Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00371
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
Copper dotatate Cu-64
|
|||||
| Synonyms |
64Cu-dotatate; Copper 64-DOTA-tate; Copper Cu-64 dotatate; Copper dotatate Cu-64; Copper oxodotreotide Cu-64
Click to Show/Hide
|
|||||
| PDC Status |
Approved
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
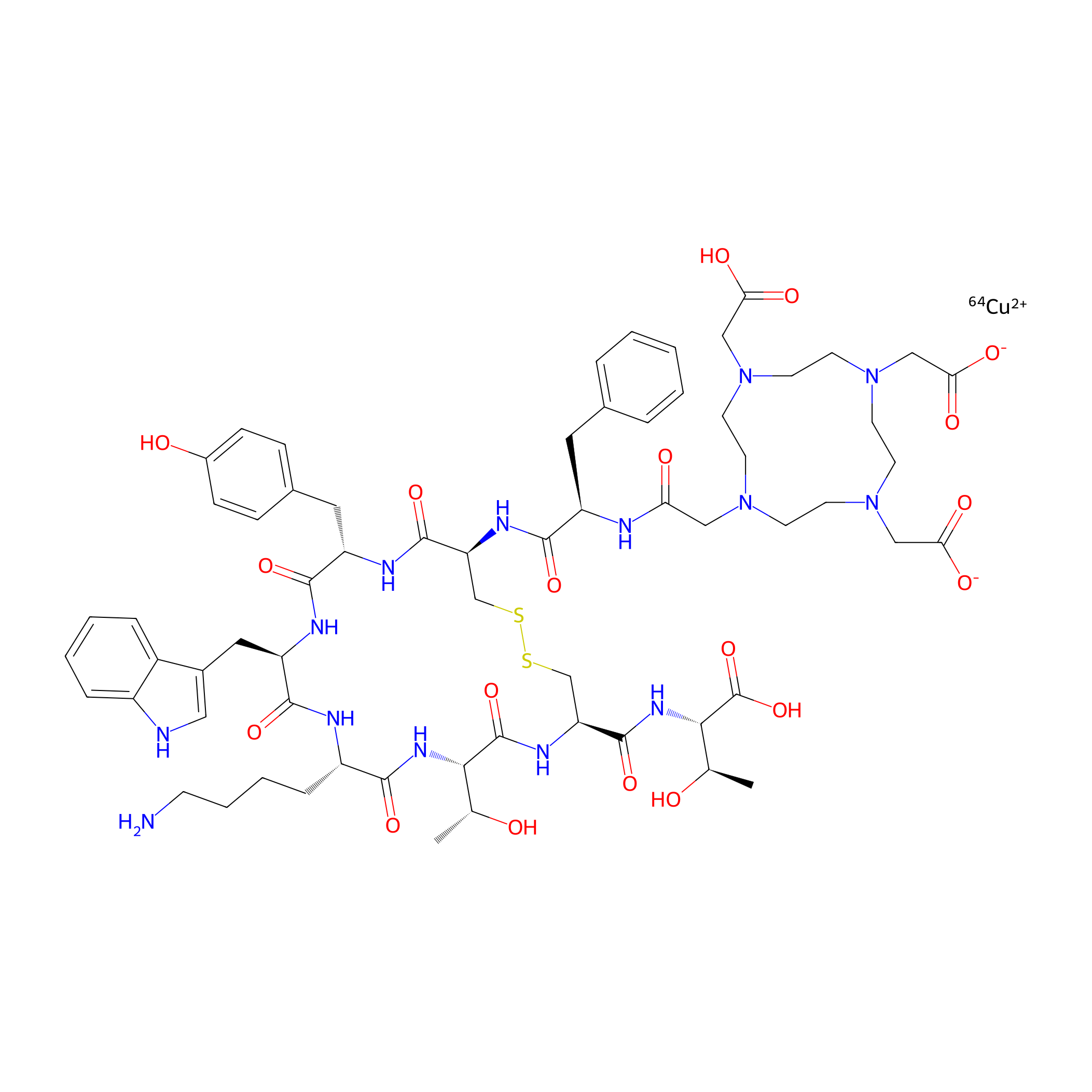
|
|||||
| Peptide Name |
3-Tyr-Octreotate
|
Peptide Info | ||||
| Receptor Name |
Somatostatin receptor type 1 (SSTR1)
|
Receptor Info | ||||
| Drug Name |
Copper-64
|
Drug Info | ||||
| Therapeutic Target |
Human Deoxyribonucleic acid (hDNA)
|
Target Info | ||||
| Linker Name |
Amide bond
|
Linker Info | ||||
| Peptide Modified Type |
Amino acid modifications; Cyclization modification
|
|||||
| Modified Segment |
D-amino acids
|
|||||
| Ternimal Modification |
N-terminal modification
|
|||||
| Drugbank ID | ||||||
| Formula |
C65H88CuN14O19S2
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 1497.561764 | ||||
| Lipid-water partition coefficient (xlogp) | -6.0465 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 15 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 24 | |||||
| Rotatable Bond Count (rotbonds) | 26 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Neuroendocrine neoplasms | ||||
| Efficacy Data | PET50% overall sensitivity |
100%
|
|||
| Patients Enrolled |
38 patients with NEN.
|
||||
| Administration Dosage | 142 MBq | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
Somatostatin analogues radiolabeled with the radioisotope 68Ga, e.g. [68Ga]Ga-DOTATATE and [68Ga]Ga-DOTATOC, are currently the predominately used tracers for PET imaging of patients with NEN. However, the 64Cu-labeled SSR targeting somatostatin analogue [64Cu]Cu-DOTATATE is emerging as an alternative to 68Ga-labeled radiotracers. Although 64Cu has a lower branching ratio for + decay than 68Ga (18% vs 89%), 64Cu has the advantage of a longer half-life (12.7 h vs 68 min) and shorter positron range (0.7 mm vs 3.5 mm) where the latter at least in theory should lead to better spatial resolution. The effective whole-body dose is 6.3 mSv for a 200 MBq injection of [64Cu]Cu-DOTATATE or 4.7 mSv for the United States Food and Drug Administration (FDA) recommended dose of 148 MBq.
Click to Show/Hide
|
||||
| Description |
The median [64Cu]Cu-DOTATATE activity dose could be reduced from 191 to 142 MBq without decline in diagnostic image quality (P = 0.62), diagnostic lesion confidence (P = 1.0), or number of lesions detected in major organs or regions (P = 0.19-0.71). Sensitivity and specificity for detection of liver disease were 100% (26/26 patients) and 100% (12/12), respectively, for both PET75% and PET50%. Overall sensitivity for detection of NEN was 100% (26/26) for both PET75% and PET50%, and overall specificities were 92% (11/12) and 100% (12/12) for PET75 and PET50, respectively. Following dose-blinded post hoc review, the PET75% specificity was adjusted to 100% (12/12).
Click to Show/Hide
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Neuroendocrine neoplasms | ||||
| Efficacy Data | PET50% overall specificities |
100%
|
|||
| Patients Enrolled |
38 patients with NEN.
|
||||
| Administration Dosage | 142 MBq | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
Somatostatin analogues radiolabeled with the radioisotope 68Ga, e.g. [68Ga]Ga-DOTATATE and [68Ga]Ga-DOTATOC, are currently the predominately used tracers for PET imaging of patients with NEN. However, the 64Cu-labeled SSR targeting somatostatin analogue [64Cu]Cu-DOTATATE is emerging as an alternative to 68Ga-labeled radiotracers. Although 64Cu has a lower branching ratio for + decay than 68Ga (18% vs 89%), 64Cu has the advantage of a longer half-life (12.7 h vs 68 min) and shorter positron range (0.7 mm vs 3.5 mm) where the latter at least in theory should lead to better spatial resolution. The effective whole-body dose is 6.3 mSv for a 200 MBq injection of [64Cu]Cu-DOTATATE or 4.7 mSv for the United States Food and Drug Administration (FDA) recommended dose of 148 MBq.
Click to Show/Hide
|
||||
| Description |
The median [64Cu]Cu-DOTATATE activity dose could be reduced from 191 to 142 MBq without decline in diagnostic image quality (P = 0.62), diagnostic lesion confidence (P = 1.0), or number of lesions detected in major organs or regions (P = 0.19-0.71). Sensitivity and specificity for detection of liver disease were 100% (26/26 patients) and 100% (12/12), respectively, for both PET75% and PET50%. Overall sensitivity for detection of NEN was 100% (26/26) for both PET75% and PET50%, and overall specificities were 92% (11/12) and 100% (12/12) for PET75 and PET50, respectively. Following dose-blinded post hoc review, the PET75% specificity was adjusted to 100% (12/12).
Click to Show/Hide
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Neuroendocrine neoplasms | ||||
| Efficacy Data | PET75% overall sensitivity |
100%
|
|||
| Patients Enrolled |
38 patients with NEN.
|
||||
| Administration Dosage | 142 MBq | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
Somatostatin analogues radiolabeled with the radioisotope 68Ga, e.g. [68Ga]Ga-DOTATATE and [68Ga]Ga-DOTATOC, are currently the predominately used tracers for PET imaging of patients with NEN. However, the 64Cu-labeled SSR targeting somatostatin analogue [64Cu]Cu-DOTATATE is emerging as an alternative to 68Ga-labeled radiotracers. Although 64Cu has a lower branching ratio for + decay than 68Ga (18% vs 89%), 64Cu has the advantage of a longer half-life (12.7 h vs 68 min) and shorter positron range (0.7 mm vs 3.5 mm) where the latter at least in theory should lead to better spatial resolution. The effective whole-body dose is 6.3 mSv for a 200 MBq injection of [64Cu]Cu-DOTATATE or 4.7 mSv for the United States Food and Drug Administration (FDA) recommended dose of 148 MBq.
Click to Show/Hide
|
||||
| Description |
The median [64Cu]Cu-DOTATATE activity dose could be reduced from 191 to 142 MBq without decline in diagnostic image quality (P = 0.62), diagnostic lesion confidence (P = 1.0), or number of lesions detected in major organs or regions (P = 0.19-0.71). Sensitivity and specificity for detection of liver disease were 100% (26/26 patients) and 100% (12/12), respectively, for both PET75% and PET50%. Overall sensitivity for detection of NEN was 100% (26/26) for both PET75% and PET50%, and overall specificities were 92% (11/12) and 100% (12/12) for PET75 and PET50, respectively. Following dose-blinded post hoc review, the PET75% specificity was adjusted to 100% (12/12).
Click to Show/Hide
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Neuroendocrine neoplasms | ||||
| Efficacy Data | PET75% overall specificities |
92%
|
|||
| Patients Enrolled |
38 patients with NEN.
|
||||
| Administration Dosage | 142 MBq | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
Somatostatin analogues radiolabeled with the radioisotope 68Ga, e.g. [68Ga]Ga-DOTATATE and [68Ga]Ga-DOTATOC, are currently the predominately used tracers for PET imaging of patients with NEN. However, the 64Cu-labeled SSR targeting somatostatin analogue [64Cu]Cu-DOTATATE is emerging as an alternative to 68Ga-labeled radiotracers. Although 64Cu has a lower branching ratio for + decay than 68Ga (18% vs 89%), 64Cu has the advantage of a longer half-life (12.7 h vs 68 min) and shorter positron range (0.7 mm vs 3.5 mm) where the latter at least in theory should lead to better spatial resolution. The effective whole-body dose is 6.3 mSv for a 200 MBq injection of [64Cu]Cu-DOTATATE or 4.7 mSv for the United States Food and Drug Administration (FDA) recommended dose of 148 MBq.
Click to Show/Hide
|
||||
| Description |
The median [64Cu]Cu-DOTATATE activity dose could be reduced from 191 to 142 MBq without decline in diagnostic image quality (P = 0.62), diagnostic lesion confidence (P = 1.0), or number of lesions detected in major organs or regions (P = 0.19-0.71). Sensitivity and specificity for detection of liver disease were 100% (26/26 patients) and 100% (12/12), respectively, for both PET75% and PET50%. Overall sensitivity for detection of NEN was 100% (26/26) for both PET75% and PET50%, and overall specificities were 92% (11/12) and 100% (12/12) for PET75 and PET50, respectively. Following dose-blinded post hoc review, the PET75% specificity was adjusted to 100% (12/12).
Click to Show/Hide
|
||||
References
