Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00218
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
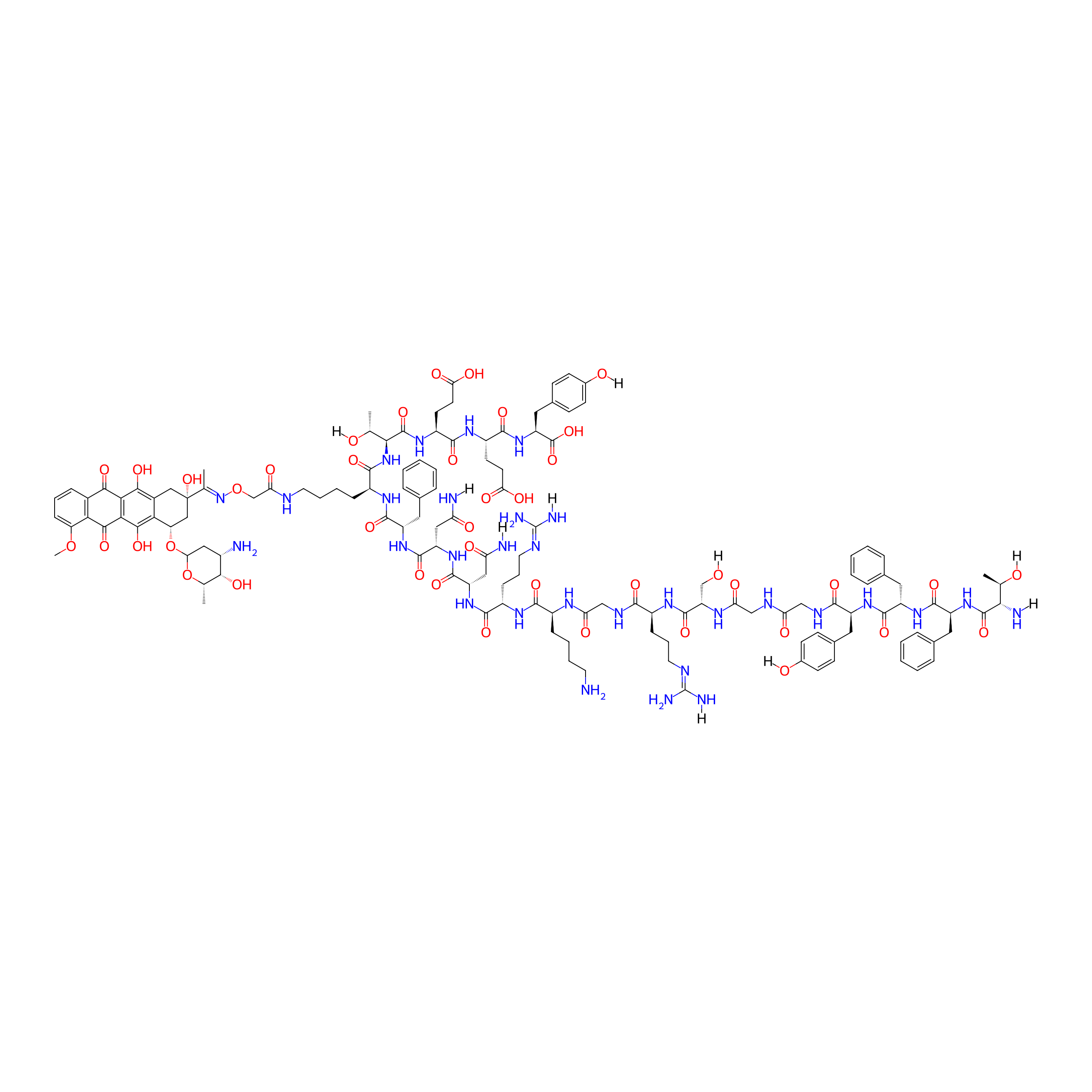
H-TFFYGGSRGKRNNFK(Dau=Aoa)TEEY-OH
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
|
|||||
| Peptide Name |
Angiopep-2
|
Peptide Info | ||||
| Receptor Name |
Prolow-density lipoprotein receptor-related protein 1; Low-density lipoprotein receptor-related protein 2 (LRP1; LRP2)
|
Receptor Info | ||||
| Drug Name |
Daunorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
Aminooxyacetic acid
|
Linker Info | ||||
| Formula |
C133H179N31O42
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 2884.07 | ||||
| Lipid-water partition coefficient (xlogp) | -10.7534 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 40 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 45 | |||||
| Rotatable Bond Count (rotbonds) | 90 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
30.2 ± 6.4 µM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The blood-brain barrier (BBB) is a semipermeable system, and, therefore, most of the active substances are poorly transported through this barrier, resulting in decreased therapeutic effects. Angiopep-2 (TFFYGGSRGKRNNFKTEEY) is a peptide ligand of low-density lipoprotein receptor-related protein-1 (LRP1), which can cross the BBB via receptor-mediated transcytosis and simultaneously target glioblastomas. Angiopep-2 contains three amino groups that have previously been used to produce drug-peptide conjugates, although the role and importance of each position have not yet been investigated. Thus, we studied the number and position of drug molecules in Angiopep-2 based conjugates. Conjugates containing one, two, and three daunomycin molecules conjugated via oxime linkage in all possible variations were prepared. The in vitro cytostatic effect and cellular uptake of the conjugates were investigated on U87 human glioblastoma cells. Degradation studies in the presence of rat liver lysosomal homogenates were also performed in order for us to better understand the structure-activity relationship and to determine the smallest metabolites. Conjugates with the best cytostatic effects had a drug molecule at the N-terminus. We demonstrated that the increasing number of drug molecules does not necessarily increase the efficacy of the conjugates, and proved that modification of the different conjugation sites results in differing biological effectiveness.
Click to Show/Hide
|
||||
| Description |
The in vitro cytostatic effect of the synthesized peptide (1) and daunomycin-peptide conjugates (2-8) was investigated on U87 human glioblastoma cells. The cells were treated with the peptide and conjugates at different concentrations (0.05-50 uM) for 24 h, and after a washing step, the cells were incubated for another 48 h at 37 C. The cytostatic effect of the compounds was determined using the MTT test. The measured IC50values are shown in. The free Angiopep-2 did not show any effect on tumor cells up to a 50 μM concentration. However, no clear correlation could be obtained between the number of the daunomycin and the cytostatic efficacy. Among the conjugates with only one Dau, compound3,in which Dau was attached to theN-terminus, showed the highest in vitro cytostatic effect on glioblastoma cells. When the Dau was conjugated to the Lys side chain in position 15 (compound2), the effect decreased significantly, and conjugate4was also not significantly better. In case of conjugates with two drug molecules, a similar tendency was observed. Compound7,with Dau at theN-terminus and on the Lys side chain in position 10, showed the highest activity. When the Lys side chains were used as conjugation sites, the formed compound (6) had moderate activity on U87 cells. Surprisingly, conjugate5,with Dau at theN-terminus and Lys side chain in position 15, showed the lowest efficacy on the U87 cells. Compound8,with three Dau, showed moderate activity. Thus, the effectiveness of Angiopep-2-daunomycin conjugates does not primarily depend on the number of drug molecules, but rather on their position within the molecule.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
References
