Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00244
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
MPD3
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
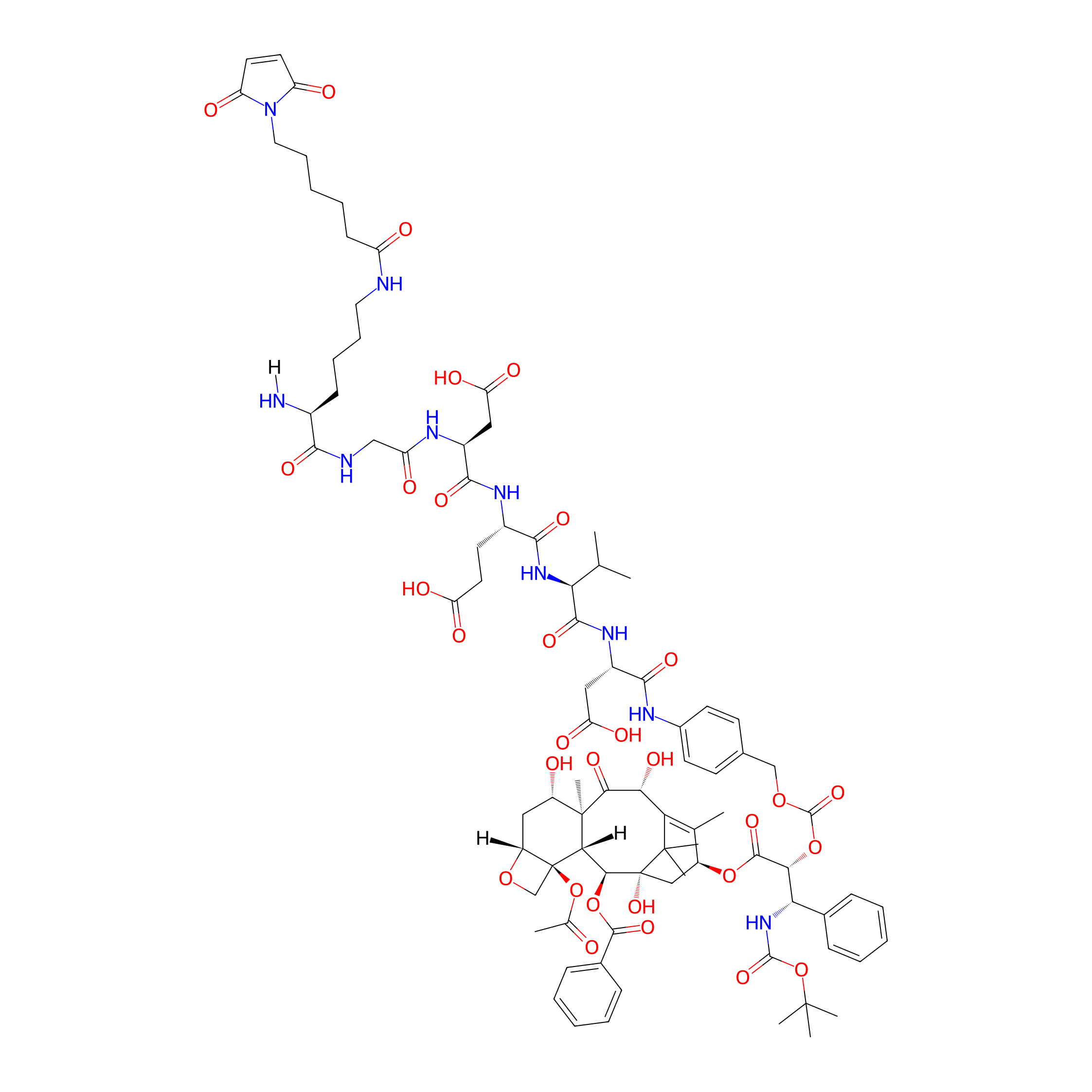
|
|||||
| Peptide Name |
DEVD
|
Peptide Info | ||||
| Receptor Name |
Caspase-3 (CASP3)
|
Receptor Info | ||||
| Drug Name |
Docetaxel
|
Drug Info | ||||
| Therapeutic Target |
Microtubule (MT)
|
Target Info | ||||
| Linker Name |
(4-Aminophenyl)methyl hydrogen carbonate
|
Linker Info | ||||
| Formula |
C87H112N10O31
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 1793.892 | ||||
| Lipid-water partition coefficient (xlogp) | 2.184 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 15 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 29 | |||||
| Rotatable Bond Count (rotbonds) | 43 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic triple-negative breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
48.40%
|
|||
| Administration Time | 3 weeks | ||||
| Administration Dosage | 3 mg/kg | ||||
| MOA of PDC |
To demonstrate our strategy, we developed a peptide-drug conjugate (PDC) composed of the caspase-3 cleavablepeptide sequence(DEVD), an albumin-binding functional moiety, and docetaxel linked to the peptide via self-immolated linker. Functionalization of albumin-binding domain enables intravenously injected PDC to spontaneously bind to circulatingserum albumin, which is employed as a drug carrier capable of inducing macropinocytic uptake of the conjugate. Furthermore, in order to combat tumoral heterogeneity, a unique peptide sequence of DEVD is deployed, which is a core component of the PDC that propagates bystander killing of cancer cells. More specifically, caspase-3 is triggered from albumin-metabolism induced apoptosis in PTEN-loss cancer cells, which in turn recognizes and activates extracellular PDC to release the payload. The released hydrophobic payload subsequently penetrates neighboring cancer cells, thereby inducing bystander killing in a non-selective manner, leading to the continual amplification ofin-situapoptosis. Additionally, to boost the apoptosis of initial cancer cells and caspase-3 expression, we used olaparib as a combination therapeutic agent. Given that olaparib is the first targeted therapy approved against metastatic TNBC with BRCA mutation, we reasoned that combination therapy with olaparib has the potential to induce apoptosis in BRCA mutant cancer cells and intensify tumoral apoptosis. In this study, we demonstrate the therapeutic potential of exploiting PTEN-loss drivenbiomimeticdrug delivery and the significant therapeutic benefit of combination therapy in the treatment of metastatic TNBC with altered PTEN and BRCA using our novel PDC.
Click to Show/Hide
|
||||
| Description |
To examine whether BRCA and PTEN alteration status affect the anti-tumor efficacy of olaparib/MPD3 combination, we performedin vivoexperiments on Hs578t-tumor and BT549-tumor bearing balb/cnude mice. As expected, olaparib monotherapy in Hs578t or BT549 tumors did not show detectable tumor suppressing efficacy, which could be ascribed to the poor sensitivity of olaparib in BRCA wild-type tumors. Interestingly, Hs578t tumors showed delayed onset of tumor growth inhibition effect by MPD3 monotherapy, producing 48.4% TGI rate on the last day of observation. Following an unexpected response to MPD3 in PTEN wild-type tumors, different control tumor specimens at day 1, 16, and 28 were examined by immunohistochemical staining to investigate tumor microenvironmental changes during the drug treatment course. We found evidence of increasing pattern of active caspase-3 expression over the course of time, which is thought to be associated with activation of localized extracellular prodrug and late tumor inhibiting efficacy in Hs578t tumors. Owing to the poor response to olaparib therapy, both MPD3 and olaparib/MPD3 displayed similar tumor inhibiting efficacies in Hs578t tumor bearing mice, suggesting that olaparib triggered apoptosis is needed to generate synergistic efficacy with MPD3. Lastly,in vivoexperiment results with BT549 tumor xenografted model confirmed the contribution of PTEN-loss alteration to thein vivoactivity of MPD3. MPD3 monotherapy was able to induce substantial tumor growth inhibition with 63.4% TGI. We ascribe these results mainly to the PTEN-loss induced upregulation of the macropinocytosis level, witnessed by significantly increased albumin uptake level in BT549cells. However, as with Hs578t xenograft model, the addition of olaparib did not cause significant tumor growth inhibition. Note that despite the comparable anti-cancer activity of olaparib/docetaxel to that of olaparib/MPD3, docetaxel combination treatment caused severebody weight loss, with 60% and 20% lethality in Hs578t and in BT549 tumor bearing mice, respectively. The results ofin vivoanti-cancer efficacies of MPD3 and olaparib in TNBC tumor models expressing different biomarkers are summarized inTable 1. Coefficients of drug interaction (CDI) values of the combination of olaparib/MPD3 were 0.04, 1.22 and 0.42 in MDA-MB-436, Hs578t, BT549 xenograft models, respectively, where CDI of <1, =1, or >1 indicates that the combination therapy is synergistic, additive, or antagonistic. These results provide compelling evidence that the BRCA and PTEN alteration status influence the efficacy of our combination strategy, showing that BRCA mutation and PTEN-loss contribute to the favorable anti-cancer efficacy of the strategy.
Click to Show/Hide
|
||||
| In Vivo Model | BRCA/PTEN wt Hs578t cells female BALB/cnude mice xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma of no special type | BRCA/PTEN WT Hs578t cell | CVCL_0332 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic triple-negative breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
63.40%
|
|||
| Administration Time | 3 weeks | ||||
| Administration Dosage | 3 mg/kg | ||||
| MOA of PDC |
To demonstrate our strategy, we developed a peptide-drug conjugate (PDC) composed of the caspase-3 cleavablepeptide sequence(DEVD), an albumin-binding functional moiety, and docetaxel linked to the peptide via self-immolated linker. Functionalization of albumin-binding domain enables intravenously injected PDC to spontaneously bind to circulatingserum albumin, which is employed as a drug carrier capable of inducing macropinocytic uptake of the conjugate. Furthermore, in order to combat tumoral heterogeneity, a unique peptide sequence of DEVD is deployed, which is a core component of the PDC that propagates bystander killing of cancer cells. More specifically, caspase-3 is triggered from albumin-metabolism induced apoptosis in PTEN-loss cancer cells, which in turn recognizes and activates extracellular PDC to release the payload. The released hydrophobic payload subsequently penetrates neighboring cancer cells, thereby inducing bystander killing in a non-selective manner, leading to the continual amplification ofin-situapoptosis. Additionally, to boost the apoptosis of initial cancer cells and caspase-3 expression, we used olaparib as a combination therapeutic agent. Given that olaparib is the first targeted therapy approved against metastatic TNBC with BRCA mutation, we reasoned that combination therapy with olaparib has the potential to induce apoptosis in BRCA mutant cancer cells and intensify tumoral apoptosis. In this study, we demonstrate the therapeutic potential of exploiting PTEN-loss drivenbiomimeticdrug delivery and the significant therapeutic benefit of combination therapy in the treatment of metastatic TNBC with altered PTEN and BRCA using our novel PDC.
Click to Show/Hide
|
||||
| Description |
To examine whether BRCA and PTEN alteration status affect the anti-tumor efficacy of olaparib/MPD3 combination, we performedin vivoexperiments on Hs578t-tumor and BT549-tumor bearing balb/cnude mice. As expected, olaparib monotherapy in Hs578t or BT549 tumors did not show detectable tumor suppressing efficacy, which could be ascribed to the poor sensitivity of olaparib in BRCA wild-type tumors. Interestingly, Hs578t tumors showed delayed onset of tumor growth inhibition effect by MPD3 monotherapy, producing 48.4% TGI rate on the last day of observation. Following an unexpected response to MPD3 in PTEN wild-type tumors, different control tumor specimens at day 1, 16, and 28 were examined by immunohistochemical staining to investigate tumor microenvironmental changes during the drug treatment course. We found evidence of increasing pattern of active caspase-3 expression over the course of time, which is thought to be associated with activation of localized extracellular prodrug and late tumor inhibiting efficacy in Hs578t tumors. Owing to the poor response to olaparib therapy, both MPD3 and olaparib/MPD3 displayed similar tumor inhibiting efficacies in Hs578t tumor bearing mice, suggesting that olaparib triggered apoptosis is needed to generate synergistic efficacy with MPD3. Lastly,in vivoexperiment results with BT549 tumor xenografted model confirmed the contribution of PTEN-loss alteration to thein vivoactivity of MPD3. MPD3 monotherapy was able to induce substantial tumor growth inhibition with 63.4% TGI. We ascribe these results mainly to the PTEN-loss induced upregulation of the macropinocytosis level, witnessed by significantly increased albumin uptake level in BT549cells. However, as with Hs578t xenograft model, the addition of olaparib did not cause significant tumor growth inhibition. Note that despite the comparable anti-cancer activity of olaparib/docetaxel to that of olaparib/MPD3, docetaxel combination treatment caused severebody weight loss, with 60% and 20% lethality in Hs578t and in BT549 tumor bearing mice, respectively. The results ofin vivoanti-cancer efficacies of MPD3 and olaparib in TNBC tumor models expressing different biomarkers are summarized inTable 1. Coefficients of drug interaction (CDI) values of the combination of olaparib/MPD3 were 0.04, 1.22 and 0.42 in MDA-MB-436, Hs578t, BT549 xenograft models, respectively, where CDI of <1, =1, or >1 indicates that the combination therapy is synergistic, additive, or antagonistic. These results provide compelling evidence that the BRCA and PTEN alteration status influence the efficacy of our combination strategy, showing that BRCA mutation and PTEN-loss contribute to the favorable anti-cancer efficacy of the strategy.
Click to Show/Hide
|
||||
| In Vivo Model | BRCA wt BT549 cells female BALB/cnude mice xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma of no special type | BRCA WT BT-549 cell | CVCL_1092 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic triple-negative breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
64.60%
|
|||
| Administration Time | 3 weeks | ||||
| Administration Dosage | 3 mg/kg | ||||
| MOA of PDC |
To demonstrate our strategy, we developed a peptide-drug conjugate (PDC) composed of the caspase-3 cleavablepeptide sequence(DEVD), an albumin-binding functional moiety, and docetaxel linked to the peptide via self-immolated linker. Functionalization of albumin-binding domain enables intravenously injected PDC to spontaneously bind to circulatingserum albumin, which is employed as a drug carrier capable of inducing macropinocytic uptake of the conjugate. Furthermore, in order to combat tumoral heterogeneity, a unique peptide sequence of DEVD is deployed, which is a core component of the PDC that propagates bystander killing of cancer cells. More specifically, caspase-3 is triggered from albumin-metabolism induced apoptosis in PTEN-loss cancer cells, which in turn recognizes and activates extracellular PDC to release the payload. The released hydrophobic payload subsequently penetrates neighboring cancer cells, thereby inducing bystander killing in a non-selective manner, leading to the continual amplification ofin-situapoptosis. Additionally, to boost the apoptosis of initial cancer cells and caspase-3 expression, we used olaparib as a combination therapeutic agent. Given that olaparib is the first targeted therapy approved against metastatic TNBC with BRCA mutation, we reasoned that combination therapy with olaparib has the potential to induce apoptosis in BRCA mutant cancer cells and intensify tumoral apoptosis. In this study, we demonstrate the therapeutic potential of exploiting PTEN-loss drivenbiomimeticdrug delivery and the significant therapeutic benefit of combination therapy in the treatment of metastatic TNBC with altered PTEN and BRCA using our novel PDC.
Click to Show/Hide
|
||||
| Description |
To examine whether BRCA and PTEN alteration status affect the anti-tumor efficacy of olaparib/MPD3 combination, we performedin vivoexperiments on Hs578t-tumor and BT549-tumor bearing balb/cnude mice. As expected, olaparib monotherapy in Hs578t or BT549 tumors did not show detectable tumor suppressing efficacy, which could be ascribed to the poor sensitivity of olaparib in BRCA wild-type tumors. Interestingly, Hs578t tumors showed delayed onset of tumor growth inhibition effect by MPD3 monotherapy, producing 48.4% TGI rate on the last day of observation. Following an unexpected response to MPD3 in PTEN wild-type tumors, different control tumor specimens at day 1, 16, and 28 were examined by immunohistochemical staining to investigate tumor microenvironmental changes during the drug treatment course. We found evidence of increasing pattern of active caspase-3 expression over the course of time, which is thought to be associated with activation of localized extracellular prodrug and late tumor inhibiting efficacy in Hs578t tumors. Owing to the poor response to olaparib therapy, both MPD3 and olaparib/MPD3 displayed similar tumor inhibiting efficacies in Hs578t tumor bearing mice, suggesting that olaparib triggered apoptosis is needed to generate synergistic efficacy with MPD3. Lastly,in vivoexperiment results with BT549 tumor xenografted model confirmed the contribution of PTEN-loss alteration to thein vivoactivity of MPD3. MPD3 monotherapy was able to induce substantial tumor growth inhibition with 63.4% TGI. We ascribe these results mainly to the PTEN-loss induced upregulation of the macropinocytosis level, witnessed by significantly increased albumin uptake level in BT549cells. However, as with Hs578t xenograft model, the addition of olaparib did not cause significant tumor growth inhibition. Note that despite the comparable anti-cancer activity of olaparib/docetaxel to that of olaparib/MPD3, docetaxel combination treatment caused severebody weight loss, with 60% and 20% lethality in Hs578t and in BT549 tumor bearing mice, respectively. The results ofin vivoanti-cancer efficacies of MPD3 and olaparib in TNBC tumor models expressing different biomarkers are summarized inTable 1. Coefficients of drug interaction (CDI) values of the combination of olaparib/MPD3 were 0.04, 1.22 and 0.42 in MDA-MB-436, Hs578t, BT549 xenograft models, respectively, where CDI of <1, =1, or >1 indicates that the combination therapy is synergistic, additive, or antagonistic. These results provide compelling evidence that the BRCA and PTEN alteration status influence the efficacy of our combination strategy, showing that BRCA mutation and PTEN-loss contribute to the favorable anti-cancer efficacy of the strategy.
Click to Show/Hide
|
||||
| In Vivo Model | BRCA1 mut MDA-MB-436 cells female BALB/cnude mice xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma of no special type | BRCA1 mut MDA-MB-436 cell | CVCL_0623 | ||
References
