Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00004
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
[18F]AlF-NOTA-octreotide
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 2 Indication(s)
|
|||||
| Structure |
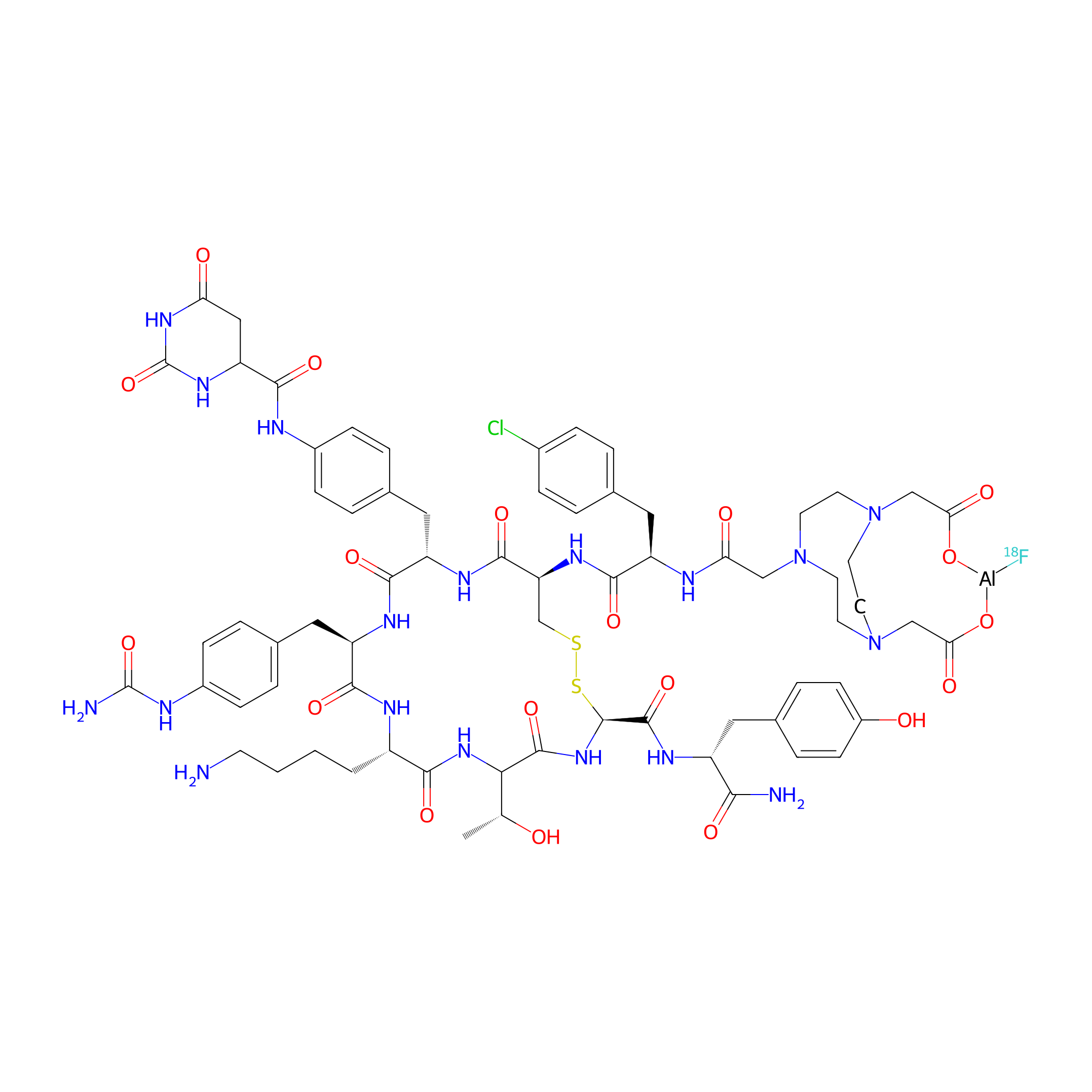
|
|||||
| Peptide Name |
Octreotide
|
Peptide Info | ||||
| Receptor Name |
Somatostatin receptor type 1 (SSTR1)
|
Receptor Info | ||||
| Drug Name |
Fluorine-18
|
Drug Info | ||||
| Therapeutic Target |
Human Deoxyribonucleic acid (hDNA)
|
Target Info | ||||
| Linker Name |
Amide bond
|
Linker Info | ||||
| Peptide Modified Type |
Amino acid modifications; Cyclization modification
|
|||||
| Modified Segment |
D-amino acids
|
|||||
| Ternimal Modification |
N-terminal modification
|
|||||
| Formula |
C69H87AlClFN18O19S2
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 1617.131938 | ||||
| Lipid-water partition coefficient (xlogp) | -3.4566 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 17 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 25 | |||||
| Rotatable Bond Count (rotbonds) | 24 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor-induced osteomalacia | ||||
| Efficacy Data | Accuracy |
88.20%
|
|||
| Patients Enrolled |
17 patients with hypophosphatemic osteomalacia suspected to be TIO.
|
||||
| Administration Dosage | 3.7 MBq (0.10 mCi) per kilogram of body weight | ||||
| Evaluation Method | 18F-OC PET/CT assay | ||||
| MOA of PDC |
18F-OC PET/CT scan is useful in the detection of tumors causing TIO. Further studies with larger patient populations are needed to validate the result.
|
||||
| Description |
The 18F-OC PET/CT scans were positive in 14 patients. Furthermore, 4 of 14 patients were scanned with both 18F-OC and 68Ga-DOTATATE PET/CT. Both studies were able to localize the tumor in all 4 patients. In total, 14 patients had surgery to remove the lesions. Postsurgical pathological examination confirmed causative tumors in these patients, whose symptoms diminished promptly. Serum phosphate levels normalized, confirming the diagnosis of TIO. 18F-OC PET/CT sensitivity, specificity, and accuracy were 87.5%, 100%, and 88.2% respectively. 18F-OC PET/CT findings affected patient management in 88.2% of cases.
Click to Show/Hide
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Neuroendocrine tumour | ||||
| Efficacy Data | Difference in detection ratio |
15.80%
|
|||
| Patients Enrolled |
75 patients with histologically confirmed neuroendocrine tumours and routine clinical.
|
||||
| MOA of PDC |
18F-AlF-OC is noninferior and even superior to 68Ga-DOTATATE/NOC PET in NET patients. This validates 18F-AlF-OC as an option for clinical practice somatostatin receptor PET.
|
||||
| Description |
The resulting mean DDR was 15.8%, with a lower margin of the 95% CI (95% CI, 9.6%-22.0%) higher than -15%, which is the prespecified boundary for noninferiority. The mean DDRs for the 68Ga-DOTATATE and 68Ga-DOTANOC subgroups were 11.8% (95% CI, 4.3-19.3) and 27.5% (95% CI, 17.8-37.1), respectively.
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Neuroendocrine tumour | ||||
| Efficacy Data | Median detection ratio |
91.10%
|
|||
| Patients Enrolled |
75 patients with histologically confirmed neuroendocrine tumours and routine clinical.
|
||||
| MOA of PDC |
18F-AlF-OC is noninferior and even superior to 68Ga-DOTATATE/NOC PET in NET patients. This validates 18F-AlF-OC as an option for clinical practice somatostatin receptor PET.
|
||||
| Description |
In total, 4,709 different tumor lesions were detected: 3,454 with 68Ga-DOTATATE/NOC and 4,278 with 18F-AlF-OC. The mean DR with 18F-AlF-OC was significantly higher than with 68Ga-DOTATATE/NOC (91.1% vs. 75.3%; P < 10-5).
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor-induced osteomalacia | ||||
| Efficacy Data | Sensitivity |
87.50%
|
|||
| Patients Enrolled |
17 patients with hypophosphatemic osteomalacia suspected to be TIO.
|
||||
| Administration Dosage | 3.7 MBq (0.10 mCi) per kilogram of body weight | ||||
| Evaluation Method | 18F-OC PET/CT assay | ||||
| MOA of PDC |
18F-OC PET/CT scan is useful in the detection of tumors causing TIO. Further studies with larger patient populations are needed to validate the result.
|
||||
| Description |
The 18F-OC PET/CT scans were positive in 14 patients. Furthermore, 4 of 14 patients were scanned with both 18F-OC and 68Ga-DOTATATE PET/CT. Both studies were able to localize the tumor in all 4 patients. In total, 14 patients had surgery to remove the lesions. Postsurgical pathological examination confirmed causative tumors in these patients, whose symptoms diminished promptly. Serum phosphate levels normalized, confirming the diagnosis of TIO. 18F-OC PET/CT sensitivity, specificity, and accuracy were 87.5%, 100%, and 88.2% respectively. 18F-OC PET/CT findings affected patient management in 88.2% of cases.
Click to Show/Hide
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor-induced osteomalacia | ||||
| Efficacy Data | Specificity |
100%
|
|||
| Patients Enrolled |
17 patients with hypophosphatemic osteomalacia suspected to be TIO.
|
||||
| Administration Dosage | 3.7 MBq (0.10 mCi) per kilogram of body weight | ||||
| Evaluation Method | 18F-OC PET/CT assay | ||||
| MOA of PDC |
18F-OC PET/CT scan is useful in the detection of tumors causing TIO. Further studies with larger patient populations are needed to validate the result.
|
||||
| Description |
The 18F-OC PET/CT scans were positive in 14 patients. Furthermore, 4 of 14 patients were scanned with both 18F-OC and 68Ga-DOTATATE PET/CT. Both studies were able to localize the tumor in all 4 patients. In total, 14 patients had surgery to remove the lesions. Postsurgical pathological examination confirmed causative tumors in these patients, whose symptoms diminished promptly. Serum phosphate levels normalized, confirming the diagnosis of TIO. 18F-OC PET/CT sensitivity, specificity, and accuracy were 87.5%, 100%, and 88.2% respectively. 18F-OC PET/CT findings affected patient management in 88.2% of cases.
Click to Show/Hide
|
||||
References
