Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00198
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
GnRH-III-[3D-Tic, 7D-Trp 8Lys(Dau=Aoa)]
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
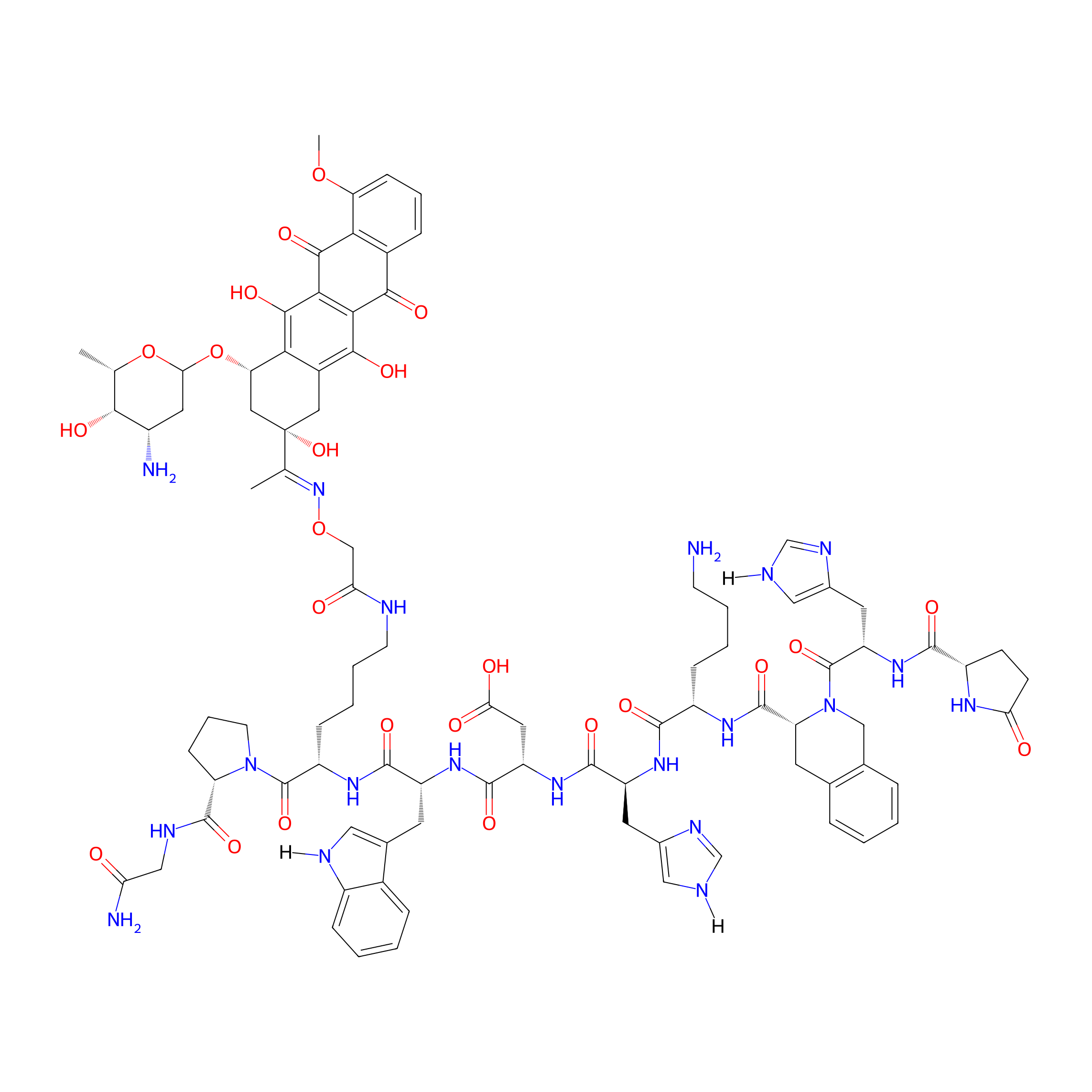
|
|||||
| Peptide Name |
GnRH-III
|
Peptide Info | ||||
| Receptor Name |
Gonadotropin-releasing hormone receptor (GNRHR)
|
Receptor Info | ||||
| Drug Name |
Daunorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
Aminooxyacetic acid
|
Linker Info | ||||
| Peptide Modified Type |
Amino acid modifications
|
|||||
| Modified Segment |
Incorporation of unnatural amino acids: pyroglutamic acid (
|
|||||
| Ternimal Modification |
N-terminal modification
|
|||||
| Formula |
C90H110N20O24
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 1855.986 | ||||
| Lipid-water partition coefficient (xlogp) | -1.8884 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 20 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 28 | |||||
| Rotatable Bond Count (rotbonds) | 41 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
3.42 ± 0.39 µM
|
|||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
15.34 ± 2.63 µM
|
|||
| Description |
We obtained IC50 values in the low micromolar range on MCF-7 cells varying between 0.14 and 6.64 μM. In the case of HT-29 colon cancer cells, the determined IC50 values were slightly higher and within a range of 3.31-19.10 μM. With exception of compound 10, no significant difference of the cancer cell growth inhibitory effect of the novel bioconjugates and the control conjugates (K1 and K2) could be detected. However, the replacement of 3Trp by 3d-Tic in connection with the deletion of 2His led to an increased cytostatic effect of 10 on both of the analyzed cell lines. The IC50 value of bioconjugate 10 was more than 15-times lower on ER+ breast cancer MCF-7 cells and 5-times lower on colon cancer cells HT-29 compared to the control compound K2. Based on these promising findings, the growth inhibitory effects of conjugate 10 and the related 2His-3d-Tic (3, 5 and 12) conjugates, as well as the 10Gly-NH-Et (7 and 14) containing compounds, were studied on estrogen receptor negative (ER-) MDA-MB-231 breast cancer cells. The corresponding IC50 values are shown in Table 2. The GnRH-III-Dau conjugate 10 revealed also on this cell line the highest anticancer activity with an IC50 value of 2.49 μM. The comparison of the dose-dependent growth inhibitory effect of 10 and K2 on the three cancer cell lines is shown in Figure 1. Considering the results of all three cell lines, only compound 10 which contains the N-terminal modification 2His-d-Tic-Lys(Bu), displayed clearly a reduced cell viability, while the conjugates bearing other substitutions yielded IC50 values which vary only slightly in comparison to the controls.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
References
