Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00327
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
SG3299
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 2 Indication(s)
|
|||||
| Structure |
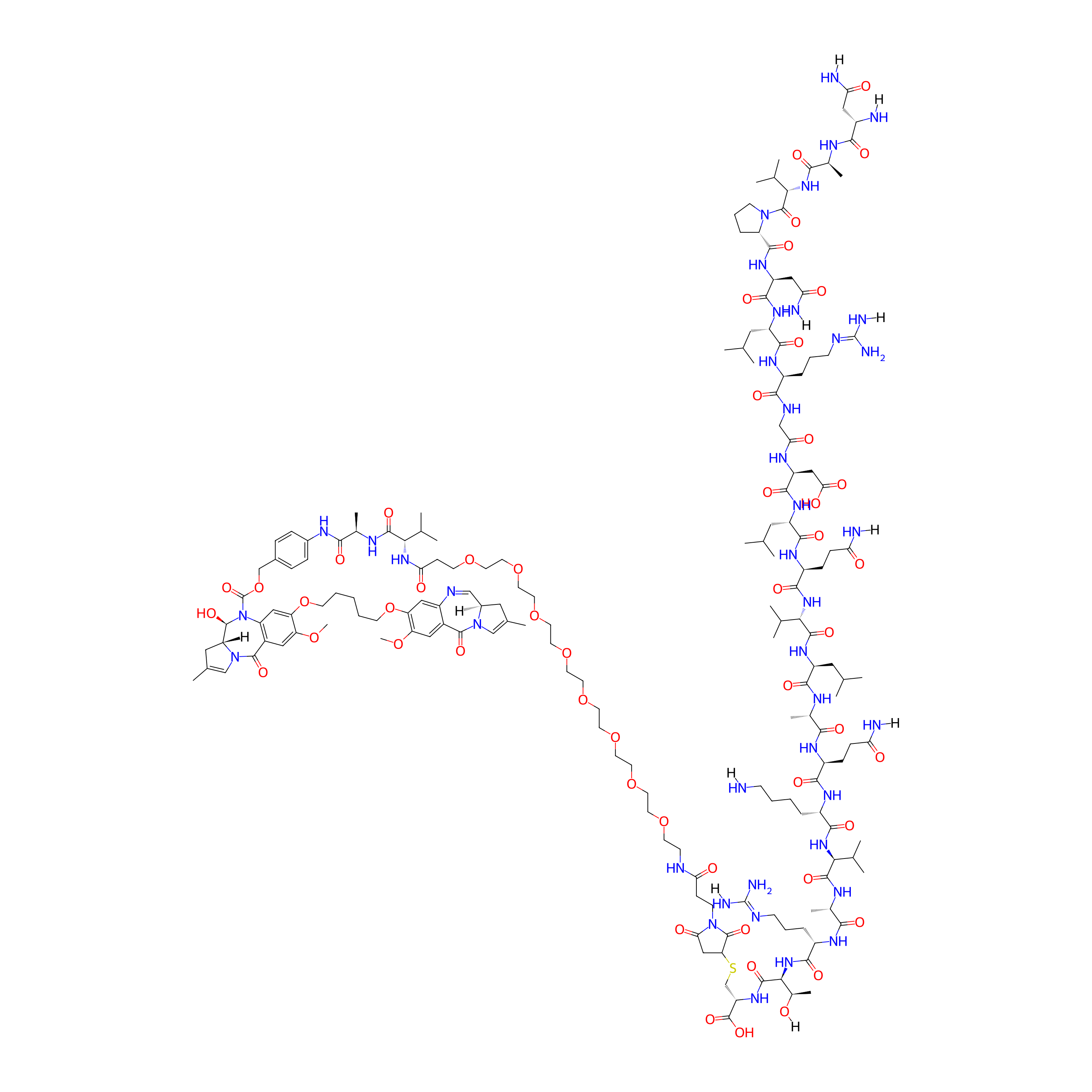
|
|||||
| Peptide Name |
A20FMDV2
|
Peptide Info | ||||
| Receptor Name |
Integrin alpha-V; Integrin beta-6 (ITGAV; ITGB6)
|
Receptor Info | ||||
| Drug Name |
Pyrrolobenzodiazepine (PBD) dimer
|
Drug Info | ||||
| Therapeutic Target |
Human Deoxyribonucleic acid (hDNA)
|
Target Info | ||||
| Linker Name |
Val-Ala
|
Linker Info | ||||
| Formula |
C171H269N41O52S
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 3763.335 | ||||
| Lipid-water partition coefficient (xlogp) | -6.9815 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 37 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 56 | |||||
| Rotatable Bond Count (rotbonds) | 128 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
53.9 ± 23.7%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 10 µg/kg | ||||
| Evaluation Method | Tumor volume detection assay | ||||
| MOA of PDC |
SG3299 Treatment Reduces Pancreatic Tumor Growth by inducing DNA Damage and Apoptosis
|
||||
| Description |
Tumors treated with SG3511 and SG3299 exhibited significant reductions in size (P<0.0001, 53.9±23.7% and 34.8±4.6% after 21 days respectively) but there was no significant difference in the effect of either treatment (P=0.24, after 30 days).
|
||||
| In Vivo Model | Athymic CD1Nu/Nu female mice bearing human A375Ppuro xenograft tumours. | ||||
| In Vitro Model | Amelanotic melanoma | A375P-puro cell | CVCL_5F66 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
76%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 20 µg/kg | ||||
| Evaluation Method | Tumor volume detection assay | ||||
| MOA of PDC |
SG3299 Treatment Reduces Pancreatic Tumor Growth by inducing DNA Damage and Apoptosis
|
||||
| Description |
SG3299 significantly reduced Panc0403/PS1 xenograft tumor growth by 75.8±6% (P<0.001) compared with PBS treatment and by 60.4±9.8% (P<0.05) compared with SG3511 therapy.
|
||||
| In Vivo Model | Athymic CD1Nu/Nu female mice bearing human A375Pbeta6 xenograft tumours. | ||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Panc 04.03 PS1 cell | CVCL_1636 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
79 ± 7%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 10 µg/kg | ||||
| Evaluation Method | Tumor volume detection assay | ||||
| MOA of PDC |
SG3299 Treatment Reduces Pancreatic Tumor Growth by inducing DNA Damage and Apoptosis
|
||||
| Description |
In contrast, both SG3299 and SG3511 reduced A375P6 tumor growth compared with PBS treatment (79±7% and 56.9±16.2% respectively, P<0.0001) and SG3299 reduced growth by 2.3-fold more than SG3511 (P<0.0001).
|
||||
| In Vivo Model | Athymic CD1Nu/Nu female mice bearing human A375Pbeta6 xenograft tumours. | ||||
| In Vitro Model | Amelanotic melanoma | A375P-beta6 cell | CVCL_5F66 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
84%
|
|||
| Administration Time | 35 days | ||||
| Administration Dosage | 10 µg/kg | ||||
| Evaluation Method | Tumor volume detection assay | ||||
| MOA of PDC |
SG3299 Treatment Reduces Pancreatic Tumor Growth by inducing DNA Damage and Apoptosis
|
||||
| Description |
Capan-1 xenografts responded in a similar manner to A375P6 xenografts upon 10 ug/kg tri-weekly treatment, with significant growth inhibition with SG3511 and SG3299 (P<0.0001). Again, SG3299 inhibited tumor growth significantly more than SG3511 (P<0.0001).
|
||||
| In Vivo Model | Athymic CD1Nu/Nu female mice bearing human Capan-1 xenograft tumours. | ||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Capan-1 cell | CVCL_0237 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
97.7 ± 2
|
|||
| Administration Time | Bi-weekly for 4 weeks | ||||
| Administration Dosage | 25 µg/kg | ||||
| Evaluation Method | Tumor volume detection assay | ||||
| MOA of PDC |
SG3299 Treatment Reduces Pancreatic Tumor Growth by inducing DNA Damage and Apoptosis
|
||||
| Description |
Again, SG3299 significantly reduced Capan-1 tumor growth achieving 97.7±2% (P<0.0001) and 96.1±3.4% (P<0.0001) reductions compared to PBS and SG3511, respectively.
|
||||
| In Vivo Model | Athymic CD1Nu/Nu female mice bearing human A375Pbeta6 xenograft tumours. | ||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Capan-1 cell | CVCL_0237 | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
24 nM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | WST-1 assay | ||||
| MOA of PDC |
The integrin vβ6 is expressed on ˜85% of pancreatic cancers with minimal expression in healthy tissues, and thus is a valid therapeutic target. We previously developed the A20FMDV2 peptide that binds with high-affinity to vβ6. SG3299 is a peptide-toxin conjugate that conjugates A20FMDV2 to a synthetic pyrrolobenzodiazepine (PBD) dimer cytotoxic warhead with a cathepsin B-cleavable valine-alanine linker. We have shown that SG3299 is highly effective in subcutaneous pancreatic cancer xenografts in immunodeficient models, with prolonged survival and tumour eliminations.
Click to Show/Hide
|
||||
| Description |
The relative specificity of SG3299 for v6, and the in vitro cytotoxic effect of SG3299 on v6-expressing murine cancer cells was confirmed with a growth inhibition assay performed on TB32043mb6s2 and TB32043 cells (high & negative v6 expression respectively). Cell viability was evaluated with a WST-1 assay following treatment with 0-105 nM of SG3299 or SG3511. The IC50 of v6-targeted SG3299 in TB32043mb6s2 was over 15-fold lower than in TB32043 cells (24 nM vs 418 nM, p < 0.001). There was no significant difference in the IC50 values for the non-targeted SG3511 between TB32043mb6s2 and TB32043 cells (223 vs 300 nM, p = 0.17).
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Pancreatic ductal adenocarcinoma cell TB32043mb6s2 | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
418 nM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | WST-1 assay | ||||
| MOA of PDC |
The integrin vβ6 is expressed on ˜85% of pancreatic cancers with minimal expression in healthy tissues, and thus is a valid therapeutic target. We previously developed the A20FMDV2 peptide that binds with high-affinity to vβ6. SG3299 is a peptide-toxin conjugate that conjugates A20FMDV2 to a synthetic pyrrolobenzodiazepine (PBD) dimer cytotoxic warhead with a cathepsin B-cleavable valine-alanine linker. We have shown that SG3299 is highly effective in subcutaneous pancreatic cancer xenografts in immunodeficient models, with prolonged survival and tumour eliminations.
Click to Show/Hide
|
||||
| Description |
The relative specificity of SG3299 for v6, and the in vitro cytotoxic effect of SG3299 on v6-expressing murine cancer cells was confirmed with a growth inhibition assay performed on TB32043mb6s2 and TB32043 cells (high & negative v6 expression respectively). Cell viability was evaluated with a WST-1 assay following treatment with 0-105 nM of SG3299 or SG3511. The IC50 of v6-targeted SG3299 in TB32043mb6s2 was over 15-fold lower than in TB32043 cells (24 nM vs 418 nM, p < 0.001). There was no significant difference in the IC50 values for the non-targeted SG3511 between TB32043mb6s2 and TB32043 cells (223 vs 300 nM, p = 0.17).
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Pancreatic ductal adenocarcinoma cell TB32043 | Homo sapiens | ||
References
