Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00122
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
DauQ=AoaGFLGGE11HbPG
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
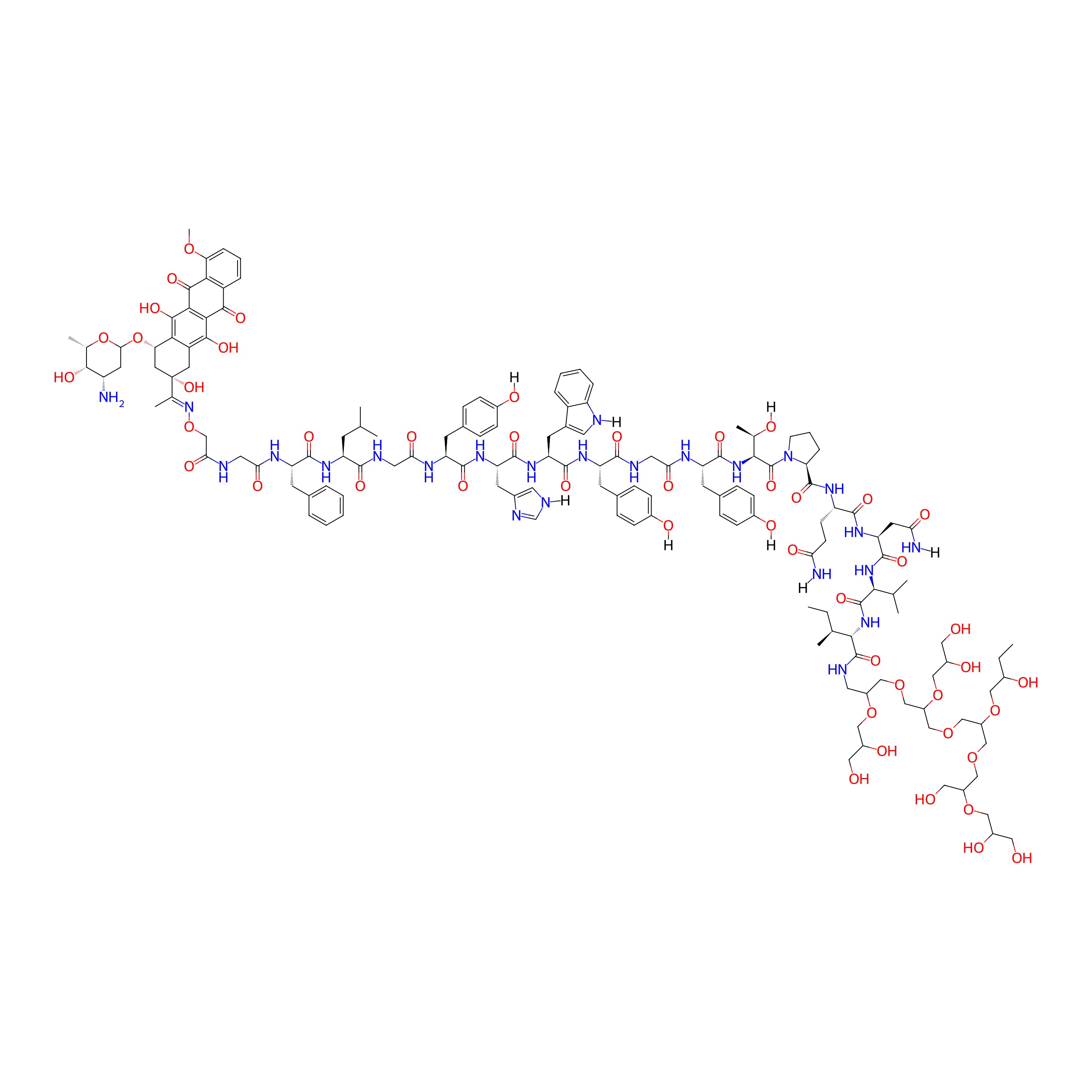
|
|||||
| Peptide Name |
GE11
|
Peptide Info | ||||
| Receptor Name |
Pro-epidermal growth factor (EGF)
|
Receptor Info | ||||
| Drug Name |
Daunorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
GFLG
|
Linker Info | ||||
| Peptide Modified Type |
The modification of binding with chemical macromolecules
|
|||||
| Modified Segment |
HbPG
|
|||||
| Formula |
C148H204N24O48
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 3087.38 | ||||
| Lipid-water partition coefficient (xlogp) | -6.4335 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 37 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 51 | |||||
| Rotatable Bond Count (rotbonds) | 96 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
9.3 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | xCELLigence SP system assay | ||||
| MOA of PDC |
As observed, the cathepsin B labile spacer (GFLG), which was incorporated between the targeting peptide and the daunomycin to ensure the efficient release of the active metabolite, highly increases the hydrophobicity of the conjugates. As demonstrated, the solubility problem of these conjugates can be solved by hydrophilic polymer coupling, not only by using the well-known PEG but also by using the amino-monofunctional HbPG. To the best of our knowledge, 1:1 covalent peptide-polymer conjugates with well-defined monofunctional HbPG have been reported here for the first time. The results of the in vitro cell viability and cellular uptake measurements on HT-29 human colon adenocarcinoma cells prove that the HbPG and the PEG highly influenced the biological activity of the drug-peptide-polymer conjugates. In both peptide conjugate series, one GE11 and one D4 targeting peptide-based conjugate were found with outstanding cellular uptake and cytotoxicity values, namely Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG. According to our results, the PEG is suitable for longer targeting peptides (e.g. GE11), but the G5 spacer is not suitable irrespective of the length of the peptide because it may decrease the biological effect by increasing the flexibility of the polymer and shading of the targeting moiety. In contrast, the use of the hydrophilic hyperbranched polyglycerol (HbPG) is advantageous for short targeting peptides (e.g. D4) but only with a G5 spacer, which provides accessibility of the peptide for receptor binding and cellular uptake resulting in outstanding cytotoxicity.
Click to Show/Hide
|
||||
| Description |
The polymers influenced the cellular uptake of the conjugates in a different manner, but in both groups (GE11 and D4 containing conjugates), one of the compounds was outstanding. The HT-29 cells could uptake Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG to the highest extent. The G5 spacer increased the uptake of the D4-HbPG derivative, presumably because the increased distance between the globular HbPG and the very short peptide sequence provides a better receptor binding. In sharp contrast, the G5 spacer decreases the uptake of the conjugates in all other cases. The most significant difference was observed in the case of GE11-PEG, where the G5 spacer completely demolished the internalization. Probably, here the G5 spacer provides more flexibility for the linear PEG chain resulting in decreased receptor binding. There was one outstanding conjugate from each group (Dau[double bond, length as m-dash]Aoa-GFLG-GE11-PEG and Dau[double bond, length as m-dash]Aoa-GFLG-D4-G5-HbPG) in the cytotoxicity measurements that correlated well with the results of the internalization studies. These conjugates were found to be the most potent ones in the viability measurement and were proved to be taken up by HT-29 cells the most effectively. Depending on the type of polymer, the incorporation of the G5 spacer had an opposite effect on the cytotoxic activity of the conjugates. In the presence of the G5 spacer, the antitumour activity of the PEGylated conjugates decreased, while the cytotoxicity of the HbPG-containing conjugates increased, especially in the case of those with the D4 targeting peptide. We observed that some of the conjugates (Dau[double bond, length as m-dash]Aoa-GFLG-D4-HbPG and Dau[double bond, length as m-dash]Aoa-GFLG-GE11-G5-PEG) could not cause complete cell death, i.e., ˜0% viability value - characteristic for cell-free culturing medium - was not achieved even at the highest concentration, since their dose-response curves reached a plateau in a lower concentration range. In our opinion, this can be explained by the different characteristics of the highly hydrophobic peptide chain and the highly hydrophilic polymer segment. Due to this amphiphilic character, self-aggregation of the conjugates may occur, which then may block the accessibility of the targeting peptide for receptor binding, thereby decreasing the efficiency of the conjugate as well. This assumption is also confirmed by the turbidity results, since the observed low turbidity values may be caused by the possible formation of nanosized aggregates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
References
