] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 2.5 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 5.4 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 3 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 14.7 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 49.4 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | > 150 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 6 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | > 150 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
c[DKP-RGD]-PEG4-sC18(dau=Aoa-GFLG-Lys8) [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 2.7 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 3.9 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 7.8 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 11 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 5 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 23.8 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 89.1 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
c[DKP-RGD]-PEG4-sC18(dau=Aoa-Lys8) [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 3 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 2 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 5.6 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 9.1 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 12.5 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds 1b and 3b demonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure 6). Importantly, 1b was more active than 3b demonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that 2b was less efficient than 1b but marginally more active than 3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that 3c is still significantly more active in U87 cells expressing integrin receptors. By contrast, 1c kept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to 1b.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 50.5 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds 1b and 3b demonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure 6). Importantly, 1b was more active than 3b demonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that 2b was less efficient than 1b but marginally more active than 3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that 3c is still significantly more active in U87 cells expressing integrin receptors. By contrast, 1c kept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to 1b.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 6 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 53.4 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds 1b and 3b demonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure 6). Importantly, 1b was more active than 3b demonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that 2b was less efficient than 1b but marginally more active than 3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that 3c is still significantly more active in U87 cells expressing integrin receptors. By contrast, 1c kept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to 1b.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
c[DKP-RGD]-PEG4-Aoa=dau [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 3 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 9.2 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 3 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 21.7 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 56.9 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | > 150 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 6 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | > 150 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
sC18(dau-Aoa-Lys8) [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 5 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 5.8 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 9.2 μM | |||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 4 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 25.1 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to3b.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | 42.2 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [33] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | > 100 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to2b.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
[C15]-NPY-Dauno-HYD [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [34] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability | 35.00% | |||
| Evaluation Method | XTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 1 μM | ||||
| MOA of PDC |
In this study, we investigate the usefulness of peptides as carriers for which the receptors are overexpressed on tumor cells. Covalent linking of the drug to the peptide could be used for chemotherapy and would lead to selective addressing of the tumor cells. As a model peptide, we used neuropeptide Y (NPY) because its receptors are produced in a number of neuroblastoma and the thereof derived cell lines. NPY is a 36-amino acid peptide of the pancreatic polypeptide family. It is expressed in the peripheral and central nervous systems and is one of the most abundant neuropeptides in the brain. Five distinct NPY receptors have been cloned, which have been named Y1, Y2, Y4, Y5, and y6 receptor subtypes. Upon binding to the G-protein coupled receptors, the ligand-receptor complex is internalized, which provides a convenient way to enter the cell by receptor-mediated endocytosis. Because structure-activity relationships (SARs) of NPY are well-known, position 15 of NPY was used for attaching maleimide anthracycline derivatives which would presumably not lead to a significant loss of binding activity for the Y1 receptor. Because the NPY-receptor complex is internalized and undergoes a pH shift from 7.4 to approximately 5.0 during endosomal trafficking, two different anthracycline derivatives that differ in the acid sensitivity of the bond between the drug and the spacer were selected (see Figure 1). Doxo-MBS and Dauno-MBS are characterized by a stable amide bond at the 3-amino position of the anthracycline; Dauno-HYD is a daunorubicin derivative incorporating an acid-sensitive hydrazone linker at the 13-keto position. The maleimide moiety was introduced into daunorubicin and doxorubicin in order to selectively label the sulfhydryl group of [C15]-NPY.
Click to Show/Hide
|
||||
| Description |
[C15]-NPY, [C15]-NPY-Dauno-MBS, and [C15]-NPY-Doxo-MBS showed no or only marginal effects. In contrast, [C15]-NPY-Dauno-HYD was as effective as free daunorubicin with respect to cytotoxicity, and the compounds were able to reduce cell growth by 66.9 ± 2.5% and 68.6 ± 0.4%, respectively.
|
||||
| In Vitro Model | Askin tumor | SK-N-MC cell | CVCL_0530 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [34] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability | 60.00% | |||
| Administration Time | 36 h | ||||
| Administration Dosage | 1 μM with BIBP 3226 (100 μM) | ||||
| MOA of PDC |
In this study, we investigate the usefulness of peptides as carriers for which the receptors are overexpressed on tumor cells. Covalent linking of the drug to the peptide could be used for chemotherapy and would lead to selective addressing of the tumor cells. As a model peptide, we used neuropeptide Y (NPY) because its receptors are produced in a number of neuroblastoma and the thereof derived cell lines. NPY is a 36-amino acid peptide of the pancreatic polypeptide family. It is expressed in the peripheral and central nervous systems and is one of the most abundant neuropeptides in the brain. Five distinct NPY receptors have been cloned, which have been named Y1, Y2, Y4, Y5, and y6 receptor subtypes. Upon binding to the G-protein coupled receptors, the ligand-receptor complex is internalized, which provides a convenient way to enter the cell by receptor-mediated endocytosis. Because structure-activity relationships (SARs) of NPY are well-known, position 15 of NPY was used for attaching maleimide anthracycline derivatives which would presumably not lead to a significant loss of binding activity for the Y1 receptor. Because the NPY-receptor complex is internalized and undergoes a pH shift from 7.4 to approximately 5.0 during endosomal trafficking, two different anthracycline derivatives that differ in the acid sensitivity of the bond between the drug and the spacer were selected (see Figure 1). Doxo-MBS and Dauno-MBS are characterized by a stable amide bond at the 3-amino position of the anthracycline; Dauno-HYD is a daunorubicin derivative incorporating an acid-sensitive hydrazone linker at the 13-keto position. The maleimide moiety was introduced into daunorubicin and doxorubicin in order to selectively label the sulfhydryl group of [C15]-NPY.
Click to Show/Hide
|
||||
| Description |
Pretreatment with 100 μM of the Y1 receptor antagonist BIBP 322629,30 antagonized the cytotoxicity of [C15]-NPY-Dauno-HYD (cell viability remained at 89.1 ± 4.6%), but not that of daunorubicin. To investigate the minimal required concentration of the administered conjugates, dilutions were made and tested on SK-N-MC cells; the cytotoxic effects of [C15]-NPY-Dauno-HYD and daunorubicin were visible starting at 1 μM concentrations.
Click to Show/Hide
|
||||
| In Vitro Model | Askin tumor | SK-N-MC cell | CVCL_0530 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [34] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability | 100.00% | |||
| Administration Time | 48 h | ||||
| Administration Dosage | 1 μM | ||||
| MOA of PDC |
In this study, we investigate the usefulness of peptides as carriers for which the receptors are overexpressed on tumor cells. Covalent linking of the drug to the peptide could be used for chemotherapy and would lead to selective addressing of the tumor cells. As a model peptide, we used neuropeptide Y (NPY) because its receptors are produced in a number of neuroblastoma and the thereof derived cell lines. NPY is a 36-amino acid peptide of the pancreatic polypeptide family. It is expressed in the peripheral and central nervous systems and is one of the most abundant neuropeptides in the brain. Five distinct NPY receptors have been cloned, which have been named Y1, Y2, Y4, Y5, and y6 receptor subtypes. Upon binding to the G-protein coupled receptors, the ligand-receptor complex is internalized, which provides a convenient way to enter the cell by receptor-mediated endocytosis. Because structure-activity relationships (SARs) of NPY are well-known, position 15 of NPY was used for attaching maleimide anthracycline derivatives which would presumably not lead to a significant loss of binding activity for the Y1 receptor. Because the NPY-receptor complex is internalized and undergoes a pH shift from 7.4 to approximately 5.0 during endosomal trafficking, two different anthracycline derivatives that differ in the acid sensitivity of the bond between the drug and the spacer were selected (see Figure 1). Doxo-MBS and Dauno-MBS are characterized by a stable amide bond at the 3-amino position of the anthracycline; Dauno-HYD is a daunorubicin derivative incorporating an acid-sensitive hydrazone linker at the 13-keto position. The maleimide moiety was introduced into daunorubicin and doxorubicin in order to selectively label the sulfhydryl group of [C15]-NPY.
Click to Show/Hide
|
||||
| Description |
The same compounds were tested on glioblastoma XF-498L cells that do not express NPY receptors.31 Daunorubicin was able to reduce cell growth by 47.3 ± 5%, whereas [C15]-NPY-Dauno-HYD showed no cytotoxicity (Figure 3C).
|
||||
| In Vitro Model | Glioblastoma | XF 498 cell | CVCL_8928 | ||
[C15]-NPY-Dauno-MBS [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [34] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability | 100.00% | |||
| Evaluation Method | XTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 1 μM | ||||
| MOA of PDC |
In this study, we investigate the usefulness of peptides as carriers for which the receptors are overexpressed on tumor cells. Covalent linking of the drug to the peptide could be used for chemotherapy and would lead to selective addressing of the tumor cells. As a model peptide, we used neuropeptide Y (NPY) because its receptors are produced in a number of neuroblastoma and the thereof derived cell lines. NPY is a 36-amino acid peptide of the pancreatic polypeptide family. It is expressed in the peripheral and central nervous systems and is one of the most abundant neuropeptides in the brain. Five distinct NPY receptors have been cloned, which have been named Y1, Y2, Y4, Y5, and y6 receptor subtypes. Upon binding to the G-protein coupled receptors, the ligand-receptor complex is internalized, which provides a convenient way to enter the cell by receptor-mediated endocytosis. Because structure-activity relationships (SARs) of NPY are well-known, position 15 of NPY was used for attaching maleimide anthracycline derivatives which would presumably not lead to a significant loss of binding activity for the Y1 receptor. Because the NPY-receptor complex is internalized and undergoes a pH shift from 7.4 to approximately 5.0 during endosomal trafficking, two different anthracycline derivatives that differ in the acid sensitivity of the bond between the drug and the spacer were selected (see Figure 1). Doxo-MBS and Dauno-MBS are characterized by a stable amide bond at the 3-amino position of the anthracycline; Dauno-HYD is a daunorubicin derivative incorporating an acid-sensitive hydrazone linker at the 13-keto position. The maleimide moiety was introduced into daunorubicin and doxorubicin in order to selectively label the sulfhydryl group of [C15]-NPY.
Click to Show/Hide
|
||||
| Description |
[C15]-NPY, [C15]-NPY-Dauno-MBS, and [C15]-NPY-Doxo-MBS showed no or only marginal effects. In contrast, [C15]-NPY-Dauno-HYD was as effective as free daunorubicin with respect to cytotoxicity, and the compounds were able to reduce cell growth by 66.9 ± 2.5% and 68.6 ± 0.4%, respectively.
|
||||
| In Vitro Model | Askin tumor | SK-N-MC cell | CVCL_0530 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [34] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability | 100.00% | |||
| Administration Time | 36 h | ||||
| Administration Dosage | 1 μM with BIBP 3226 (100 μM) | ||||
| MOA of PDC |
In this study, we investigate the usefulness of peptides as carriers for which the receptors are overexpressed on tumor cells. Covalent linking of the drug to the peptide could be used for chemotherapy and would lead to selective addressing of the tumor cells. As a model peptide, we used neuropeptide Y (NPY) because its receptors are produced in a number of neuroblastoma and the thereof derived cell lines. NPY is a 36-amino acid peptide of the pancreatic polypeptide family. It is expressed in the peripheral and central nervous systems and is one of the most abundant neuropeptides in the brain. Five distinct NPY receptors have been cloned, which have been named Y1, Y2, Y4, Y5, and y6 receptor subtypes. Upon binding to the G-protein coupled receptors, the ligand-receptor complex is internalized, which provides a convenient way to enter the cell by receptor-mediated endocytosis. Because structure-activity relationships (SARs) of NPY are well-known, position 15 of NPY was used for attaching maleimide anthracycline derivatives which would presumably not lead to a significant loss of binding activity for the Y1 receptor. Because the NPY-receptor complex is internalized and undergoes a pH shift from 7.4 to approximately 5.0 during endosomal trafficking, two different anthracycline derivatives that differ in the acid sensitivity of the bond between the drug and the spacer were selected (see Figure 1). Doxo-MBS and Dauno-MBS are characterized by a stable amide bond at the 3-amino position of the anthracycline; Dauno-HYD is a daunorubicin derivative incorporating an acid-sensitive hydrazone linker at the 13-keto position. The maleimide moiety was introduced into daunorubicin and doxorubicin in order to selectively label the sulfhydryl group of [C15]-NPY.
Click to Show/Hide
|
||||
| Description |
Pretreatment with 100 μM of the Y1 receptor antagonist BIBP 322629,30 antagonized the cytotoxicity of [C15]-NPY-Dauno-HYD (cell viability remained at 89.1 ± 4.6%), but not that of daunorubicin. To investigate the minimal required concentration of the administered conjugates, dilutions were made and tested on SK-N-MC cells; the cytotoxic effects of [C15]-NPY-Dauno-HYD and daunorubicin were visible starting at 1 μM concentrations.
Click to Show/Hide
|
||||
| In Vitro Model | Askin tumor | SK-N-MC cell | CVCL_0530 | ||
References
