Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_02029
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
LLC2B-Mal-DM1
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
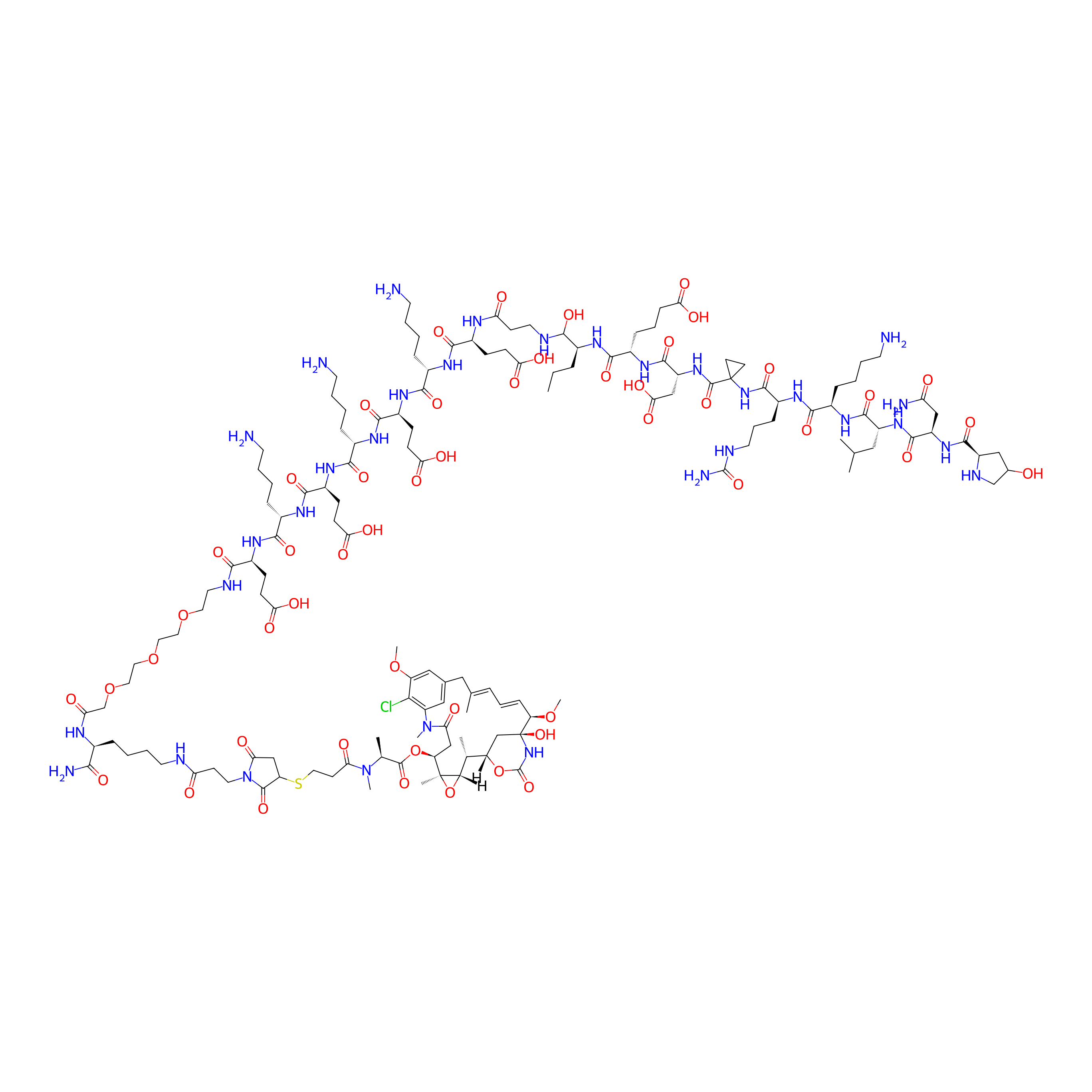
|
|||||
| Peptide Name |
LLC2B
|
Peptide Info | ||||
| Drug Name |
Mertansine DM1
|
Drug Info | ||||
| Therapeutic Target |
Microtubule (MT)
|
Target Info | ||||
| Linker Name |
Thiol group (SH) and maleimide
|
Linker Info | ||||
| Peptide Modified Type |
Amino acid modifications
|
|||||
| Modified Segment |
Incorporation of unnatural amino acids
|
|||||
| Formula |
C143H229ClN32O50S
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 3264.099 | ||||
| Lipid-water partition coefficient (xlogp) | -8.0902 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 38 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 51 | |||||
| Rotatable Bond Count (rotbonds) | 109 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
28.66%
|
|||
| Administration Time | 6 d | ||||
| Administration Dosage | 4.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
34.15%
|
|||
| Administration Time | 6 d | ||||
| Administration Dosage | 2.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
36.81%
|
|||
| Administration Time | 12 d | ||||
| Administration Dosage | 4.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
37.21%
|
|||
| Administration Time | 2 d | ||||
| Administration Dosage | 4.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
38.55%
|
|||
| Administration Time | 10 d | ||||
| Administration Dosage | 4.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
38.73%
|
|||
| Administration Time | 8 d | ||||
| Administration Dosage | 4.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
39.16%
|
|||
| Administration Time | Once a week for 3 weeks | ||||
| Administration Dosage | 4.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
44.93%
|
|||
| Administration Time | 8 d | ||||
| Administration Dosage | 2.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
46.77%
|
|||
| Administration Time | 18 d | ||||
| Administration Dosage | 1.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
47.60%
|
|||
| Administration Time | 16 d | ||||
| Administration Dosage | 4.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
47.63%
|
|||
| Administration Time | Once a week for 3 weeks | ||||
| Administration Dosage | 2.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
48.73%
|
|||
| Administration Time | 16 d | ||||
| Administration Dosage | 2.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 13 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
49.13%
|
|||
| Administration Time | 14 d | ||||
| Administration Dosage | 4.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 14 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
49.33%
|
|||
| Administration Time | 2 d | ||||
| Administration Dosage | 2.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 15 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
50.36%
|
|||
| Administration Time | 18 d | ||||
| Administration Dosage | 4.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 16 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
50.81%
|
|||
| Administration Time | 12 d | ||||
| Administration Dosage | 2.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 17 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
51.89%
|
|||
| Administration Time | 18 d | ||||
| Administration Dosage | 2.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 18 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
53.10%
|
|||
| Administration Time | 14 d | ||||
| Administration Dosage | 2.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 19 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
54.75%
|
|||
| Administration Time | 12 d | ||||
| Administration Dosage | 1.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 20 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
54.77%
|
|||
| Administration Time | 16 d | ||||
| Administration Dosage | 1.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 21 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
57.18%
|
|||
| Administration Time | 10 d | ||||
| Administration Dosage | 1.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 22 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
57.94%
|
|||
| Administration Time | 10 d | ||||
| Administration Dosage | 2.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 23 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
59.26%
|
|||
| Administration Time | 14 d | ||||
| Administration Dosage | 1.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 24 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
60.37%
|
|||
| Administration Time | 8 d | ||||
| Administration Dosage | 1.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 25 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
63.86%
|
|||
| Administration Time | 6 d | ||||
| Administration Dosage | 1.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 26 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
65.78%
|
|||
| Administration Time | Once a week for 3 weeks | ||||
| Administration Dosage | 1.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 27 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor growth inhibition value (TGI) |
71.61%
|
|||
| Administration Time | 2 d | ||||
| Administration Dosage | 1.0 mg DM1 equiv/kg | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
We investigated the antitumor efficacy of those two PDCs on xenografted mice in vivo. LLC2B-Mal-DM1 at 1.0, 2.0 and 4.0 mg DM1 equiv./kg per week was injected into individual mice according to the tolerated concentrations of DM1 in mice: 1.0-1.5 mg/kg once per week. However, LLC2B-SS-DM1 at 0.5, 1.0, 2.0 and 4.0 mg DM1 equiv./kg was injected in the test due to its higher suppressive effect compared to LLC2B-Mal-DM in the clone formation experiment (the drug concentration used was based on the amount of the substance DM1). As shown in Figure 6A,C, LLC2B-Mal-DM1 at 2.0 and 4.0 mg DM1 equiv./kg and LLC2B-SS-DM1 with 0.5 mg DM1 equiv./kg had clear antitumor effects compared to the DM1 control groups. According to the mouse body weights, there were probably no acute life-threatening conditions in any of the groups. Moreover, the comparison of both PDCs indicated that LLC2B-SS-DM1 had a better effect than LLC2B-Mal-DM1 against the tumors.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice MCF-7 cells xenograft model. | ||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Completely suppress concentration |
1 μg DM1 equiv./mL
|
|||
| Administration Time | 12 h | ||||
| Evaluation Method | Clone formation experiment | ||||
| MOA of PDC |
A maytansin derivative, DM1, is a promising therapeutic compound for treating tumors, but is also a highly poisonous substance with various side effects. For clinical expansion, we tried to develop novel peptide-drug conjugates (PDCs) with DM1. In the study, a one-bead one-compound (OBOC) platform was used to screen and identify a novel, highly stable, non-natural amino acid peptide targeting the tyrosine receptor FGFR2. Then, the identified peptide, named LLC2B, was conjugated with the cytotoxin DM1. Our results show that LLC2B has high affinity for the FGFR2 protein according to an isothermal titration calorimetry (ITC) test. LLC2B-Cy5.5 binding to FGFR2-positive cancer cells was confirmed by fluorescent microscopic imaging and flow cytometry in vitro. Using xenografted nude mouse models established with breast cancer MCF-7 cells and esophageal squamous cell carcinoma KYSE180 cells, respectively, LLC2B-Cy5.5 was observed to specifically target tumor tissues 24 h after tail vein injection. Incubation assays, both in aqueous solution at room temperature and in human plasma at 37 °C, suggested that LLC2B has high stability and strong anti-proteolytic ability. Then, we used two different linkers, one of molecular disulfide bonds and another of a maleimide group, to couple LLC2B to the toxin DM1. The novel peptide-drug conjugates (PDCs) inhibited tumor growth and significantly increased the maximum tolerated dose of DM1 in xenografted mice. In brief, our results suggest that LLC2B-DM1 can be developed into a potential PDC for tumor treatment in the future.
Click to Show/Hide
|
||||
| Description |
The results show that DM1 could completely suppress the clone formation of MCF-7 cells at 0.001 μg/mL, while LLC2B-SS-DM1 could completely suppress the clone formation of MCF-7 cells at 0.1 μg DM1 equiv./mL. A concentration of 1 μg DM1 equiv./mL was required for LLC2B-Mal-DM1 to achieve the same suppression effect.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
References
