Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00237
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
M1-RGD-PTX
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 2 Indication(s)
|
|||||
| Structure |
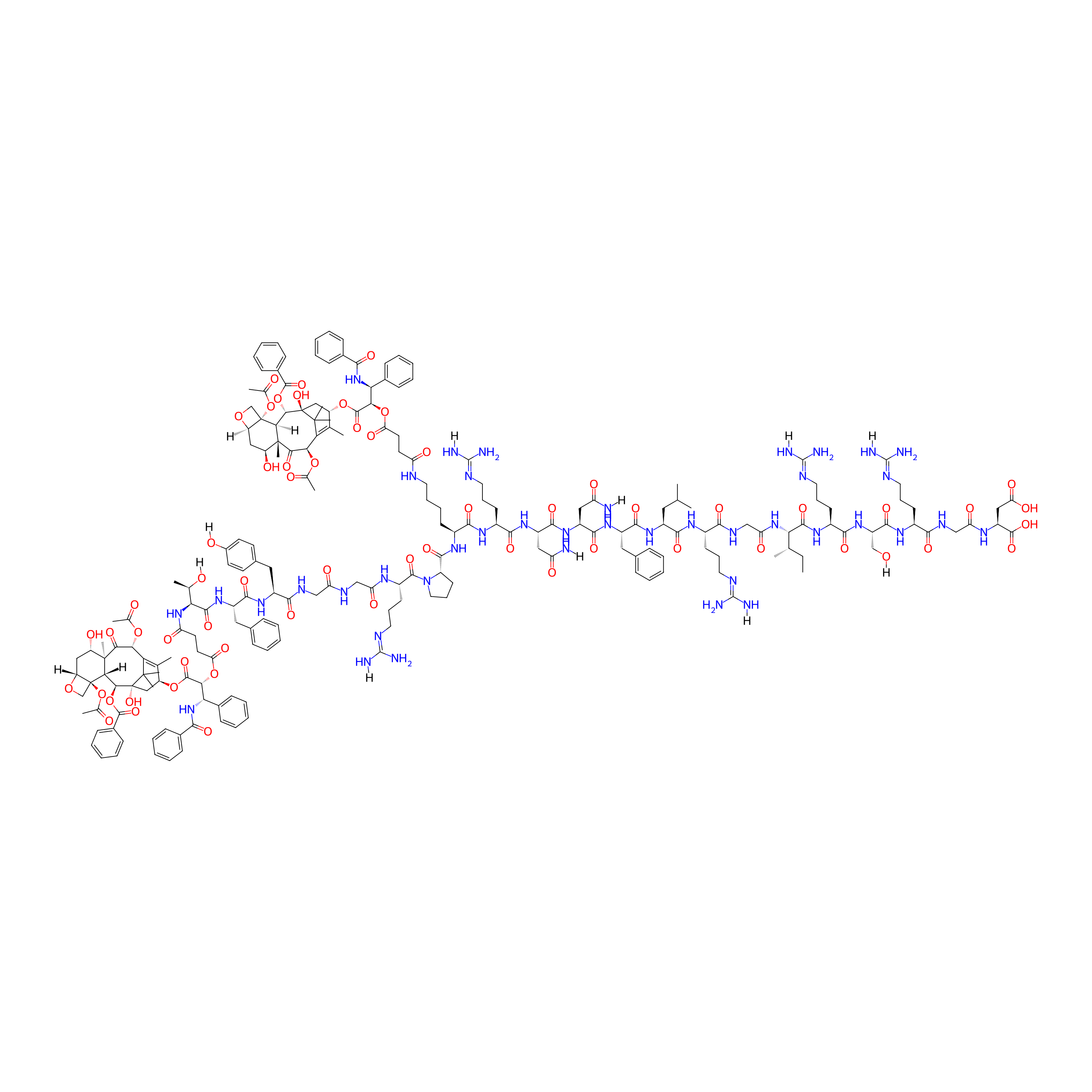
|
|||||
| Peptide Name |
M1-RGD
|
Peptide Info | ||||
| Drug Name |
Paclitaxel
|
Drug Info | ||||
| Therapeutic Target |
Microtubule (MT)
|
Target Info | ||||
| Linker Name |
Succinic Acid
|
Linker Info | ||||
| Peptide Modified Type |
Amino acid modifications
|
|||||
| Modified Segment |
RGD
|
|||||
| Ternimal Modification |
C-terminal modification
|
|||||
| Formula |
C209H277N41O61
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 4339.741 | ||||
| Lipid-water partition coefficient (xlogp) | -6.8766 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 44 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 64 | |||||
| Rotatable Bond Count (rotbonds) | 115 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Glioma | ||||
| Efficacy Data | Percent survival |
20%
|
|||
| Administration Time | 25 days | ||||
| Administration Dosage | Normalized to 17.3 mg/kg PTX | ||||
| Description |
The survival of the PTX group was markedly lower than that of the Vehicle group. M1-PTX did not improve survival, whereas the survival of the M1-RGD-PTX group was increased.
|
||||
| In Vivo Model | U87MG-Luc-bearing xenograft model. | ||||
| In Vitro Model | Glioblastoma | U87MG-Luc cell | CVCL_5J15 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Percent survival |
50%
|
|||
| Administration Time | 20 days | ||||
| Administration Dosage | 44 mg/kg | ||||
| Description |
PTX did not increase survival, while the PDC M1-RGD-PTX markedly increased survival.
|
||||
| In Vivo Model | MDA-MB-231BR mouse model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Glioma | ||||
| Efficacy Data | Cell viability |
55%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 100 nM | ||||
| Evaluation Method | MTS assay | ||||
| Description |
Both PDCs showed higher in vitro cytotoxicity than free PTX.
|
||||
| In Vitro Model | Glioblastoma | U-87MG cell | CVCL_0022 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Glioma | ||||
| Efficacy Data | Median lethal dose (LD50) |
152 mg/kg
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 100 nM | ||||
| Description |
The fitted LD50 for M1-RGD-PTX was 152 mg/kg (equivalent to 60 mg/kg PTX).
|
||||
| In Vitro Model | Glioblastoma | U-87MG cell | CVCL_0022 | ||
References
