Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00122
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
c(RGDfK)
|
|||||
| Structure |
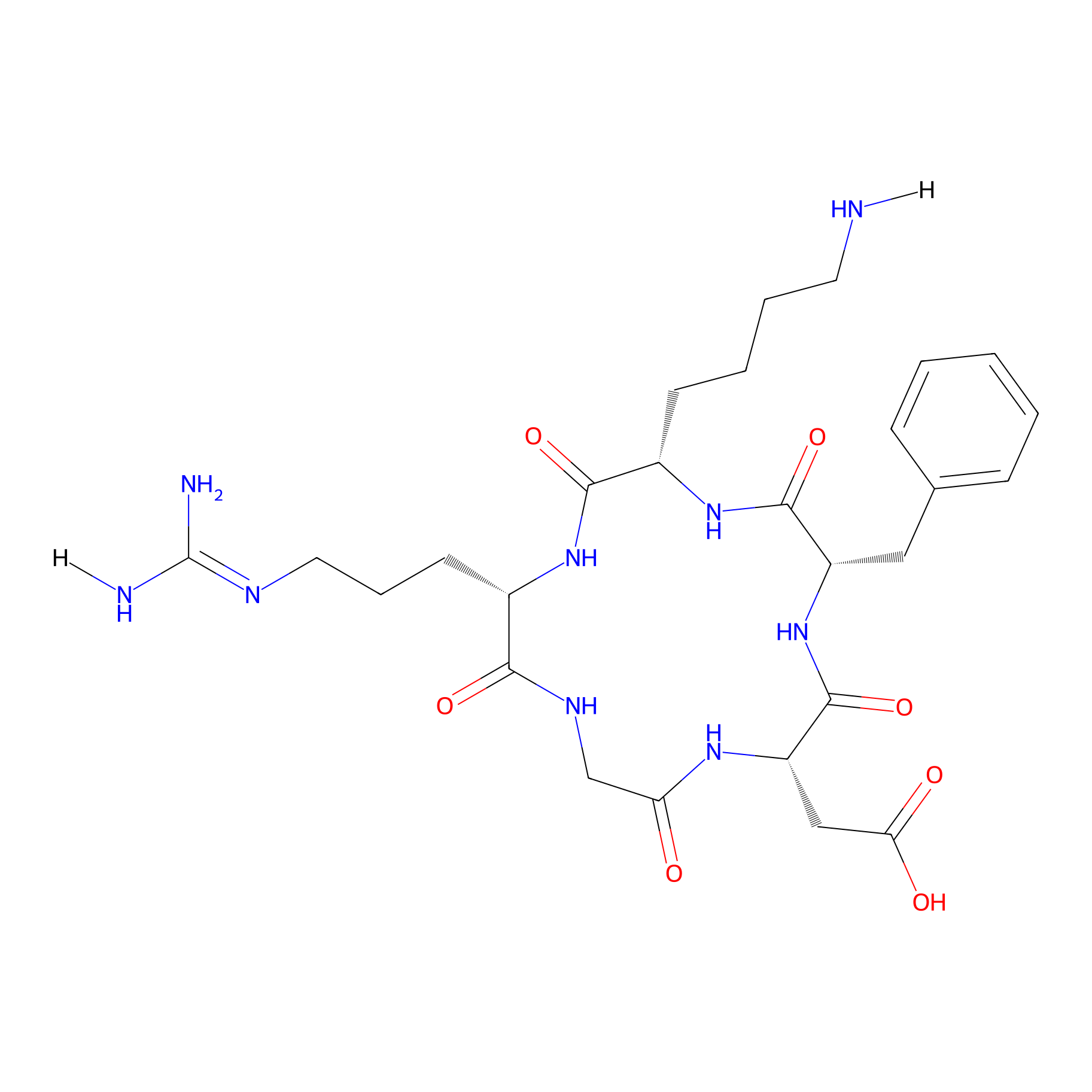
|
|||||
| Sequence |
RGDFK
|
|||||
| Peptide Type |
Cyclic
|
|||||
| Receptor Name |
Integrin alpha-V; Integrin beta-3 (ITGAV; ITGB3)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C27H41N9O7
|
|||||
| Isosmiles |
[H]NCCCC[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CC(=O)O)NC(=O)CNC(=O)[C@H](CCC/N=C(\N)N[H])NC1=O
|
|||||
| InChI |
InChI=1S/C27H41N9O7/c28-11-5-4-9-18-24(41)34-17(10-6-12-31-27(29)30)23(40)32-15-21(37)33-20(14-22(38)39)26(43)36-19(25(42)35-18)13-16-7-2-1-3-8-16/h1-3,7-8,17-20H,4-6,9-15,28H2,(H,32,40)(H,33,37)(H,34,41)(H,35,42)(H,36,43)(H,38,39)(H4,29,30,31)/t17-,18-,19-,20-/m0/s1
|
|||||
| InChIKey |
NVHPXYIRNJFKTE-MUGJNUQGSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
603.681
|
Polar area
|
273.22
|
||
|
Complexity
|
603.3128947
|
xlogp Value
|
-3.0446
|
|||
|
Heavy Count
|
43
|
Rot Bonds
|
13
|
|||
|
Hbond acc
|
8
|
Hbond Donor
|
9
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
99mTc-3PRGD2 [Phase 3]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Primary malignant lung tumors | ||||
| Efficacy Data | Specificity |
88.24%
|
|||
| Patients Enrolled |
26 patients with primary malignant lung tumors.
|
||||
| Administration Time | 40-50 minutes | ||||
| Administration Dosage | 11.1 MBq (0.3 mCi)/kg | ||||
| MOA of PDC |
The integrin vβ3 receptor is a transmembrane heterodimer that mediates cell-cell and cell-extracellular matrix adhesions. It is ubiquitously present during the development and progression of malignant tumors and is closely associated with angiogenesis and metastasis. This receptor is highly expressed in proliferating tumor cells and activated endothelial cells, but is either not expressed or expressed at very low levels in normal endothelial cells, dormant vascular cells, and other normal cells, exhibiting a certain specificity. 99mtechnetium-three polyethylene glycol spacers-arginine-glycine-aspartic acid (99mTc-3PRGD2) is a single-photon emission computed tomography (SPECT) imaging agent that targets the integrin vβ33 receptor. Previous studies have confirmed the feasibility of 99mTc-3PRGD2 SPECT imaging for the diagnosis of lung cancer, breast cancer, esophageal cancer, and other malignant tumors and their related lymph node metastases. This study aimed to further investigate the performance of 99mTc-3PRGD2 SPECT/CT imaging in the diagnosis of lymph node metastasis in primary malignant lung tumors by comparing it with 18F-FDG PET/CT imaging.
Click to Show/Hide
|
||||
| Description |
Total 42 stations had metastatic lymph nodes and 136 stations had benign lymph nodes. The differences between metastatic and benign lymph nodes in the visual qualitative and semiquantitative analyses of 99mTc-3PRGD2 SPECT/CT and 18F-FDG PET/CT were statistically significant (all P < 0.001). The area under the receiver operating characteristic curve (AUC) in the semi-quantitative analysis of 99mTc-3PRGD2 SPECT/CT was 0.908 (95% confidence interval [CI], 0.851-0.966), and the sensitivity, specificity, positive predictive value, and negative predictive value were 0.86 (36/42), 0.88 (120/136), 0.69 (36/52), and 0.95 (120/126), respectively. Among the 26 patients (including two patients each with two lung tumors), 15 had pathologically confirmed lymph node metastasis. The difference between primary lung lesions in patients with and without lymph node metastasis was statistically significant only in the semi-quantitative analysis of 99mTc-3PRGD2 SPECT/CT (P = 0.007), with an AUC of 0.807 (95% CI, 0.641-0.974).
Click to Show/Hide
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Primary malignant lung tumors | ||||
| Efficacy Data | Sensitivity |
85.71%
|
|||
| Patients Enrolled |
26 patients with primary malignant lung tumors.
|
||||
| Administration Time | 40-50 minutes | ||||
| Administration Dosage | 11.1 MBq (0.3 mCi)/kg | ||||
| MOA of PDC |
The integrin vβ3 receptor is a transmembrane heterodimer that mediates cell-cell and cell-extracellular matrix adhesions. It is ubiquitously present during the development and progression of malignant tumors and is closely associated with angiogenesis and metastasis. This receptor is highly expressed in proliferating tumor cells and activated endothelial cells, but is either not expressed or expressed at very low levels in normal endothelial cells, dormant vascular cells, and other normal cells, exhibiting a certain specificity. 99mtechnetium-three polyethylene glycol spacers-arginine-glycine-aspartic acid (99mTc-3PRGD2) is a single-photon emission computed tomography (SPECT) imaging agent that targets the integrin vβ33 receptor. Previous studies have confirmed the feasibility of 99mTc-3PRGD2 SPECT imaging for the diagnosis of lung cancer, breast cancer, esophageal cancer, and other malignant tumors and their related lymph node metastases. This study aimed to further investigate the performance of 99mTc-3PRGD2 SPECT/CT imaging in the diagnosis of lymph node metastasis in primary malignant lung tumors by comparing it with 18F-FDG PET/CT imaging.
Click to Show/Hide
|
||||
| Description |
Total 42 stations had metastatic lymph nodes and 136 stations had benign lymph nodes. The differences between metastatic and benign lymph nodes in the visual qualitative and semiquantitative analyses of 99mTc-3PRGD2 SPECT/CT and 18F-FDG PET/CT were statistically significant (all P < 0.001). The area under the receiver operating characteristic curve (AUC) in the semi-quantitative analysis of 99mTc-3PRGD2 SPECT/CT was 0.908 (95% confidence interval [CI], 0.851-0.966), and the sensitivity, specificity, positive predictive value, and negative predictive value were 0.86 (36/42), 0.88 (120/136), 0.69 (36/52), and 0.95 (120/126), respectively. Among the 26 patients (including two patients each with two lung tumors), 15 had pathologically confirmed lymph node metastasis. The difference between primary lung lesions in patients with and without lymph node metastasis was statistically significant only in the semi-quantitative analysis of 99mTc-3PRGD2 SPECT/CT (P = 0.007), with an AUC of 0.807 (95% CI, 0.641-0.974).
Click to Show/Hide
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Primary malignant lung tumors | ||||
| Efficacy Data | Area under the curve (AUC) |
0.908
|
|||
| Patients Enrolled |
26 patients with primary malignant lung tumors.
|
||||
| Administration Time | 40-50 minutes | ||||
| Administration Dosage | 11.1 MBq (0.3 mCi)/kg | ||||
| MOA of PDC |
The integrin vβ3 receptor is a transmembrane heterodimer that mediates cell-cell and cell-extracellular matrix adhesions. It is ubiquitously present during the development and progression of malignant tumors and is closely associated with angiogenesis and metastasis. This receptor is highly expressed in proliferating tumor cells and activated endothelial cells, but is either not expressed or expressed at very low levels in normal endothelial cells, dormant vascular cells, and other normal cells, exhibiting a certain specificity. 99mtechnetium-three polyethylene glycol spacers-arginine-glycine-aspartic acid (99mTc-3PRGD2) is a single-photon emission computed tomography (SPECT) imaging agent that targets the integrin vβ33 receptor. Previous studies have confirmed the feasibility of 99mTc-3PRGD2 SPECT imaging for the diagnosis of lung cancer, breast cancer, esophageal cancer, and other malignant tumors and their related lymph node metastases. This study aimed to further investigate the performance of 99mTc-3PRGD2 SPECT/CT imaging in the diagnosis of lymph node metastasis in primary malignant lung tumors by comparing it with 18F-FDG PET/CT imaging.
Click to Show/Hide
|
||||
| Description |
Total 42 stations had metastatic lymph nodes and 136 stations had benign lymph nodes. The differences between metastatic and benign lymph nodes in the visual qualitative and semiquantitative analyses of 99mTc-3PRGD2 SPECT/CT and 18F-FDG PET/CT were statistically significant (all P < 0.001). The area under the receiver operating characteristic curve (AUC) in the semi-quantitative analysis of 99mTc-3PRGD2 SPECT/CT was 0.908 (95% confidence interval [CI], 0.851-0.966), and the sensitivity, specificity, positive predictive value, and negative predictive value were 0.86 (36/42), 0.88 (120/136), 0.69 (36/52), and 0.95 (120/126), respectively. Among the 26 patients (including two patients each with two lung tumors), 15 had pathologically confirmed lymph node metastasis. The difference between primary lung lesions in patients with and without lymph node metastasis was statistically significant only in the semi-quantitative analysis of 99mTc-3PRGD2 SPECT/CT (P = 0.007), with an AUC of 0.807 (95% CI, 0.641-0.974).
Click to Show/Hide
|
||||
BGC-0222 [Phase 1]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values |
9.21%
|
|||
| Administration Time | 32 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values |
9.87%
|
|||
| Administration Time | 29 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values |
16.90%
|
|||
| Administration Time | 25 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values |
27.60%
|
|||
| Administration Time | 22 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 5 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values |
57.00%
|
|||
| Administration Time | 18 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 6 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values |
88.80%
|
|||
| Administration Time | 15 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 7 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values |
100%
|
|||
| Administration Time | 12 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 8 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
90%
|
|||
| Administration Time | 32 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
It was worth noting that, similar with that in HT-29 mice model, as shown in Fig. 3, BGC0222 also exhibited better antitumor effect than irinotecan and NKTR-102 in MIA PaCa-2(B), NCI-H446(C), U-87 MG(D) and MDA-MB-231(E) xenograft models, with lower RTV and T/C values. Moreover, in these model assays, there were also no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 9 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
94%
|
|||
| Administration Time | 40 days | ||||
| Administration Dosage | 60 mg/kg, QW3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
It was worth noting that, similar with that in HT-29 mice model, as shown in Fig. 3, BGC0222 also exhibited better antitumor effect than irinotecan and NKTR-102 in MIA PaCa-2(B), NCI-H446(C), U-87 MG(D) and MDA-MB-231(E) xenograft models, with lower RTV and T/C values. Moreover, in these model assays, there were also no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse U-87MG cells xenograft model. | ||||
| In Vitro Model | Glioblastoma | U-87MG cell | CVCL_0022 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Small cell lung cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
95%
|
|||
| Administration Time | 30 days | ||||
| Administration Dosage | 20 mg/kg, QW3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
It was worth noting that, similar with that in HT-29 mice model, as shown in Fig. 3, BGC0222 also exhibited better antitumor effect than irinotecan and NKTR-102 in MIA PaCa-2(B), NCI-H446(C), U-87 MG(D) and MDA-MB-231(E) xenograft models, with lower RTV and T/C values. Moreover, in these model assays, there were also no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse NCI-H446 cells xenograft model. | ||||
| In Vitro Model | Lung small cell carcinoma | NCI-H446 cell | CVCL_1562 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
98%
|
|||
| Administration Time | 31 days | ||||
| Administration Dosage | 20 mg/kg, QW3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
It was worth noting that, similar with that in HT-29 mice model, as shown in Fig. 3, BGC0222 also exhibited better antitumor effect than irinotecan and NKTR-102 in MIA PaCa-2(B), NCI-H446(C), U-87 MG(D) and MDA-MB-231(E) xenograft models, with lower RTV and T/C values. Moreover, in these model assays, there were also no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse MIA PaCa-2 cells xenograft model. | ||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | MIA PaCa-2 cell | CVCL_0428 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
99%
|
|||
| Administration Time | 36 days | ||||
| Administration Dosage | 20 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
It was worth noting that, similar with that in HT-29 mice model, as shown in Fig. 3, BGC0222 also exhibited better antitumor effect than irinotecan and NKTR-102 in MIA PaCa-2(B), NCI-H446(C), U-87 MG(D) and MDA-MB-231(E) xenograft models, with lower RTV and T/C values. Moreover, in these model assays, there were also no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse MDA-MB-231 cells xenograft model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 13 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) |
0.72
|
|||
| Administration Time | 29 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 14 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) |
0.82
|
|||
| Administration Time | 32 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 15 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) |
0.88
|
|||
| Administration Time | 25 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 16 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) |
1
|
|||
| Administration Time | 12 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 17 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) |
1.15
|
|||
| Administration Time | 22 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 18 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) |
1.47
|
|||
| Administration Time | 15 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 19 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) |
1.56
|
|||
| Administration Time | 18 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.68 ± 0.04 μM
|
|||
| Evaluation Method | Graph Pad Prism 5.0 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
Compared with irinotecan and NKTR-102, compound BGC0222 exhibited better antiproliferative activity in all these assayed cell lines. In the antiproliferative activity assay with the HT29 cell line, compound BGC0222 displayed higher antiproliferative activity than irinotecan (IC50 = 7.64 0.19 μM) and NKTR-102 (IC50 = 2.02 0.10 μM), with IC50 value of 1.83 0.09 μM. In addition, compound BGC0222 showed better antiproliferative activity than irinotecan (IC50 = 16.2 0.22 μM) and NKTR-102 (IC50 = 4.68 0.15 μM) against MIA PaCa-2 cells line, with IC50 value of 3.95 0.16 μM, while compound BGC0222 also exhibited stronger antiproliferative activity than irinotecan (IC50 = 2.69 0.12 μM) and NKTR-102 (IC50 = 0.71 0.05 μM) in MCF-7 cells line assay, with IC50 value of 0.68 0.04 μM, respectively. Clearly, the order of in vitro antiproliferative effect against the assay cell lines was: BGC0222>NKTR-102 > irinotecan. The structure-activity relationship was then investigated. The success of NKTR-102 proved that the introducing of PEG should lead to better antiproliferative activity. In addition, by the comparison of the structure of BGC0222 with that of NKTR-102, it should be found the presence of cRGD lead to better antiproliferative effect. Therefore, based on the above observation, it could be concluded that the introduction of PEG and cRGD to irinotecan should efficiently improve the antiproliferative effect, consistent with our expectation.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.83 ± 0.09 μM
|
|||
| Evaluation Method | Graph Pad Prism 5.0 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
Compared with irinotecan and NKTR-102, compound BGC0222 exhibited better antiproliferative activity in all these assayed cell lines. In the antiproliferative activity assay with the HT29 cell line, compound BGC0222 displayed higher antiproliferative activity than irinotecan (IC50 = 7.64 0.19 μM) and NKTR-102 (IC50 = 2.02 0.10 μM), with IC50 value of 1.83 0.09 μM. In addition, compound BGC0222 showed better antiproliferative activity than irinotecan (IC50 = 16.2 0.22 μM) and NKTR-102 (IC50 = 4.68 0.15 μM) against MIA PaCa-2 cells line, with IC50 value of 3.95 0.16 μM, while compound BGC0222 also exhibited stronger antiproliferative activity than irinotecan (IC50 = 2.69 0.12 μM) and NKTR-102 (IC50 = 0.71 0.05 μM) in MCF-7 cells line assay, with IC50 value of 0.68 0.04 μM, respectively. Clearly, the order of in vitro antiproliferative effect against the assay cell lines was: BGC0222>NKTR-102 > irinotecan. The structure-activity relationship was then investigated. The success of NKTR-102 proved that the introducing of PEG should lead to better antiproliferative activity. In addition, by the comparison of the structure of BGC0222 with that of NKTR-102, it should be found the presence of cRGD lead to better antiproliferative effect. Therefore, based on the above observation, it could be concluded that the introduction of PEG and cRGD to irinotecan should efficiently improve the antiproliferative effect, consistent with our expectation.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
3.95 ± 0.16 μM
|
|||
| Evaluation Method | Graph Pad Prism 5.0 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
Compared with irinotecan and NKTR-102, compound BGC0222 exhibited better antiproliferative activity in all these assayed cell lines. In the antiproliferative activity assay with the HT29 cell line, compound BGC0222 displayed higher antiproliferative activity than irinotecan (IC50 = 7.64 0.19 μM) and NKTR-102 (IC50 = 2.02 0.10 μM), with IC50 value of 1.83 0.09 μM. In addition, compound BGC0222 showed better antiproliferative activity than irinotecan (IC50 = 16.2 0.22 μM) and NKTR-102 (IC50 = 4.68 0.15 μM) against MIA PaCa-2 cells line, with IC50 value of 3.95 0.16 μM, while compound BGC0222 also exhibited stronger antiproliferative activity than irinotecan (IC50 = 2.69 0.12 μM) and NKTR-102 (IC50 = 0.71 0.05 μM) in MCF-7 cells line assay, with IC50 value of 0.68 0.04 μM, respectively. Clearly, the order of in vitro antiproliferative effect against the assay cell lines was: BGC0222>NKTR-102 > irinotecan. The structure-activity relationship was then investigated. The success of NKTR-102 proved that the introducing of PEG should lead to better antiproliferative activity. In addition, by the comparison of the structure of BGC0222 with that of NKTR-102, it should be found the presence of cRGD lead to better antiproliferative effect. Therefore, based on the above observation, it could be concluded that the introduction of PEG and cRGD to irinotecan should efficiently improve the antiproliferative effect, consistent with our expectation.
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | MIA PaCa-2 cell | CVCL_0428 | ||
cRGD-SS-DM1 [Investigative]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumer volume |
1800 mm3
|
|||
| Administration Time | Given every other day for a total of five times | ||||
| Administration Dosage | 400 µg/kg (calculated by free DM1) | ||||
| Description |
It was demonstrated here, RCCD@NPs and RSSD@NPs exhibited significantly better tumor-growth inhibition compared with that of the free DM1, QCCD@NPs or QSSD@NPs (Figure (Figure4A).4A). RSSD@NPs showed the most suppression on B16 tumor up to 25 days, and resulted in a tumor volume 4 times smaller than the saline group at the end of the experiment. The final tumor volumes in various nano-DDS groups ranked from the greatest to the least: QCCD@NPs, QSSD@NPs>DM1>RCCD@NPs>RSSD@NPs. Almost the same trends were found in terms of tumor weight and tumor size (Figures (Figures4B,4B, C).
Click to Show/Hide
|
||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
20.42 ± 4.70 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
21.38 ± 4.32 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Melanoma | B16 cell | CVCL_F936 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
302.00 ± 72.91 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
cRGD-SMCC-DM1 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Uric acid levels |
185
|
|||
| Administration Time | 21d | ||||
| Description |
Remarkably, leucopenia and myelosuppression did not occur during the use of PDC. In addition, the values of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP), as the biomarkers of hepatic function, were all in the normal range in comparison to the saline group (Figure 5F-H). Meanwhile, normal renal function was also identified after treatment by monitoring CREA, UA, and BUN (Figure 5I-K).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumer volume |
1000 mm3
|
|||
| Administration Time | 21d | ||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumer volume |
2100 mm3
|
|||
| Administration Time | Given every other day for a total of five times | ||||
| Administration Dosage | 400 µg/kg (calculated by free DM1) | ||||
| Description |
It was demonstrated here, RCCD@NPs and RSSD@NPs exhibited significantly better tumor-growth inhibition compared with that of the free DM1, QCCD@NPs or QSSD@NPs (Figure (Figure4A).4A). RSSD@NPs showed the most suppression on B16 tumor up to 25 days, and resulted in a tumor volume 4 times smaller than the saline group at the end of the experiment. The final tumor volumes in various nano-DDS groups ranked from the greatest to the least: QCCD@NPs, QSSD@NPs>DM1>RCCD@NPs>RSSD@NPs. Almost the same trends were found in terms of tumor weight and tumor size (Figures (Figures4B,4B, C).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing C57BL/6 mice model. | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | BUN level |
7
|
|||
| Administration Time | 21d | ||||
| Description |
Remarkably, leucopenia and myelosuppression did not occur during the use of PDC. In addition, the values of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP), as the biomarkers of hepatic function, were all in the normal range in comparison to the saline group (Figure 5F-H). Meanwhile, normal renal function was also identified after treatment by monitoring CREA, UA, and BUN (Figure 5I-K).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | AST level |
175
|
|||
| Administration Time | 21d | ||||
| Description |
Remarkably, leucopenia and myelosuppression did not occur during the use of PDC. In addition, the values of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP), as the biomarkers of hepatic function, were all in the normal range in comparison to the saline group (Figure 5F-H). Meanwhile, normal renal function was also identified after treatment by monitoring CREA, UA, and BUN (Figure 5I-K).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | ALT level |
60
|
|||
| Administration Time | 21d | ||||
| Description |
Remarkably, leucopenia and myelosuppression did not occur during the use of PDC. In addition, the values of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP), as the biomarkers of hepatic function, were all in the normal range in comparison to the saline group (Figure 5F-H). Meanwhile, normal renal function was also identified after treatment by monitoring CREA, UA, and BUN (Figure 5I-K).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | ALP level |
80
|
|||
| Administration Time | 21d | ||||
| Description |
Remarkably, leucopenia and myelosuppression did not occur during the use of PDC. In addition, the values of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP), as the biomarkers of hepatic function, were all in the normal range in comparison to the saline group (Figure 5F-H). Meanwhile, normal renal function was also identified after treatment by monitoring CREA, UA, and BUN (Figure 5I-K).
Click to Show/Hide
|
||||
| In Vivo Model | Tumor-bearing nude mice. | ||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
33.84 ± 6.30 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
68.5 nM
|
|||
| Description |
After the MDA-MB-231 cell line was confirmed to be integrin v3-positive by immunofluorescence assay, it was demonstrated that cRGD-SMCC-DM1 had an IC50 value of 68.5 nM to MDA-MB-231 cells.
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
102.33 ± 38.92 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Melanoma | B16 cell | CVCL_F936 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
407.38 ± 54.05 nM
|
|||
| Administration Time | 48 h | ||||
| Description |
The IC50 of QCCD@NPs and QSSD@NPs (389.05±75.25 nM and 245.47±37.54 nM) on B16 cells seemed much higher than the values of RCCD@NPs and RSSD@NPs (102.33±38.92 nM and 21.38±4.32 nM). The cytotoxicity of APDC@NPs on HUVEC showed a similar pattern.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
References
