Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_02015
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
Piflufolastat F-18
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
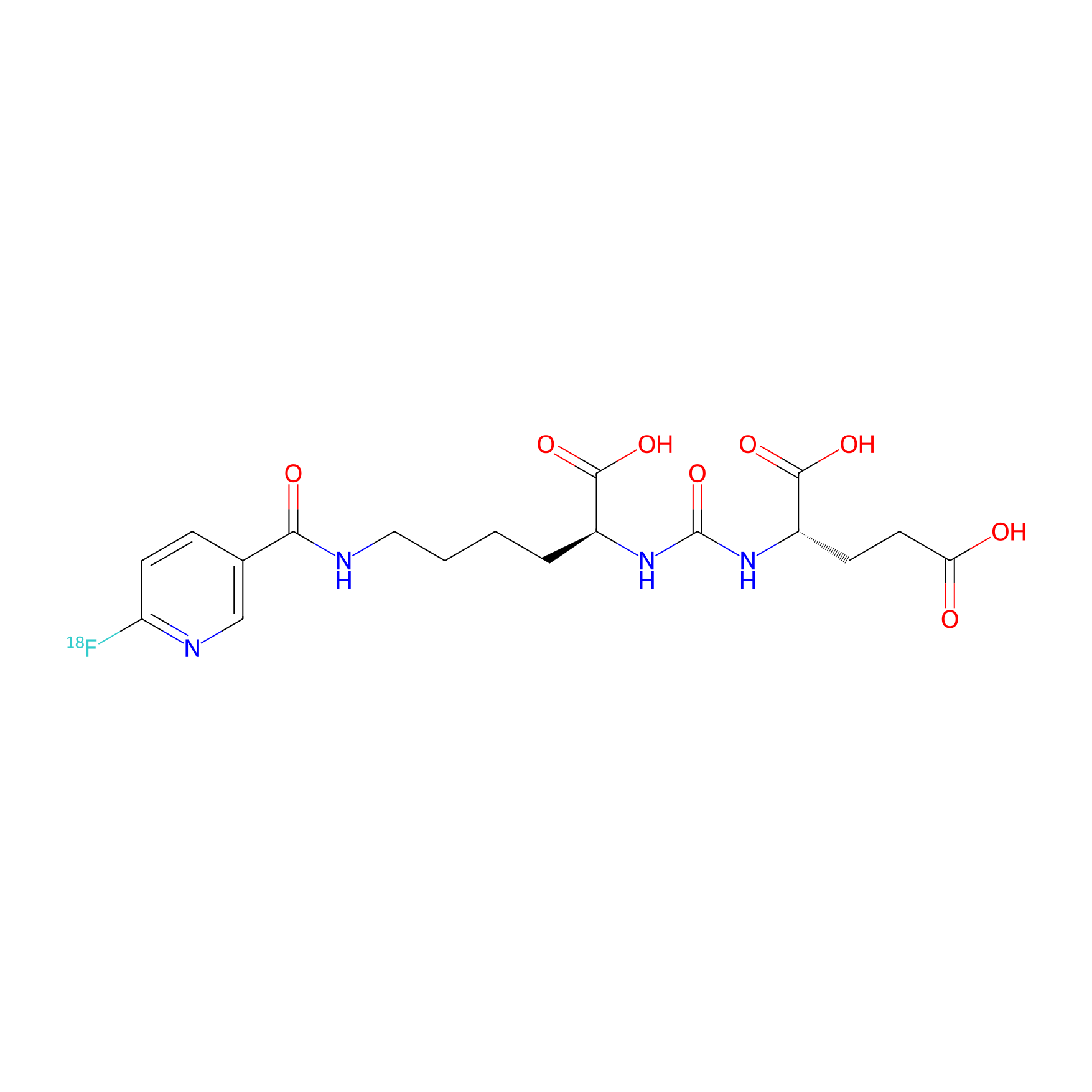
|
|||||
| Peptide Name |
PSMA-617
|
Peptide Info | ||||
| Receptor Name |
Glutamate carboxypeptidase 2 (FOLH1)
|
Receptor Info | ||||
| Drug Name |
Fluorine-18
|
Drug Info | ||||
| Therapeutic Target |
Human Deoxyribonucleic acid (hDNA)
|
Target Info | ||||
| Linker Name |
Amide bond
|
Linker Info | ||||
| Peptide Modified Type |
The modification of binding with chemical molecules
|
|||||
| Modified Segment |
Urea
|
|||||
| Formula |
C18H23FN4O8
|
|||||
| #Ro5 Violations (Lipinski): 2 | Molecular Weight | 441.402938 | ||||
| Lipid-water partition coefficient (xlogp) | 0.1912 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 6 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | |||||
| Rotatable Bond Count (rotbonds) | 13 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Dysgeusia |
3%
|
|||
| Patients Enrolled |
345 patients with prostate cancer.
|
||||
| Administration Dosage | 8-10 mCi | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
PSMA, also known as glutamate carboxypeptidase 2 or folate hydrolase 1, is highly expressed in nearly all prostate cancers. Structurally, PSMA is a large extracellular carboxypeptidase domain that is tethered to the cellular membrane by a short transmembrane domain. Physiologically, PSMA is also expressed in normal tissue, including non-malignant prostate tissue, the kidney, the small intestine, and the central nervous system. In most tissues, PSMA likely contributes to folate uptake by cleaving the C-terminal glutamate from folic acid (Figure 1). Within the brain, PSMA regulates the level of N-acetylaspartylglutamate (NAAG) by catalyzing its degradation to glutamate. Structure of PSMA substrates and inhibitors. Left: structure of the PSMA substrate folic acid. The peptide bond (dashed box) linking the P1 glutamate residue (blue) to the remainder of the substrate (P1, red) is cleaved by PSMA. Right: Structure of the PSMA-PET agents 18F-DCFPyL and 68Ga-PSMA-11. The scissile peptide bond has been replaced by a non-hydrolyzable urea motif (red) to generate PMSA inhibitors. The P1 glutamate residue is essential for binding to PSMA, but a wide degree of variability is tolerated in the opposing end of the structure.
Click to Show/Hide
|
||||
| Description |
Within cohort A, the primary end point was the sensitivity and specificity for detection of metastases to the pelvic lymph nodes by 18F-DCFPyL PET/CT. These findings were validated by histopathology after radical proctectomy and lymph node dissection (Figure 6). When all confirmed positive lymph node metastases were considered, the sensitivity of 18F-DCFPyL PET/CT was 40%, with a 98% specificity. It is perhaps unsurprising that histopathology is more sensitivity for the detection of early metastases as a sufficient number of malignant cells must accumulate before any imaging modality will be positive. However, a post-hoc analysis considering only lesions >5 mm found the sensitivity of 18F-DCFPyL PET/CT increased to 60%. Despite this, across all lesions, 18F-DCFPyL PET/CT significantly outperformed conventional CT pelvic imaging with markedly improved specificity (97.7% vs. 65.1%) and positive predictive value (85.1% vs. 28.3%) for pelvic lymph node metastases, as well as sensitivity for disease within the prostate (96.8% vs. 35.9%).
Click to Show/Hide
|
||||
| Half life period | 8 h | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Fatigue |
1%
|
|||
| Patients Enrolled |
345 patients with prostate cancer.
|
||||
| Administration Dosage | 8-10 mCi | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
PSMA, also known as glutamate carboxypeptidase 2 or folate hydrolase 1, is highly expressed in nearly all prostate cancers. Structurally, PSMA is a large extracellular carboxypeptidase domain that is tethered to the cellular membrane by a short transmembrane domain. Physiologically, PSMA is also expressed in normal tissue, including non-malignant prostate tissue, the kidney, the small intestine, and the central nervous system. In most tissues, PSMA likely contributes to folate uptake by cleaving the C-terminal glutamate from folic acid (Figure 1). Within the brain, PSMA regulates the level of N-acetylaspartylglutamate (NAAG) by catalyzing its degradation to glutamate. Structure of PSMA substrates and inhibitors. Left: structure of the PSMA substrate folic acid. The peptide bond (dashed box) linking the P1 glutamate residue (blue) to the remainder of the substrate (P1, red) is cleaved by PSMA. Right: Structure of the PSMA-PET agents 18F-DCFPyL and 68Ga-PSMA-11. The scissile peptide bond has been replaced by a non-hydrolyzable urea motif (red) to generate PMSA inhibitors. The P1 glutamate residue is essential for binding to PSMA, but a wide degree of variability is tolerated in the opposing end of the structure.
Click to Show/Hide
|
||||
| Description |
Within cohort A, the primary end point was the sensitivity and specificity for detection of metastases to the pelvic lymph nodes by 18F-DCFPyL PET/CT. These findings were validated by histopathology after radical proctectomy and lymph node dissection (Figure 6). When all confirmed positive lymph node metastases were considered, the sensitivity of 18F-DCFPyL PET/CT was 40%, with a 98% specificity. It is perhaps unsurprising that histopathology is more sensitivity for the detection of early metastases as a sufficient number of malignant cells must accumulate before any imaging modality will be positive. However, a post-hoc analysis considering only lesions >5 mm found the sensitivity of 18F-DCFPyL PET/CT increased to 60%. Despite this, across all lesions, 18F-DCFPyL PET/CT significantly outperformed conventional CT pelvic imaging with markedly improved specificity (97.7% vs. 65.1%) and positive predictive value (85.1% vs. 28.3%) for pelvic lymph node metastases, as well as sensitivity for disease within the prostate (96.8% vs. 35.9%).
Click to Show/Hide
|
||||
| Half life period | 8 h | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Fatigue |
1%
|
|||
| Patients Enrolled |
208 patients with prostate cancer.
|
||||
| Administration Dosage | 8-10 mCi | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
PSMA, also known as glutamate carboxypeptidase 2 or folate hydrolase 1, is highly expressed in nearly all prostate cancers. Structurally, PSMA is a large extracellular carboxypeptidase domain that is tethered to the cellular membrane by a short transmembrane domain. Physiologically, PSMA is also expressed in normal tissue, including non-malignant prostate tissue, the kidney, the small intestine, and the central nervous system. In most tissues, PSMA likely contributes to folate uptake by cleaving the C-terminal glutamate from folic acid (Figure 1). Within the brain, PSMA regulates the level of N-acetylaspartylglutamate (NAAG) by catalyzing its degradation to glutamate. Structure of PSMA substrates and inhibitors. Left: structure of the PSMA substrate folic acid. The peptide bond (dashed box) linking the P1 glutamate residue (blue) to the remainder of the substrate (P1, red) is cleaved by PSMA. Right: Structure of the PSMA-PET agents 18F-DCFPyL and 68Ga-PSMA-11. The scissile peptide bond has been replaced by a non-hydrolyzable urea motif (red) to generate PMSA inhibitors. The P1 glutamate residue is essential for binding to PSMA, but a wide degree of variability is tolerated in the opposing end of the structure.
Click to Show/Hide
|
||||
| Description |
All patients were dosed with 9 mCi of 18F-DCFPyL and PET/CT images were obtained. 18F-DCFPyL avid lesions were detected in ~60% of the patients. To confirm the metastatic nature of these lesions, biopsies were obtained, if possible. If the lesion was not amendable to biopsy, validation is obtained by focused conventional follow-up imaging or biochemical response to external beam radiotherapy. The differentiation between the number of true metastases validated by other techniques (i.e. true positives, TP) and false positives (FP) allows for the calculation of the correct localization rate (CLR), where the CLR = TP/(TP + FP) * 100 (essentially, PPV with the added requirement of anatomic co-localization). Across all patients, the CLR ranged from 85% to 87% between readers, however the CLR improved as the baseline PSA increased, from 75% to 96% (PSA < 1.0 to PSA ≥ 5.0, respectively). The PPV of the 18F-DCFPyL PET/CT was found to vary by anatomic region and was highest for prostatic lesions (80%) and pelvic lymph nodes (71%), but lower for extrapelvic visceral/soft tissue masses (29%).
Click to Show/Hide
|
||||
| Half life period | 8 h | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Headache rate |
2%
|
|||
| Patients Enrolled |
345 patients with prostate cancer.
|
||||
| Administration Dosage | 8-10 mCi | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
PSMA, also known as glutamate carboxypeptidase 2 or folate hydrolase 1, is highly expressed in nearly all prostate cancers. Structurally, PSMA is a large extracellular carboxypeptidase domain that is tethered to the cellular membrane by a short transmembrane domain. Physiologically, PSMA is also expressed in normal tissue, including non-malignant prostate tissue, the kidney, the small intestine, and the central nervous system. In most tissues, PSMA likely contributes to folate uptake by cleaving the C-terminal glutamate from folic acid (Figure 1). Within the brain, PSMA regulates the level of N-acetylaspartylglutamate (NAAG) by catalyzing its degradation to glutamate. Structure of PSMA substrates and inhibitors. Left: structure of the PSMA substrate folic acid. The peptide bond (dashed box) linking the P1 glutamate residue (blue) to the remainder of the substrate (P1, red) is cleaved by PSMA. Right: Structure of the PSMA-PET agents 18F-DCFPyL and 68Ga-PSMA-11. The scissile peptide bond has been replaced by a non-hydrolyzable urea motif (red) to generate PMSA inhibitors. The P1 glutamate residue is essential for binding to PSMA, but a wide degree of variability is tolerated in the opposing end of the structure.
Click to Show/Hide
|
||||
| Description |
Within cohort A, the primary end point was the sensitivity and specificity for detection of metastases to the pelvic lymph nodes by 18F-DCFPyL PET/CT. These findings were validated by histopathology after radical proctectomy and lymph node dissection (Figure 6). When all confirmed positive lymph node metastases were considered, the sensitivity of 18F-DCFPyL PET/CT was 40%, with a 98% specificity. It is perhaps unsurprising that histopathology is more sensitivity for the detection of early metastases as a sufficient number of malignant cells must accumulate before any imaging modality will be positive. However, a post-hoc analysis considering only lesions >5 mm found the sensitivity of 18F-DCFPyL PET/CT increased to 60%. Despite this, across all lesions, 18F-DCFPyL PET/CT significantly outperformed conventional CT pelvic imaging with markedly improved specificity (97.7% vs. 65.1%) and positive predictive value (85.1% vs. 28.3%) for pelvic lymph node metastases, as well as sensitivity for disease within the prostate (96.8% vs. 35.9%).
Click to Show/Hide
|
||||
| Half life period | 8 h | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Headache rate |
2%
|
|||
| Patients Enrolled |
208 patients with prostate cancer.
|
||||
| Administration Dosage | 8-10 mCi | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
PSMA, also known as glutamate carboxypeptidase 2 or folate hydrolase 1, is highly expressed in nearly all prostate cancers. Structurally, PSMA is a large extracellular carboxypeptidase domain that is tethered to the cellular membrane by a short transmembrane domain. Physiologically, PSMA is also expressed in normal tissue, including non-malignant prostate tissue, the kidney, the small intestine, and the central nervous system. In most tissues, PSMA likely contributes to folate uptake by cleaving the C-terminal glutamate from folic acid (Figure 1). Within the brain, PSMA regulates the level of N-acetylaspartylglutamate (NAAG) by catalyzing its degradation to glutamate. Structure of PSMA substrates and inhibitors. Left: structure of the PSMA substrate folic acid. The peptide bond (dashed box) linking the P1 glutamate residue (blue) to the remainder of the substrate (P1, red) is cleaved by PSMA. Right: Structure of the PSMA-PET agents 18F-DCFPyL and 68Ga-PSMA-11. The scissile peptide bond has been replaced by a non-hydrolyzable urea motif (red) to generate PMSA inhibitors. The P1 glutamate residue is essential for binding to PSMA, but a wide degree of variability is tolerated in the opposing end of the structure.
Click to Show/Hide
|
||||
| Description |
All patients were dosed with 9 mCi of 18F-DCFPyL and PET/CT images were obtained. 18F-DCFPyL avid lesions were detected in ~60% of the patients. To confirm the metastatic nature of these lesions, biopsies were obtained, if possible. If the lesion was not amendable to biopsy, validation is obtained by focused conventional follow-up imaging or biochemical response to external beam radiotherapy. The differentiation between the number of true metastases validated by other techniques (i.e. true positives, TP) and false positives (FP) allows for the calculation of the correct localization rate (CLR), where the CLR = TP/(TP + FP) * 100 (essentially, PPV with the added requirement of anatomic co-localization). Across all patients, the CLR ranged from 85% to 87% between readers, however the CLR improved as the baseline PSA increased, from 75% to 96% (PSA < 1.0 to PSA ≥ 5.0, respectively). The PPV of the 18F-DCFPyL PET/CT was found to vary by anatomic region and was highest for prostatic lesions (80%) and pelvic lymph nodes (71%), but lower for extrapelvic visceral/soft tissue masses (29%).
Click to Show/Hide
|
||||
| Half life period | 8 h | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Hypertension |
1%
|
|||
| Patients Enrolled |
208 patients with prostate cancer.
|
||||
| Administration Dosage | 8-10 mCi | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
PSMA, also known as glutamate carboxypeptidase 2 or folate hydrolase 1, is highly expressed in nearly all prostate cancers. Structurally, PSMA is a large extracellular carboxypeptidase domain that is tethered to the cellular membrane by a short transmembrane domain. Physiologically, PSMA is also expressed in normal tissue, including non-malignant prostate tissue, the kidney, the small intestine, and the central nervous system. In most tissues, PSMA likely contributes to folate uptake by cleaving the C-terminal glutamate from folic acid (Figure 1). Within the brain, PSMA regulates the level of N-acetylaspartylglutamate (NAAG) by catalyzing its degradation to glutamate. Structure of PSMA substrates and inhibitors. Left: structure of the PSMA substrate folic acid. The peptide bond (dashed box) linking the P1 glutamate residue (blue) to the remainder of the substrate (P1, red) is cleaved by PSMA. Right: Structure of the PSMA-PET agents 18F-DCFPyL and 68Ga-PSMA-11. The scissile peptide bond has been replaced by a non-hydrolyzable urea motif (red) to generate PMSA inhibitors. The P1 glutamate residue is essential for binding to PSMA, but a wide degree of variability is tolerated in the opposing end of the structure.
Click to Show/Hide
|
||||
| Description |
All patients were dosed with 9 mCi of 18F-DCFPyL and PET/CT images were obtained. 18F-DCFPyL avid lesions were detected in ~60% of the patients. To confirm the metastatic nature of these lesions, biopsies were obtained, if possible. If the lesion was not amendable to biopsy, validation is obtained by focused conventional follow-up imaging or biochemical response to external beam radiotherapy. The differentiation between the number of true metastases validated by other techniques (i.e. true positives, TP) and false positives (FP) allows for the calculation of the correct localization rate (CLR), where the CLR = TP/(TP + FP) * 100 (essentially, PPV with the added requirement of anatomic co-localization). Across all patients, the CLR ranged from 85% to 87% between readers, however the CLR improved as the baseline PSA increased, from 75% to 96% (PSA < 1.0 to PSA ≥ 5.0, respectively). The PPV of the 18F-DCFPyL PET/CT was found to vary by anatomic region and was highest for prostatic lesions (80%) and pelvic lymph nodes (71%), but lower for extrapelvic visceral/soft tissue masses (29%).
Click to Show/Hide
|
||||
| Half life period | 8 h | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Negative predictive value |
83.00%
|
|||
| Patients Enrolled |
252 patients with prostate cancer.
|
||||
| Administration Dosage | 8-10 mCi | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
PSMA, also known as glutamate carboxypeptidase 2 or folate hydrolase 1, is highly expressed in nearly all prostate cancers. Structurally, PSMA is a large extracellular carboxypeptidase domain that is tethered to the cellular membrane by a short transmembrane domain. Physiologically, PSMA is also expressed in normal tissue, including non-malignant prostate tissue, the kidney, the small intestine, and the central nervous system. In most tissues, PSMA likely contributes to folate uptake by cleaving the C-terminal glutamate from folic acid (Figure 1). Within the brain, PSMA regulates the level of N-acetylaspartylglutamate (NAAG) by catalyzing its degradation to glutamate. Structure of PSMA substrates and inhibitors. Left: structure of the PSMA substrate folic acid. The peptide bond (dashed box) linking the P1 glutamate residue (blue) to the remainder of the substrate (P1, red) is cleaved by PSMA. Right: Structure of the PSMA-PET agents 18F-DCFPyL and 68Ga-PSMA-11. The scissile peptide bond has been replaced by a non-hydrolyzable urea motif (red) to generate PMSA inhibitors. The P1 glutamate residue is essential for binding to PSMA, but a wide degree of variability is tolerated in the opposing end of the structure.
Click to Show/Hide
|
||||
| Description |
Within cohort A, the primary end point was the sensitivity and specificity for detection of metastases to the pelvic lymph nodes by 18F-DCFPyL PET/CT. These findings were validated by histopathology after radical proctectomy and lymph node dissection (Figure 6). When all confirmed positive lymph node metastases were considered, the sensitivity of 18F-DCFPyL PET/CT was 40%, with a 98% specificity. It is perhaps unsurprising that histopathology is more sensitivity for the detection of early metastases as a sufficient number of malignant cells must accumulate before any imaging modality will be positive. However, a post-hoc analysis considering only lesions >5 mm found the sensitivity of 18F-DCFPyL PET/CT increased to 60%. Despite this, across all lesions, 18F-DCFPyL PET/CT significantly outperformed conventional CT pelvic imaging with markedly improved specificity (97.7% vs. 65.1%) and positive predictive value (85.1% vs. 28.3%) for pelvic lymph node metastases, as well as sensitivity for disease within the prostate (96.8% vs. 35.9%).
Click to Show/Hide
|
||||
| Half life period | 8 h | ||||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Sensitivity |
40%
|
|||
| Patients Enrolled |
252 patients with prostate cancer.
|
||||
| Administration Dosage | 8-10 mCi | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
PSMA, also known as glutamate carboxypeptidase 2 or folate hydrolase 1, is highly expressed in nearly all prostate cancers. Structurally, PSMA is a large extracellular carboxypeptidase domain that is tethered to the cellular membrane by a short transmembrane domain. Physiologically, PSMA is also expressed in normal tissue, including non-malignant prostate tissue, the kidney, the small intestine, and the central nervous system. In most tissues, PSMA likely contributes to folate uptake by cleaving the C-terminal glutamate from folic acid (Figure 1). Within the brain, PSMA regulates the level of N-acetylaspartylglutamate (NAAG) by catalyzing its degradation to glutamate. Structure of PSMA substrates and inhibitors. Left: structure of the PSMA substrate folic acid. The peptide bond (dashed box) linking the P1 glutamate residue (blue) to the remainder of the substrate (P1, red) is cleaved by PSMA. Right: Structure of the PSMA-PET agents 18F-DCFPyL and 68Ga-PSMA-11. The scissile peptide bond has been replaced by a non-hydrolyzable urea motif (red) to generate PMSA inhibitors. The P1 glutamate residue is essential for binding to PSMA, but a wide degree of variability is tolerated in the opposing end of the structure.
Click to Show/Hide
|
||||
| Description |
Within cohort A, the primary end point was the sensitivity and specificity for detection of metastases to the pelvic lymph nodes by 18F-DCFPyL PET/CT. These findings were validated by histopathology after radical proctectomy and lymph node dissection (Figure 6). When all confirmed positive lymph node metastases were considered, the sensitivity of 18F-DCFPyL PET/CT was 40%, with a 98% specificity. It is perhaps unsurprising that histopathology is more sensitivity for the detection of early metastases as a sufficient number of malignant cells must accumulate before any imaging modality will be positive. However, a post-hoc analysis considering only lesions >5 mm found the sensitivity of 18F-DCFPyL PET/CT increased to 60%. Despite this, across all lesions, 18F-DCFPyL PET/CT significantly outperformed conventional CT pelvic imaging with markedly improved specificity (97.7% vs. 65.1%) and positive predictive value (85.1% vs. 28.3%) for pelvic lymph node metastases, as well as sensitivity for disease within the prostate (96.8% vs. 35.9%).
Click to Show/Hide
|
||||
| Half life period | 8 h | ||||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Sensitivity |
86%
|
|||
| Patients Enrolled |
208 patients with prostate cancer.
|
||||
| Administration Dosage | 8-10 mCi | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
PSMA, also known as glutamate carboxypeptidase 2 or folate hydrolase 1, is highly expressed in nearly all prostate cancers. Structurally, PSMA is a large extracellular carboxypeptidase domain that is tethered to the cellular membrane by a short transmembrane domain. Physiologically, PSMA is also expressed in normal tissue, including non-malignant prostate tissue, the kidney, the small intestine, and the central nervous system. In most tissues, PSMA likely contributes to folate uptake by cleaving the C-terminal glutamate from folic acid (Figure 1). Within the brain, PSMA regulates the level of N-acetylaspartylglutamate (NAAG) by catalyzing its degradation to glutamate. Structure of PSMA substrates and inhibitors. Left: structure of the PSMA substrate folic acid. The peptide bond (dashed box) linking the P1 glutamate residue (blue) to the remainder of the substrate (P1, red) is cleaved by PSMA. Right: Structure of the PSMA-PET agents 18F-DCFPyL and 68Ga-PSMA-11. The scissile peptide bond has been replaced by a non-hydrolyzable urea motif (red) to generate PMSA inhibitors. The P1 glutamate residue is essential for binding to PSMA, but a wide degree of variability is tolerated in the opposing end of the structure.
Click to Show/Hide
|
||||
| Description |
All patients were dosed with 9 mCi of 18F-DCFPyL and PET/CT images were obtained. 18F-DCFPyL avid lesions were detected in ~60% of the patients. To confirm the metastatic nature of these lesions, biopsies were obtained, if possible. If the lesion was not amendable to biopsy, validation is obtained by focused conventional follow-up imaging or biochemical response to external beam radiotherapy. The differentiation between the number of true metastases validated by other techniques (i.e. true positives, TP) and false positives (FP) allows for the calculation of the correct localization rate (CLR), where the CLR = TP/(TP + FP) * 100 (essentially, PPV with the added requirement of anatomic co-localization). Across all patients, the CLR ranged from 85% to 87% between readers, however the CLR improved as the baseline PSA increased, from 75% to 96% (PSA < 1.0 to PSA ≥ 5.0, respectively). The PPV of the 18F-DCFPyL PET/CT was found to vary by anatomic region and was highest for prostatic lesions (80%) and pelvic lymph nodes (71%), but lower for extrapelvic visceral/soft tissue masses (29%).
Click to Show/Hide
|
||||
| Half life period | 8 h | ||||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Sensitivity |
96%
|
|||
| Patients Enrolled |
93 patients with prostate cancer.
|
||||
| Administration Dosage | 8-10 mCi | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
PSMA, also known as glutamate carboxypeptidase 2 or folate hydrolase 1, is highly expressed in nearly all prostate cancers. Structurally, PSMA is a large extracellular carboxypeptidase domain that is tethered to the cellular membrane by a short transmembrane domain. Physiologically, PSMA is also expressed in normal tissue, including non-malignant prostate tissue, the kidney, the small intestine, and the central nervous system. In most tissues, PSMA likely contributes to folate uptake by cleaving the C-terminal glutamate from folic acid (Figure 1). Within the brain, PSMA regulates the level of N-acetylaspartylglutamate (NAAG) by catalyzing its degradation to glutamate. Structure of PSMA substrates and inhibitors. Left: structure of the PSMA substrate folic acid. The peptide bond (dashed box) linking the P1 glutamate residue (blue) to the remainder of the substrate (P1, red) is cleaved by PSMA. Right: Structure of the PSMA-PET agents 18F-DCFPyL and 68Ga-PSMA-11. The scissile peptide bond has been replaced by a non-hydrolyzable urea motif (red) to generate PMSA inhibitors. The P1 glutamate residue is essential for binding to PSMA, but a wide degree of variability is tolerated in the opposing end of the structure.
Click to Show/Hide
|
||||
| Description |
Within cohort A, the primary end point was the sensitivity and specificity for detection of metastases to the pelvic lymph nodes by 18F-DCFPyL PET/CT. These findings were validated by histopathology after radical proctectomy and lymph node dissection (Figure 6). When all confirmed positive lymph node metastases were considered, the sensitivity of 18F-DCFPyL PET/CT was 40%, with a 98% specificity. It is perhaps unsurprising that histopathology is more sensitivity for the detection of early metastases as a sufficient number of malignant cells must accumulate before any imaging modality will be positive. However, a post-hoc analysis considering only lesions >5 mm found the sensitivity of 18F-DCFPyL PET/CT increased to 60%. Despite this, across all lesions, 18F-DCFPyL PET/CT significantly outperformed conventional CT pelvic imaging with markedly improved specificity (97.7% vs. 65.1%) and positive predictive value (85.1% vs. 28.3%) for pelvic lymph node metastases, as well as sensitivity for disease within the prostate (96.8% vs. 35.9%).
Click to Show/Hide
|
||||
| Half life period | 8 h | ||||
| Experiment 11 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Specificity |
98%
|
|||
| Patients Enrolled |
252 patients with prostate cancer.
|
||||
| Administration Dosage | 8-10 mCi | ||||
| Evaluation Method | PET/CT | ||||
| MOA of PDC |
PSMA, also known as glutamate carboxypeptidase 2 or folate hydrolase 1, is highly expressed in nearly all prostate cancers. Structurally, PSMA is a large extracellular carboxypeptidase domain that is tethered to the cellular membrane by a short transmembrane domain. Physiologically, PSMA is also expressed in normal tissue, including non-malignant prostate tissue, the kidney, the small intestine, and the central nervous system. In most tissues, PSMA likely contributes to folate uptake by cleaving the C-terminal glutamate from folic acid (Figure 1). Within the brain, PSMA regulates the level of N-acetylaspartylglutamate (NAAG) by catalyzing its degradation to glutamate. Structure of PSMA substrates and inhibitors. Left: structure of the PSMA substrate folic acid. The peptide bond (dashed box) linking the P1 glutamate residue (blue) to the remainder of the substrate (P1, red) is cleaved by PSMA. Right: Structure of the PSMA-PET agents 18F-DCFPyL and 68Ga-PSMA-11. The scissile peptide bond has been replaced by a non-hydrolyzable urea motif (red) to generate PMSA inhibitors. The P1 glutamate residue is essential for binding to PSMA, but a wide degree of variability is tolerated in the opposing end of the structure.
Click to Show/Hide
|
||||
| Description |
Within cohort A, the primary end point was the sensitivity and specificity for detection of metastases to the pelvic lymph nodes by 18F-DCFPyL PET/CT. These findings were validated by histopathology after radical proctectomy and lymph node dissection (Figure 6). When all confirmed positive lymph node metastases were considered, the sensitivity of 18F-DCFPyL PET/CT was 40%, with a 98% specificity. It is perhaps unsurprising that histopathology is more sensitivity for the detection of early metastases as a sufficient number of malignant cells must accumulate before any imaging modality will be positive. However, a post-hoc analysis considering only lesions >5 mm found the sensitivity of 18F-DCFPyL PET/CT increased to 60%. Despite this, across all lesions, 18F-DCFPyL PET/CT significantly outperformed conventional CT pelvic imaging with markedly improved specificity (97.7% vs. 65.1%) and positive predictive value (85.1% vs. 28.3%) for pelvic lymph node metastases, as well as sensitivity for disease within the prostate (96.8% vs. 35.9%).
Click to Show/Hide
|
||||
| Half life period | 8 h | ||||
References
